
Ellingham diagram
Encyclopedia
An Ellingham diagram is a graph showing the temperature dependence of the stability for compounds. This analysis is usually used to evaluate the ease of reduction of metal oxide
s and sulphides
. These diagrams were first constructed by Harold Ellingham
in 1944. In metallurgy
, the Ellingham diagram is used to predict the equilibrium temperature between a metal
, its oxide
and oxygen
, and by extension, reactions of a metal with sulphur, nitrogen
, and other non-metals. The diagrams are useful in predicting the conditions under which a metal ore
will be reduced to the metal. The analysis is thermodynamic in nature and ignores reaction kinetics. Thus, processes that are predicted to be favourable by the Ellingham diagram can still be slow.
[ΔG = ΔH - TΔS]
and are a particular graphical form of it. ΔG is the Gibbs Free Energy Change,ΔH is the Enthalpy Change and ΔS is the Entropy Change]
The Ellingham diagram plots the Gibbs free energy
change (ΔG) for the oxidation reaction versus the temperature. In the temperature ranges commonly used, the metal and the oxide are in a condensed state (liquid or solid) with the oxygen gaseous, the reactions may be exothermic or endothermic, but the ΔG of the oxidation always becomes more negative with lower temperature, and thus the reaction becomes more probable statistically. At a sufficiently high temperature, the sign of ΔG may invert (becoming negative) and the oxide can spontaneously reduce to the metal.
As with any chemical reaction prediction based on purely energetic grounds the reaction may or may not take place spontaneously on kinetic grounds if one or more stages in the reaction pathway have very high Activation Energies EA.
If two metals are present, two equilibriums have to be considered, so that the metal with the more negative ΔG reduces, the other oxidizes.
). Moreover, when carbon reacts with oxygen it forms gaseous oxides carbon monoxide
and carbon dioxide
, therefore the dynamics of its oxidation is different from that for metals: its oxidation has a more negative ΔG with higher temperatures. Carbon can thus serve as reducing agent
. Using this property, reduction of metals may be performed as a double redox
reaction at relatively low temperature.
industry, where it helps to select the best reducing agent for various ores in the extraction process. It also helps to guide the purification of metals, especially the removal of tramp elements. The direct reduction process for making iron rests firmly on the guidance of Ellingham diagrams, which show that hydrogen can alone reduce iron oxides to the metal.
Is subject to a less negative free energy change than the process:
Thus, in the blast furnace haematite is reduced by CO even in the presence of carbon. The overall equation for this carbothermic reaction
is:
 The Ellingham curve for aluminium
The Ellingham curve for aluminium
lies below the curves of most metals such chromium
, iron
, etc. This fact indicates that aluminum can be used as the reducing agent for oxides of all these metals. This result is illustrated as below:
The free energies of formation of chromium oxide and aluminum oxide per mole of oxygen consumed are -540kJ and -827kJ respectively. The processes are:
So Aluminