
Atmospheric methane
Encyclopedia
Atmospheric methane levels are of interest due to its impact on climate change
. Atmospheric methane is one of the most potent and influential greenhouse gas
es on Earth. The 100-year global warming potential
of methane is 25, i.e. over a 100-year period, it traps 25 times more heat per mass unit than carbon dioxide
.
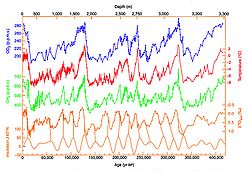
 Early in the Earth's history—about 3.5 billion years ago—there was 1,000 times as much methane in the atmosphere
Early in the Earth's history—about 3.5 billion years ago—there was 1,000 times as much methane in the atmosphere
as there is now. The earliest methane was released into the atmosphere by volcanic activity. During this time, Earth's earliest life appeared. These first, ancient bacteria added to the methane concentration by converting hydrogen and carbon dioxide into methane and water. Oxygen did not become a major part of the atmosphere until photosynthetic organisms evolved later in Earth's history. With no oxygen, methane stayed in the atmosphere longer and at higher concentrations than it does today.
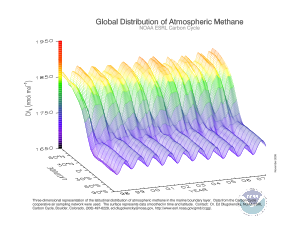
In present times, due to the increase in oxygen, the amount of methane has decreased. The average mole fraction of methane at the Earth's surface in 1998 was 1,745 nmol/mol. Its concentration is higher in the northern hemisphere as most sources (both natural and human) are larger. The concentrations vary seasonally with a minimum in the late summer mainly due to removal by the hydroxyl radical
.
Methane is created near the surface, and it is carried into the stratosphere
by rising air in the tropics
. Uncontrolled build-up of methane in Earth's atmosphere is naturally checked—although human influence can upset this natural regulation—by methane's reaction with hydroxyl radicals formed from singlet oxygen
atoms and with water vapor.
with a global warming potential
of 25 over a 100-year period. This means that a methane emission will have 25 times the impact on temperature of a carbon dioxide emission of the same mass over the following 100 years. Methane has a large effect for a brief period (a net lifetime of 8.4 years in the atmosphere), whereas carbon dioxide has a small effect for a long period (over 100 years). Because of this difference in effect and time period, the global warming potential of methane over a 20 year time period is 72. The Earth's methane concentration has increased by about 150% since 1750, and it accounts for 20% of the total radiative forcing
from all of the long-lived and globally mixed greenhouse gases. Usually, excess methane from landfills and other natural producers of methane are burned so CO2 is released into the atmosphere instead of methane because methane is such a more effective greenhouse gas. Recently methane emitted from coal mines has been successfully converted to electricity.
, glaciers, and ice cores – A source that slowly releases methane trapped in frozen environments as global temperatures rise.
B. Wetlands – Warm temperatures and moist environments are ideal for methane production. Most of the methane makes it past methane-consuming microorganisms.
C. Forest fire – Mass burning of organic matter releases huge amounts of methane into the atmosphere.
D. Rice paddies – The warmer and moister the rice field, the more methane is produced.
E. Animals – Microorganisms breaking down difficult to digest material in the guts of ruminant livestock and termites produce methane that is then released during defecation.
F. Plants – While methane can be consumed in soil before being released into the atmosphere, plants allow for direct travel of methane up through the roots and leaves and into the atmosphere. Plants may also be direct producers of methane.
G. Landfills – Decaying organic matter and anaerobic conditions cause landfills to be a significant source of methane.
H. Waste water treatment facilities
– Anaerobic treatment of organic compounds in the water results in the production of methane.
I. Hydroxyl radical
– OH in the atmosphere is the largest sink for atmospheric methane as well as one of the most significant sources of water vapor in the upper atmosphere.
J. Chlorine radical – Free chlorine in the atmosphere also reacts with methane.
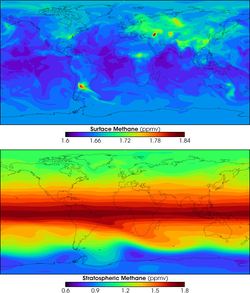
 The balance between sources and sinks is not yet fully understood. The IPCC Working Group 1 stated in chapter 2 of the Fourth Assessment Report that there are "large uncertainties in the current bottom-up estimates of components of the global source", and the balance between sources and sinks is not yet well known. The most important sink in the methane cycle is reaction with the hydroxyl radical, which is produced photochemically in the atmosphere. Production of this radical is not fully understood and has a large effect on atmospheric concentrations. This uncertainty is exemplified by observations that have shown between the year 2000 and 2006 increases in atmospheric concentration of methane ceased without reduction in anthropogenic sources, showing that methane accounting does not accurately predict methane observations.
The balance between sources and sinks is not yet fully understood. The IPCC Working Group 1 stated in chapter 2 of the Fourth Assessment Report that there are "large uncertainties in the current bottom-up estimates of components of the global source", and the balance between sources and sinks is not yet well known. The most important sink in the methane cycle is reaction with the hydroxyl radical, which is produced photochemically in the atmosphere. Production of this radical is not fully understood and has a large effect on atmospheric concentrations. This uncertainty is exemplified by observations that have shown between the year 2000 and 2006 increases in atmospheric concentration of methane ceased without reduction in anthropogenic sources, showing that methane accounting does not accurately predict methane observations.
Houweling et al. (1999) give the following values for methane emissions (Tg/a=teragrams per year):
Any process that results in the production of methane and its release into the atmosphere can be considered a "source." The two main processes that are responsible for methane production occur as a result of microorganisms anaerobic
ally converting organic compounds into methane.
Methanogenesis
, the scientific term for methane production, occurs primarily in anaerobic conditions because of the lack of availability of other oxidants. In these conditions, microscopic organisms called archaea
use acetate and hydrogen to break down essential resources in a process called fermentation
.
Acetoclastic methanogenesis- certain archaea cleave acetate
produced during anaerobic fermentation to yield methane and carbon dioxide.
H3C-COOH → CH4 + CO2
Hydrogenotrophic methanogenesis- archaea oxidize hydrogen with carbon dioxide to yield methane and water.
4H2 + CO2 → CH4 + 2H2O
While acetoclastic methanogenesis and hydrogenotrophic methanogenesis are the two major source reactions for atmospheric methane, other minor biological methane source reactions also occur.
Termite
s also contain methanogenic microorganisms in their gut. However, some of these microorganisms are so unique that they live nowhere else in the world except in the third gut of termites. These microorganisms also break down biotic components to produce ethanol
, as well as methane byproduct. However, unlike ruminants who lose 20 percent of the energy from the plants they eat, termites only lose 2 percent of their energy in the process. Thus comparatively, termites do not have to eat as much food as ruminants to obtain the same amount of energy, and give off proportionally less methane.
However the authors stress "our findings are preliminary with regard to the methane emission strength".
These findings have been called into question in a 2007 paper which found "there is no evidence for substantial aerobic methane emission by terrestrial plants, maximally 0.3% of the previously published values".
While the details of plant methane emissions have yet to be confirmed, plants as a significant methane source would help fill in the gaps of previous global methane budgets as well as explain large plumes of methane that have been observed over the tropics.
In wetlands, where rate of methane production are high, plants help methane travel into the atmosphere—acting like inverted lightning rods as they direct the gas up through the soil and into the air. They are also suspected to produce methane themselves, but because the plants would have to use aerobic conditions to produce methane, the process itself is still unidentified.
. An unknown, but possibly very large quantity of methane is trapped in this form in ocean sediments. The release of large volumes of methane gas from such sediments into the atmosphere has been suggested as a possible cause for rapid global warming
events in the Earth's distant past, such as the Paleocene–Eocene Thermal Maximum of 55 million years ago, and the Great Dying.
Theories suggest that should global warming cause them to heat up sufficiently, all of this methane gas could again be released into the atmosphere. Since methane gas is twenty-five times stronger (for a given weight, averaged over 100 years) than as a greenhouse gas; this would immensely magnify the greenhouse effect.
– land that is frozen for several years at a time – is slowly released from bogs as the permafrost melts. With rising global temperatures, the amount of permafrost melting and releasing methane continues to increase.
Although records of permafrost are limited, recent years (1999 to 2007) have seen record thawing of permafrost in Alaska
and Siberia
. Recent measurements in Siberia show that the methane released is five times greater than previously estimated. Melting yedoma
, a type of permafrost, is a significant source of atmospheric methane (about 4 Tg of CH4 per year).
A 2006 UN FAO report reported that livestock generate more greenhouse gases as measured in CO2 equivalents than the entire transportation sector. Livestock accounts for 9 percent of anthropogenic CO2, 65 percent of anthropogenic nitrous oxide and 37 percent of anthropogenic methane. A senior UN official and co-author of the report, Henning Steinfeld, said "Livestock are one of the most significant contributors to today's most serious environmental problems."
Recent NASA research has confirmed the vital role of livestock eructation (burping) in global warming. "We understand that other greenhouse gases apart from carbon dioxide are important for climate change today," said Gavin Schmidt, the lead author of the study and a researcher at NASA's Goddard Institute for Space Studies in New York, NY and Columbia University's Center for Climate Systems Research. Other recent peer reviewed NASA research published in the journal Science has also indicated that the contribution of methane to global warming has been underestimated.
Nicholas Stern, the author of the 2006 Stern Review on climate change has stated "people will need to turn vegetarian if the world is to conquer climate change". President of the National Academy of Sciences Ralph Cicerone
(an atmospheric scientist), has indicated the contribution of methane by livestock flatulence
and eructation to global warming is a “serious topic.” Cicerone states “Methane is the second-most-important greenhouse gas in the atmosphere now. The population of beef cattle and dairy cattle has grown so much that methane from cows now is big. This is not a trivial issue."
Approximately 5% of the methane is released via the flatus, whereas the other 95% is released via eructation. Vaccines are under development to reduce the amount introduced through eructation.
of sludge.
, and thus during the production, processing, storage, transmission, and distribution of natural gas, a significant amount of methane is lost into the atmosphere. This is particularly true of the oil industry.
Reaction with the hydroxyl radical- The major removal mechanism of methane from the atmosphere involves radical chemistry
; it reacts with the hydroxyl radical
(·OH) in the troposphere
or stratosphere
to create the CH·3 radical and water vapor. In addition to being the largest known sink for atmospheric methane, this reaction is one of the most important sources of water vapor in the upper atmosphere.
This reaction in the troposphere
gives a methane lifetime of 9.6 years. Two more minor sinks are soil sinks (160 year lifetime) and stratospheric loss by reaction with ·OH, ·Cl and ·O1D in the stratosphere (120 year lifetime), giving a net lifetime of 8.4 years. Oxidation of methane is the main source of water vapor in the upper stratosphere (beginning at pressure levels around 10 kPa
).
The methyl radical formed in the above reaction will, during normal daytime conditions in the troposphere, usually react with another hydroxyl radical to form formaldehyde
. Note that this is not strictly oxidative pyrolysis
as described previously. Formaldehyde can react again with a hydroxyl radical to form carbon dioxide and more water vapor. Note that sidechains in these reactions may interact with nitrogen
compounds that will likely produce ozone
, thus supplanting radicals required in the initial reaction.
Methanotrophic bacteria in soils- Methanotrophic bacteria that reside within soil use methane as a source of carbon in methane oxidation. Methane oxidation allows methanotrophic bacteria to use methane as a source of energy, reacting methane with oxygen and as a result producing carbon dioxide and water.
CH4 + 2O2→ CO2 + 2H2O
Forest soils act as good sinks for atmospheric methane because soils are optimally moist for methanotroph activity, and the movement of gases between soil and atmosphere (soil diffusivity) is high. With a lower water table, any methane in the soil has to make it past the methanotrophic bacteria before it can reach the atmosphere.
Wetland soils, however, are often sources of atmospheric methane rather than sinks because the water table is much higher, and the methane can be diffused fairly easily into the air without have to compete with the soil’s methanotrophs.
and hydrochloric acid
. This process is known as free radical halogenations.
CH4 + Cl2 → CH3Cl + HCl
The only exceptions to this drop in growth rate occurred in 1991 and 1998 when growth rates increased suddenly to 14-15 nmol/mol per year for those years, nearly double the growth rates of the years before.
The 1991 spike is believed to have occurred due to the volcanic eruption of Mt. Pinatubo in June of that year. Volcanoes affect atmospheric methane emissions when they erupt, releasing ash and sulfur dioxide into the air. As a result, photochemistry of plants is affected and the removal of methane via the tropospheric hydroxyl radical is reduced. However, growth rates quickly fell due to lower temperatures and global reduction in rainfall.
The cause of the 1998 spike is unresolved, but scientists are currently attributing it to a combination of increased wetland and rice field emissions as well as an increased amount of biomass burning. 1998 was also the warmest year since surface temperatures were first recorded, suggesting that anomalously high temperatures can induce elevated methane emission.
Data from 2007 suggested methane concentrations were beginning to rise again. This was confirmed in 2010 when a study showed methane levels were on the rise for the 3 years 2007 to 2009. After a decade of near-zero growth in methane levels, "globally averaged atmospheric methane increased by [approximately] 7 nmol/mol per year during 2007 and 2008. During the first half of 2009, globally averaged atmospheric CH4 was [approximately] 7 nmol/mol greater than it was in 2008, suggesting that the increase will continue in 2009."
Methane emissions levels vary greatly depending on the local geography. For both natural and anthropogenic sources, higher temperatures and higher water levels result in the anaerobic environment that is necessary for methane production.
For example, plants that produce methane can emit as much as two to four times more methane during the day than during the night. This is directly related to the fact that plants tend to rely on solar energy to enact chemical processes.
Additionally, methane emissions are affected by the level of water sources. Seasonal flooding during the spring and summer naturally increases the amount of methane released into the air.
A recent article from William F. Ruddiman, however, indicates that the anthropogenic change in methane may have started 5000 years prior to the industrial revolution. The methane insolation
cycles of the ice core remained stable and predictable until 5000 years ago, most likely due to some anthropogenic effect. Ruddiman suggests that the transition of humans from hunter gatherers into agricultural farming was the first instance of humans affecting methane concentration in the atmosphere. Ruddiman’s hypothesis is supported by the fact that early rice irrigation occurred approximately 5000 years ago—the same time the ice core cycles lost their predictability. Due to the inefficiency of humans first learning how to grow rice, extensive rice paddies, over flooded and filled with weeds, resulting in huge methane emitting wetlands, would have been needed to feed even a small population.
Another source of methane emissions has been identified in Russia. Near Yamburg and Urengoy exist gas fields with a methane concentration of 97 percent. The gas obtained from these fields is taken and exported to Western and Central Europe through an extensive pipeline system known as the Trans-Siberian natural gas pipeline system. In accordance with the IPCC and other natural gas emissions control groups, measurements had to be taken throughout the pipeline to measure methane emissions from technological discharges and leaks at the pipeline fittings and vents. Although the majority of the natural gas leaks were carbon dioxide, a significant amount of methane was also being consistently released from the pipeline as a result of leaks and breakdowns. In 2001, natural gas emissions from the pipeline and natural gas transportation system accounted for 1 percent of the natural gas produced. Fortunately, between 2001 and 2005, this number reduced to 0.7 percent, and even the 2001 value is still significantly less than that of 1996. Thus, it is suggested that while natural gas transportation is a significant anthropogenic source of methane, over time as technology advances and greenhouse gas emission awareness increases, methane emission growth rates decrease and natural gases are overall better managed and controlled.
For example, in order to counteract the immense amount of methane that ruminants give off, a type of drug called monensin (marketed as rumensin™) has been developed. This drug is classified as an ionophore, which is an antibiotic that is naturally produced by a harmless bacteria strain. This drug not only improves feed efficiency but also reduces the amount of methane gas emitted from the animal and its manure.
In addition to medicine, specific manure management techniques have been developed to counteract harmful emissions from livestock manure. Educational resources have even begun to be provided for small farms run by owners who do not realize the harmful effects of livestock manure on the environment. Management techniques include daily pickup and storage of manure in a completely closed off storage facility that will prevent runoff from making it into bodies of water. The manure can then be kept in storage until it is either reused for fertilizer or taken away and stored in an offsite compost. Nutrient levels of various animal manures are even provided for optimal use as compost for gardens and agriculture.
In order to reduce effects on methane oxidation in soil, several steps can be taken. Controlling the usage of nitrogen enhancing fertilizer and reducing the amount of nitrogen pollution into the air can both lower inhibition of methane oxidation—a major sink of atmospheric methane. Additionally, using drier growing conditions for crops such as rice and selecting strains of crops that produce more food per unit area can reduce the amount of land with ideal conditions for methanogenesis. Careful selection of areas of land conversion (for example, plowing down forests to create agricultural fields) can also reduce the destruction of major areas of methane oxidation.
To counteract methane emissions from landfills, on March 12, 1996, the EPA (Environmental Protection Agency) added the “Landfill Rule” to the Clean Air Act. This rule requires large landfills that have ever accepted municipal solid waste, have been used as of November 8, 1987, can hold at least 2.5 million metric tons of waste with a volume greater than 2.5 million cubic meters, and/or have nonmethane organic compound (NMOC) emissions of at least 50 metric tons per year to collect and combust emitted landfill gas
. This set of requirements excludes 96% of the landfills in the USA. While the direct result of this is landfills reducing emission of nonmethane compounds that form smog, the indirect result is reduction of methane emissions as well.
To reduce emissions from the natural gas industries, the EPA developed the Natural Gas STAR Program, also known as Gas STAR.
Another program was also developed by the EPA to reduce emissions from coal mining. The Coalbed Methane Outreach Program (CMOP) helps and encourages the mining industry to find ways to use or sell methane
that would otherwise be released from the coal mine into the atmosphere.
Climate change
Climate change is a significant and lasting change in the statistical distribution of weather patterns over periods ranging from decades to millions of years. It may be a change in average weather conditions or the distribution of events around that average...
. Atmospheric methane is one of the most potent and influential greenhouse gas
Greenhouse gas
A greenhouse gas is a gas in an atmosphere that absorbs and emits radiation within the thermal infrared range. This process is the fundamental cause of the greenhouse effect. The primary greenhouse gases in the Earth's atmosphere are water vapor, carbon dioxide, methane, nitrous oxide, and ozone...
es on Earth. The 100-year global warming potential
Global warming potential
Global-warming potential is a relative measure of how much heat a greenhouse gas traps in the atmosphere. It compares the amount of heat trapped by a certain mass of the gas in question to the amount of heat trapped by a similar mass of carbon dioxide. A GWP is calculated over a specific time...
of methane is 25, i.e. over a 100-year period, it traps 25 times more heat per mass unit than carbon dioxide
Carbon dioxide
Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
.

Atmospheric methane

Earth's atmosphere
The atmosphere of Earth is a layer of gases surrounding the planet Earth that is retained by Earth's gravity. The atmosphere protects life on Earth by absorbing ultraviolet solar radiation, warming the surface through heat retention , and reducing temperature extremes between day and night...
as there is now. The earliest methane was released into the atmosphere by volcanic activity. During this time, Earth's earliest life appeared. These first, ancient bacteria added to the methane concentration by converting hydrogen and carbon dioxide into methane and water. Oxygen did not become a major part of the atmosphere until photosynthetic organisms evolved later in Earth's history. With no oxygen, methane stayed in the atmosphere longer and at higher concentrations than it does today.
In present times, due to the increase in oxygen, the amount of methane has decreased. The average mole fraction of methane at the Earth's surface in 1998 was 1,745 nmol/mol. Its concentration is higher in the northern hemisphere as most sources (both natural and human) are larger. The concentrations vary seasonally with a minimum in the late summer mainly due to removal by the hydroxyl radical
Hydroxyl radical
The hydroxyl radical, •OH, is the neutral form of the hydroxide ion . Hydroxyl radicals are highly reactive and consequently short-lived; however, they form an important part of radical chemistry. Most notably hydroxyl radicals are produced from the decomposition of hydroperoxides or, in...
.
Methane is created near the surface, and it is carried into the stratosphere
Stratosphere
The stratosphere is the second major layer of Earth's atmosphere, just above the troposphere, and below the mesosphere. It is stratified in temperature, with warmer layers higher up and cooler layers farther down. This is in contrast to the troposphere near the Earth's surface, which is cooler...
by rising air in the tropics
Tropics
The tropics is a region of the Earth surrounding the Equator. It is limited in latitude by the Tropic of Cancer in the northern hemisphere at approximately N and the Tropic of Capricorn in the southern hemisphere at S; these latitudes correspond to the axial tilt of the Earth...
. Uncontrolled build-up of methane in Earth's atmosphere is naturally checked—although human influence can upset this natural regulation—by methane's reaction with hydroxyl radicals formed from singlet oxygen
Singlet oxygen
Singlet oxygen is the common name used for the diamagnetic form of molecular oxygen , which is less stable than the normal triplet oxygen. Because of its unusual properties, singlet oxygen can persist for over an hour at room temperature, depending on the environment...
atoms and with water vapor.
Methane as a greenhouse gas
Methane in the Earth's atmosphere is an important greenhouse gasGreenhouse gas
A greenhouse gas is a gas in an atmosphere that absorbs and emits radiation within the thermal infrared range. This process is the fundamental cause of the greenhouse effect. The primary greenhouse gases in the Earth's atmosphere are water vapor, carbon dioxide, methane, nitrous oxide, and ozone...
with a global warming potential
Global warming potential
Global-warming potential is a relative measure of how much heat a greenhouse gas traps in the atmosphere. It compares the amount of heat trapped by a certain mass of the gas in question to the amount of heat trapped by a similar mass of carbon dioxide. A GWP is calculated over a specific time...
of 25 over a 100-year period. This means that a methane emission will have 25 times the impact on temperature of a carbon dioxide emission of the same mass over the following 100 years. Methane has a large effect for a brief period (a net lifetime of 8.4 years in the atmosphere), whereas carbon dioxide has a small effect for a long period (over 100 years). Because of this difference in effect and time period, the global warming potential of methane over a 20 year time period is 72. The Earth's methane concentration has increased by about 150% since 1750, and it accounts for 20% of the total radiative forcing
Radiative forcing
In climate science, radiative forcing is generally defined as the change in net irradiance between different layers of the atmosphere. Typically, radiative forcing is quantified at the tropopause in units of watts per square meter. A positive forcing tends to warm the system, while a negative...
from all of the long-lived and globally mixed greenhouse gases. Usually, excess methane from landfills and other natural producers of methane are burned so CO2 is released into the atmosphere instead of methane because methane is such a more effective greenhouse gas. Recently methane emitted from coal mines has been successfully converted to electricity.
Global methane cycle
A. PermafrostPermafrost
In geology, permafrost, cryotic soil or permafrost soil is soil at or below the freezing point of water for two or more years. Ice is not always present, as may be in the case of nonporous bedrock, but it frequently occurs and it may be in amounts exceeding the potential hydraulic saturation of...
, glaciers, and ice cores – A source that slowly releases methane trapped in frozen environments as global temperatures rise.
B. Wetlands – Warm temperatures and moist environments are ideal for methane production. Most of the methane makes it past methane-consuming microorganisms.
C. Forest fire – Mass burning of organic matter releases huge amounts of methane into the atmosphere.
D. Rice paddies – The warmer and moister the rice field, the more methane is produced.
E. Animals – Microorganisms breaking down difficult to digest material in the guts of ruminant livestock and termites produce methane that is then released during defecation.
F. Plants – While methane can be consumed in soil before being released into the atmosphere, plants allow for direct travel of methane up through the roots and leaves and into the atmosphere. Plants may also be direct producers of methane.
G. Landfills – Decaying organic matter and anaerobic conditions cause landfills to be a significant source of methane.
H. Waste water treatment facilities
Sewage treatment
Sewage treatment, or domestic wastewater treatment, is the process of removing contaminants from wastewater and household sewage, both runoff and domestic. It includes physical, chemical, and biological processes to remove physical, chemical and biological contaminants...
– Anaerobic treatment of organic compounds in the water results in the production of methane.
I. Hydroxyl radical
Hydroxyl radical
The hydroxyl radical, •OH, is the neutral form of the hydroxide ion . Hydroxyl radicals are highly reactive and consequently short-lived; however, they form an important part of radical chemistry. Most notably hydroxyl radicals are produced from the decomposition of hydroperoxides or, in...
– OH in the atmosphere is the largest sink for atmospheric methane as well as one of the most significant sources of water vapor in the upper atmosphere.
J. Chlorine radical – Free chlorine in the atmosphere also reacts with methane.
Emissions accounting of methane


Houweling et al. (1999) give the following values for methane emissions (Tg/a=teragrams per year):
| Origin | Emission | ||
|---|---|---|---|
| Mass (Tg/a) | Type (%/a) | Total (%/a) | |
| Natural Emissions | |||
| Wetland Wetland A wetland is an area of land whose soil is saturated with water either permanently or seasonally. Wetlands are categorised by their characteristic vegetation, which is adapted to these unique soil conditions.... s (incl. Rice agriculture) |
225 | 83 | 37 |
| Termite Termite Termites are a group of eusocial insects that, until recently, were classified at the taxonomic rank of order Isoptera , but are now accepted as the epifamily Termitoidae, of the cockroach order Blattodea... s |
20 | 7 | 3 |
| Ocean Ocean An ocean is a major body of saline water, and a principal component of the hydrosphere. Approximately 71% of the Earth's surface is covered by ocean, a continuous body of water that is customarily divided into several principal oceans and smaller seas.More than half of this area is over 3,000... |
15 | 6 | 3 |
| Hydrate Hydrate Hydrate is a term used in inorganic chemistry and organic chemistry to indicate that a substance contains water. The chemical state of the water varies widely between hydrates, some of which were so labeled before their chemical structure was understood.... s |
10 | 4 | 2 |
| Natural Total | 270 | 100 | 45 |
| Anthropogenic Emissions | |||
| Energy | 110 | 33 | 18 |
| Landfill Landfill A landfill site , is a site for the disposal of waste materials by burial and is the oldest form of waste treatment... s |
40 | 12 | 7 |
| Ruminant Ruminant A ruminant is a mammal of the order Artiodactyla that digests plant-based food by initially softening it within the animal's first compartment of the stomach, principally through bacterial actions, then regurgitating the semi-digested mass, now known as cud, and chewing it again... s (Livestock) |
115 | 35 | 19 |
| Waste treatment | 25 | 8 | 4 |
| Biomass burning | 40 | 12 | 7 |
| Anthropogenic Total | 330 | 100 | 55 |
| Sinks | |||
| Soil Soil Soil is a natural body consisting of layers of mineral constituents of variable thicknesses, which differ from the parent materials in their morphological, physical, chemical, and mineralogical characteristics... s |
-30 | -5 | -5 |
| Tropospheric Troposphere The troposphere is the lowest portion of Earth's atmosphere. It contains approximately 80% of the atmosphere's mass and 99% of its water vapor and aerosols.... OH Hydroxyl A hydroxyl is a chemical group containing an oxygen atom covalently bonded with a hydrogen atom. In inorganic chemistry, the hydroxyl group is known as the hydroxide ion, and scientists and reference works generally use these different terms though they refer to the same chemical structure in... |
-510 | -88 | -85 |
| Stratospheric Stratosphere The stratosphere is the second major layer of Earth's atmosphere, just above the troposphere, and below the mesosphere. It is stratified in temperature, with warmer layers higher up and cooler layers farther down. This is in contrast to the troposphere near the Earth's surface, which is cooler... loss |
-40 | -7 | -7 |
| Sink Total | -580 | -100 | -97 |
| Emissions + Sinks | |||
| Imbalance (trend) | +20 | ~2.78 Tg/(nmol/mol) | +7.19 (nmol/mol)/a |
Any process that results in the production of methane and its release into the atmosphere can be considered a "source." The two main processes that are responsible for methane production occur as a result of microorganisms anaerobic
Anaerobic digestion
Anaerobic digestion is a series of processes in which microorganisms break down biodegradable material in the absence of oxygen. It is used for industrial or domestic purposes to manage waste and/or to release energy....
ally converting organic compounds into methane.
Methanogenesis
Methanogenesis
Methanogenesis or biomethanation is the formation of methane by microbes known as methanogens. Organisms capable of producing methane have been identified only from the domain Archaea, a group phylogenetically distinct from both eukaryotes and bacteria, although many live in close association with...
, the scientific term for methane production, occurs primarily in anaerobic conditions because of the lack of availability of other oxidants. In these conditions, microscopic organisms called archaea
Archaea
The Archaea are a group of single-celled microorganisms. A single individual or species from this domain is called an archaeon...
use acetate and hydrogen to break down essential resources in a process called fermentation
Fermentation (biochemistry)
Fermentation is the process of extracting energy from the oxidation of organic compounds, such as carbohydrates, using an endogenous electron acceptor, which is usually an organic compound. In contrast, respiration is where electrons are donated to an exogenous electron acceptor, such as oxygen,...
.
Acetoclastic methanogenesis- certain archaea cleave acetate
Acetate
An acetate is a derivative of acetic acid. This term includes salts and esters, as well as the anion found in solution. Most of the approximately 5 billion kilograms of acetic acid produced annually in industry are used in the production of acetates, which usually take the form of polymers. In...
produced during anaerobic fermentation to yield methane and carbon dioxide.
H3C-COOH → CH4 + CO2
Hydrogenotrophic methanogenesis- archaea oxidize hydrogen with carbon dioxide to yield methane and water.
4H2 + CO2 → CH4 + 2H2O
While acetoclastic methanogenesis and hydrogenotrophic methanogenesis are the two major source reactions for atmospheric methane, other minor biological methane source reactions also occur.
Natural sources of atmospheric methane
Most ecological emissions of methane relate directly to methanogens generating methane in warm, moist soils as well as in the digestive tracts of certain animals.Methanogens
Methanogens are methane producing microorganisms. In order to produce energy, they use an anaerobic process called fermentation. This process is used in lieu of aerobic, or with oxygen, processes because methanogens are typically poor competitors for the substrates, or needed materials for chemical reactions, required for aerobic processes. When acetate is broken down in fermentation, the result is the release of methane into the surrounding environment.Wetlands
Wetlands account for approximately 20 percent of atmospheric methane through emissions from soils and plants. Wetlands counteract the sinking action that normally occurs with soil because of the high water table. When the water table is low, the methane generated within the wetland soil has to come up through the soil and get past multitudes of methanotrophic bacteria. When the water table is higher, then the methane produced in the soil can more easily diffuse through the water and escape into the atmosphere.Animals
Ruminant animals, particularly cows and sheep, contain bacteria in their gastrointestinal systems that help to break down plant material. Some of these microorganisms use the acetate from the plant material to produce methane, and because these bacteria live in the stomachs and intestines of ruminants, whenever the animal “burps” or defecates, it emits methane as well. The amount of methane emitted by one cow is equivalent to the amount of methane that 2.5 acres of methanotrophic bacteria can consume.Termite
Termite
Termites are a group of eusocial insects that, until recently, were classified at the taxonomic rank of order Isoptera , but are now accepted as the epifamily Termitoidae, of the cockroach order Blattodea...
s also contain methanogenic microorganisms in their gut. However, some of these microorganisms are so unique that they live nowhere else in the world except in the third gut of termites. These microorganisms also break down biotic components to produce ethanol
Ethanol
Ethanol, also called ethyl alcohol, pure alcohol, grain alcohol, or drinking alcohol, is a volatile, flammable, colorless liquid. It is a psychoactive drug and one of the oldest recreational drugs. Best known as the type of alcohol found in alcoholic beverages, it is also used in thermometers, as a...
, as well as methane byproduct. However, unlike ruminants who lose 20 percent of the energy from the plants they eat, termites only lose 2 percent of their energy in the process. Thus comparatively, termites do not have to eat as much food as ruminants to obtain the same amount of energy, and give off proportionally less methane.
Plants
Living plants (e.g. forests) have recently been identified as a potentially important source of methane, possibly being responsible for approximately 10 to 30 percent of atmospheric methane. A 2006 paper calculated emissions of 62–236 Tg a−1, and "this newly identified source may have important implications".However the authors stress "our findings are preliminary with regard to the methane emission strength".
These findings have been called into question in a 2007 paper which found "there is no evidence for substantial aerobic methane emission by terrestrial plants, maximally 0.3% of the previously published values".
While the details of plant methane emissions have yet to be confirmed, plants as a significant methane source would help fill in the gaps of previous global methane budgets as well as explain large plumes of methane that have been observed over the tropics.
In wetlands, where rate of methane production are high, plants help methane travel into the atmosphere—acting like inverted lightning rods as they direct the gas up through the soil and into the air. They are also suspected to produce methane themselves, but because the plants would have to use aerobic conditions to produce methane, the process itself is still unidentified.
Methane gas from methane clathrates
At high pressures, such as are found on the bottom of the ocean, methane forms a solid clathrate with water, known as methane hydrateMethane clathrate
Methane clathrate, also called methane hydrate, hydromethane, methane ice, "fire ice", natural gas hydrate or just gas hydrate, is a solid clathrate compound in which a large amount of methane is trapped within a crystal structure of water, forming a solid similar to ice...
. An unknown, but possibly very large quantity of methane is trapped in this form in ocean sediments. The release of large volumes of methane gas from such sediments into the atmosphere has been suggested as a possible cause for rapid global warming
Global warming
Global warming refers to the rising average temperature of Earth's atmosphere and oceans and its projected continuation. In the last 100 years, Earth's average surface temperature increased by about with about two thirds of the increase occurring over just the last three decades...
events in the Earth's distant past, such as the Paleocene–Eocene Thermal Maximum of 55 million years ago, and the Great Dying.
Theories suggest that should global warming cause them to heat up sufficiently, all of this methane gas could again be released into the atmosphere. Since methane gas is twenty-five times stronger (for a given weight, averaged over 100 years) than as a greenhouse gas; this would immensely magnify the greenhouse effect.
Permafrost
Methane that gets frozen in permafrostPermafrost
In geology, permafrost, cryotic soil or permafrost soil is soil at or below the freezing point of water for two or more years. Ice is not always present, as may be in the case of nonporous bedrock, but it frequently occurs and it may be in amounts exceeding the potential hydraulic saturation of...
– land that is frozen for several years at a time – is slowly released from bogs as the permafrost melts. With rising global temperatures, the amount of permafrost melting and releasing methane continues to increase.
Although records of permafrost are limited, recent years (1999 to 2007) have seen record thawing of permafrost in Alaska
Alaska
Alaska is the largest state in the United States by area. It is situated in the northwest extremity of the North American continent, with Canada to the east, the Arctic Ocean to the north, and the Pacific Ocean to the west and south, with Russia further west across the Bering Strait...
and Siberia
Siberia
Siberia is an extensive region constituting almost all of Northern Asia. Comprising the central and eastern portion of the Russian Federation, it was part of the Soviet Union from its beginning, as its predecessor states, the Tsardom of Russia and the Russian Empire, conquered it during the 16th...
. Recent measurements in Siberia show that the methane released is five times greater than previously estimated. Melting yedoma
Yedoma
Yedoma is an organic-rich Pleistocene-age loess permafrost with ice content of 50–90% by volume. The amount of carbon trapped in this type of permafrost is much more prevalent than originally thought and may be about 500 Gt, that is almost 100 times the amount of carbon released into the air each...
, a type of permafrost, is a significant source of atmospheric methane (about 4 Tg of CH4 per year).
Anthropogenic sources of atmospheric methane
Slightly over half of the total emission is due to human activity. Since the Industrial Revolution humans have had a major impact on concentrations of atmospheric methane. As a result, humans have acquired the ability to affect concentrations of other gases in the atmosphere as well. For example, because methane traps heat in the atmosphere, increased methane emissions increase the temperature of the atmosphere. And, because warmer climates hold more water vapor, through methane emissions humans can indirectly increase the amount of water vapor in the atmosphere as well.Ecological conversion
Conversion of forests and natural environments into agricultural plots increases the amount of nitrogen in the soil, which inhibits methane oxidation, weakening the ability of the methanotrophic bacteria in the soil to act as sinks. Additionally, by changing the level of the water table, humans can directly affect the soil’s ability to act as a source or sink. The relationship between water table levels and methane emission is explained in the wetlands section of natural sources.Farm animals
Farming also acts to increase atmospheric methane through ruminant farm animals, such as sheep or cows. In 2006, the amount of methane emitted by farm animals alone exceeded that of the iron, steel, and cement industries combined. Two reasons can explain this immense amount of methane emission: first, the mass collections of cattle that must be used to feed the meat industry, and second, changes in natural feeding habits. In order to reduce the costs associated with the large amounts of feed needed to support livestock, humans have begun adding more nitrogen to cattle feed. The addition of nitrogen to the feed causes the cattle to need less food to feel full. This increase in nitrogen, however, feeds not only the cattle, but also the microorganisms in their stomachs. More methane producing microorganisms means higher methane emissions.A 2006 UN FAO report reported that livestock generate more greenhouse gases as measured in CO2 equivalents than the entire transportation sector. Livestock accounts for 9 percent of anthropogenic CO2, 65 percent of anthropogenic nitrous oxide and 37 percent of anthropogenic methane. A senior UN official and co-author of the report, Henning Steinfeld, said "Livestock are one of the most significant contributors to today's most serious environmental problems."
Recent NASA research has confirmed the vital role of livestock eructation (burping) in global warming. "We understand that other greenhouse gases apart from carbon dioxide are important for climate change today," said Gavin Schmidt, the lead author of the study and a researcher at NASA's Goddard Institute for Space Studies in New York, NY and Columbia University's Center for Climate Systems Research. Other recent peer reviewed NASA research published in the journal Science has also indicated that the contribution of methane to global warming has been underestimated.
Nicholas Stern, the author of the 2006 Stern Review on climate change has stated "people will need to turn vegetarian if the world is to conquer climate change". President of the National Academy of Sciences Ralph Cicerone
Ralph Cicerone
Ralph J. Cicerone is an American atmospheric scientist, a former chancellor of UC Irvine, and currently president of the National Academy of Sciences....
(an atmospheric scientist), has indicated the contribution of methane by livestock flatulence
Flatulence
Flatulence is the expulsion through the rectum of a mixture of gases that are byproducts of the digestion process of mammals and other animals. The medical term for the mixture of gases is flatus, informally known as a fart, or simply gas...
and eructation to global warming is a “serious topic.” Cicerone states “Methane is the second-most-important greenhouse gas in the atmosphere now. The population of beef cattle and dairy cattle has grown so much that methane from cows now is big. This is not a trivial issue."
Approximately 5% of the methane is released via the flatus, whereas the other 95% is released via eructation. Vaccines are under development to reduce the amount introduced through eructation.
Rice agriculture
Due to a continuously growing world population, rice agriculture has become one of the most powerful anthropogenic sources of methane. With warm weather end water-logged soil, rice paddies act like wetlands, but are generated by humans for the purpose of food production. Due to the swamp-like environment of rice fields, this crop alone is responsible for approximately 50-100 million metric tons of methane emission each year. This means that rice agriculture is responsible for approximately 15 to 20 percent of anthropogenic methane emissions. An article written by William F. Ruddiman explores the possibility that methane emissions started to rise as a result of anthropogenic activity 5000 years ago when ancient cultures started to settle and use agriculture, rice irrigation in particular, as a primary food source.Landfills
Due to the large collections of organic matter and availability of anaerobic conditions, landfills serve to be the largest source of atmospheric methane in the United States. Even after a landfill is closed, the mass amount of decaying matter continues to emit methane for years. Although the methanotrophic bacteria in the surrounding soil does oxidize some of the methane, approximately 90 percent of the methane produced in landfills escapes through the landfill cover and into the atmosphere.Waste water treatment
Waste water treatment facilities act to remove organic matter, solids, pathogens, and chemical hazards as a result of human contamination. Methane emission in waste treatment facilities occurs as a result of anaerobic treatments of organic compounds and anaerobic biodegradationBiodegradation
Biodegradation or biotic degradation or biotic decomposition is the chemical dissolution of materials by bacteria or other biological means...
of sludge.
Biomass burning
Incomplete burning of both living and dead organic matter results in the emission of methane. While natural wildfires can contribute to methane emissions, the bulk majority of biomass burning occurs as a result of humans- including everything from accidental burnings by civilians to deliberate burnings used to clear out land to biomass burnings occurring as a result of destroying waste.Natural gas distribution
Methane is a primary component of natural gasNatural gas
Natural gas is a naturally occurring gas mixture consisting primarily of methane, typically with 0–20% higher hydrocarbons . It is found associated with other hydrocarbon fuel, in coal beds, as methane clathrates, and is an important fuel source and a major feedstock for fertilizers.Most natural...
, and thus during the production, processing, storage, transmission, and distribution of natural gas, a significant amount of methane is lost into the atmosphere. This is particularly true of the oil industry.
Removal processes
Any process that consumes methane from the atmosphere can be considered a "sink" of atmospheric methane. The most prominent of these processes occur as a result of methane either being destroyed in the atmosphere or broken down in soil.Reaction with the hydroxyl radical- The major removal mechanism of methane from the atmosphere involves radical chemistry
Radical (chemistry)
Radicals are atoms, molecules, or ions with unpaired electrons on an open shell configuration. Free radicals may have positive, negative, or zero charge...
; it reacts with the hydroxyl radical
Hydroxyl radical
The hydroxyl radical, •OH, is the neutral form of the hydroxide ion . Hydroxyl radicals are highly reactive and consequently short-lived; however, they form an important part of radical chemistry. Most notably hydroxyl radicals are produced from the decomposition of hydroperoxides or, in...
(·OH) in the troposphere
Troposphere
The troposphere is the lowest portion of Earth's atmosphere. It contains approximately 80% of the atmosphere's mass and 99% of its water vapor and aerosols....
or stratosphere
Stratosphere
The stratosphere is the second major layer of Earth's atmosphere, just above the troposphere, and below the mesosphere. It is stratified in temperature, with warmer layers higher up and cooler layers farther down. This is in contrast to the troposphere near the Earth's surface, which is cooler...
to create the CH·3 radical and water vapor. In addition to being the largest known sink for atmospheric methane, this reaction is one of the most important sources of water vapor in the upper atmosphere.
This reaction in the troposphere
Troposphere
The troposphere is the lowest portion of Earth's atmosphere. It contains approximately 80% of the atmosphere's mass and 99% of its water vapor and aerosols....
gives a methane lifetime of 9.6 years. Two more minor sinks are soil sinks (160 year lifetime) and stratospheric loss by reaction with ·OH, ·Cl and ·O1D in the stratosphere (120 year lifetime), giving a net lifetime of 8.4 years. Oxidation of methane is the main source of water vapor in the upper stratosphere (beginning at pressure levels around 10 kPa
KPA
KPA may refer to:* Kenya Ports Authority* Kiln phosphoric acid, a dry process to produce phosphoric acid at high temperature in a kiln* Kilopascal , a unit of pressure* Known-plaintext attack, a method of cryptanalysis* Korean People's Army...
).
The methyl radical formed in the above reaction will, during normal daytime conditions in the troposphere, usually react with another hydroxyl radical to form formaldehyde
Formaldehyde
Formaldehyde is an organic compound with the formula CH2O. It is the simplest aldehyde, hence its systematic name methanal.Formaldehyde is a colorless gas with a characteristic pungent odor. It is an important precursor to many other chemical compounds, especially for polymers...
. Note that this is not strictly oxidative pyrolysis
Pyrolysis
Pyrolysis is a thermochemical decomposition of organic material at elevated temperatures without the participation of oxygen. It involves the simultaneous change of chemical composition and physical phase, and is irreversible...
as described previously. Formaldehyde can react again with a hydroxyl radical to form carbon dioxide and more water vapor. Note that sidechains in these reactions may interact with nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
compounds that will likely produce ozone
Ozone
Ozone , or trioxygen, is a triatomic molecule, consisting of three oxygen atoms. It is an allotrope of oxygen that is much less stable than the diatomic allotrope...
, thus supplanting radicals required in the initial reaction.
Methanotrophic bacteria in soils- Methanotrophic bacteria that reside within soil use methane as a source of carbon in methane oxidation. Methane oxidation allows methanotrophic bacteria to use methane as a source of energy, reacting methane with oxygen and as a result producing carbon dioxide and water.
CH4 + 2O2→ CO2 + 2H2O
Natural sinks of atmospheric methane
Most natural sinks occur as a result of chemical reactions in the atmosphere as well as oxidation by methane consuming bacteria in Earth’s soils.Methanotrophs in soils
Soils act as a major sink for atmospheric methane through the methanotrophic bacteria that reside within them. This occurs with two different types of bacteria. “High capacity-low affinity” methanotrophic bacteria grow in areas of high methane concentration, such as waterlogged soils in wetlands and other moist environments. And in areas of low methane concentration, “low capacity-high affinity” methanotrophic bacteria make use of the methane in the atmosphere to grow, rather than relying on methane in their immediate environment.Forest soils act as good sinks for atmospheric methane because soils are optimally moist for methanotroph activity, and the movement of gases between soil and atmosphere (soil diffusivity) is high. With a lower water table, any methane in the soil has to make it past the methanotrophic bacteria before it can reach the atmosphere.
Wetland soils, however, are often sources of atmospheric methane rather than sinks because the water table is much higher, and the methane can be diffused fairly easily into the air without have to compete with the soil’s methanotrophs.
Troposphere
The most effective sink of atmospheric methane is the hydroxyl radical in the troposphere, or the lowest portion of Earth’s atmosphere. As methane rises into the air, it reacts with the hydroxyl radical to create water vapor and carbon dioxide. The lifespan of methane in the atmosphere was estimated at 9.6 years as of 2001; however, increasing emissions of methane over time reduce the concentration of the hydroxyl radical in the atmosphere. With less OH˚ to react with, the lifespan of methane could also increase, resulting in greater concentrations of atmospheric methane.Stratosphere
Even if it is not destroyed in the troposphere, methane can usually only last 12 years before it is eventually destroyed in Earth’s next atmospheric layer: the stratosphere. Destruction in the stratosphere occurs the same way that it does in the troposphere: methane is oxidized to produce carbon dioxide and water vapor.Reaction with free chlorine
Methane also reacts with natural chlorine gas in the atmosphere to produce chloromethaneChloromethane
Chloromethane, also called methyl chloride, R-40 or HCC 40, is a chemical compound of the group of organic compounds called haloalkanes. It was once widely used as a refrigerant. It is a colorless extremely flammable gas with a minorly sweet odor, which is, however, detected at possibly toxic levels...
and hydrochloric acid
Hydrochloric acid
Hydrochloric acid is a solution of hydrogen chloride in water, that is a highly corrosive, strong mineral acid with many industrial uses. It is found naturally in gastric acid....
. This process is known as free radical halogenations.
CH4 + Cl2 → CH3Cl + HCl
Anthropogenic sinks of atmospheric methane
Humans have yet to act as any significant sink of atmospheric methane.Patterns of methane change over time
Since the 1800s, atmospheric methane concentrations have increased annually at a rate of about 0.9%. Long term atmospheric measurements of methane by NOAA show that the build up of methane has slowed dramatically over the last decade, after nearly tripling since pre-industrial times. Although scientists have yet to pinpoint the exact reason(s) for this sudden drop in growth rates, it is thought that this reduction is due to reduced industrial emissions and drought in wetland areas.The only exceptions to this drop in growth rate occurred in 1991 and 1998 when growth rates increased suddenly to 14-15 nmol/mol per year for those years, nearly double the growth rates of the years before.
The 1991 spike is believed to have occurred due to the volcanic eruption of Mt. Pinatubo in June of that year. Volcanoes affect atmospheric methane emissions when they erupt, releasing ash and sulfur dioxide into the air. As a result, photochemistry of plants is affected and the removal of methane via the tropospheric hydroxyl radical is reduced. However, growth rates quickly fell due to lower temperatures and global reduction in rainfall.
The cause of the 1998 spike is unresolved, but scientists are currently attributing it to a combination of increased wetland and rice field emissions as well as an increased amount of biomass burning. 1998 was also the warmest year since surface temperatures were first recorded, suggesting that anomalously high temperatures can induce elevated methane emission.
Data from 2007 suggested methane concentrations were beginning to rise again. This was confirmed in 2010 when a study showed methane levels were on the rise for the 3 years 2007 to 2009. After a decade of near-zero growth in methane levels, "globally averaged atmospheric methane increased by [approximately] 7 nmol/mol per year during 2007 and 2008. During the first half of 2009, globally averaged atmospheric CH4 was [approximately] 7 nmol/mol greater than it was in 2008, suggesting that the increase will continue in 2009."
Methane emissions levels vary greatly depending on the local geography. For both natural and anthropogenic sources, higher temperatures and higher water levels result in the anaerobic environment that is necessary for methane production.
Natural methane cycles
Emissions of methane into the atmosphere are directly related to temperature and moisture. Thus, the natural environmental changes that occur during seasonal change act as a major control of methane emission. Additionally, even changes in temperature during the day can affect the amount of methane that is produced and consumed.For example, plants that produce methane can emit as much as two to four times more methane during the day than during the night. This is directly related to the fact that plants tend to rely on solar energy to enact chemical processes.
Additionally, methane emissions are affected by the level of water sources. Seasonal flooding during the spring and summer naturally increases the amount of methane released into the air.
Changes in anthropogenic sources
The most clearly identified rise in atmospheric methane as a result of human activity occurred in the 1700s during the industrial revolution. As technology increased at a considerable rate, humans began to build factories and plants, burn fossil fuels for energy, and clear out forests and other vegetation for the purpose of building and agriculture. This growth continued to rise at a rate of almost 1 percent per year until around 1990 when growth rates dropped to almost zero.A recent article from William F. Ruddiman, however, indicates that the anthropogenic change in methane may have started 5000 years prior to the industrial revolution. The methane insolation
Insolation
Insolation is a measure of solar radiation energy received on a given surface area in a given time. It is commonly expressed as average irradiance in watts per square meter or kilowatt-hours per square meter per day...
cycles of the ice core remained stable and predictable until 5000 years ago, most likely due to some anthropogenic effect. Ruddiman suggests that the transition of humans from hunter gatherers into agricultural farming was the first instance of humans affecting methane concentration in the atmosphere. Ruddiman’s hypothesis is supported by the fact that early rice irrigation occurred approximately 5000 years ago—the same time the ice core cycles lost their predictability. Due to the inefficiency of humans first learning how to grow rice, extensive rice paddies, over flooded and filled with weeds, resulting in huge methane emitting wetlands, would have been needed to feed even a small population.
Another source of methane emissions has been identified in Russia. Near Yamburg and Urengoy exist gas fields with a methane concentration of 97 percent. The gas obtained from these fields is taken and exported to Western and Central Europe through an extensive pipeline system known as the Trans-Siberian natural gas pipeline system. In accordance with the IPCC and other natural gas emissions control groups, measurements had to be taken throughout the pipeline to measure methane emissions from technological discharges and leaks at the pipeline fittings and vents. Although the majority of the natural gas leaks were carbon dioxide, a significant amount of methane was also being consistently released from the pipeline as a result of leaks and breakdowns. In 2001, natural gas emissions from the pipeline and natural gas transportation system accounted for 1 percent of the natural gas produced. Fortunately, between 2001 and 2005, this number reduced to 0.7 percent, and even the 2001 value is still significantly less than that of 1996. Thus, it is suggested that while natural gas transportation is a significant anthropogenic source of methane, over time as technology advances and greenhouse gas emission awareness increases, methane emission growth rates decrease and natural gases are overall better managed and controlled.
Methane management techniques
In an effort to mitigate climate change, humans have started to develop alternative methods and medicines.For example, in order to counteract the immense amount of methane that ruminants give off, a type of drug called monensin (marketed as rumensin™) has been developed. This drug is classified as an ionophore, which is an antibiotic that is naturally produced by a harmless bacteria strain. This drug not only improves feed efficiency but also reduces the amount of methane gas emitted from the animal and its manure.
In addition to medicine, specific manure management techniques have been developed to counteract harmful emissions from livestock manure. Educational resources have even begun to be provided for small farms run by owners who do not realize the harmful effects of livestock manure on the environment. Management techniques include daily pickup and storage of manure in a completely closed off storage facility that will prevent runoff from making it into bodies of water. The manure can then be kept in storage until it is either reused for fertilizer or taken away and stored in an offsite compost. Nutrient levels of various animal manures are even provided for optimal use as compost for gardens and agriculture.
In order to reduce effects on methane oxidation in soil, several steps can be taken. Controlling the usage of nitrogen enhancing fertilizer and reducing the amount of nitrogen pollution into the air can both lower inhibition of methane oxidation—a major sink of atmospheric methane. Additionally, using drier growing conditions for crops such as rice and selecting strains of crops that produce more food per unit area can reduce the amount of land with ideal conditions for methanogenesis. Careful selection of areas of land conversion (for example, plowing down forests to create agricultural fields) can also reduce the destruction of major areas of methane oxidation.
To counteract methane emissions from landfills, on March 12, 1996, the EPA (Environmental Protection Agency) added the “Landfill Rule” to the Clean Air Act. This rule requires large landfills that have ever accepted municipal solid waste, have been used as of November 8, 1987, can hold at least 2.5 million metric tons of waste with a volume greater than 2.5 million cubic meters, and/or have nonmethane organic compound (NMOC) emissions of at least 50 metric tons per year to collect and combust emitted landfill gas
Landfill gas
Landfill gas is a complex mix of different gases created by the action of microorganisms within a landfill.-Production:Landfill gas production results from chemical reactions and microbes acting upon the waste as the putrescible materials begins to break down in the landfill...
. This set of requirements excludes 96% of the landfills in the USA. While the direct result of this is landfills reducing emission of nonmethane compounds that form smog, the indirect result is reduction of methane emissions as well.
To reduce emissions from the natural gas industries, the EPA developed the Natural Gas STAR Program, also known as Gas STAR.
Another program was also developed by the EPA to reduce emissions from coal mining. The Coalbed Methane Outreach Program (CMOP) helps and encourages the mining industry to find ways to use or sell methane
Coalbed methane
Coalbed methane or Coal Bed Methane, coalbed gas or coal mine methane is a form of natural gas extracted from coal beds. In recent decades it has become an important source of energy in United States, Canada, and other countries...
that would otherwise be released from the coal mine into the atmosphere.
External links
- Methane in tundra and oceans to be released in atmosphere
- Methane hydrate stability and anthropogenic climate change Biogeosciences Discuss., 4, 993-1057, 2007
- Methane: A Scientific Journey from Obscurity to Climate Super-Stardom Good Sept. 2004 background report from NASA GISS

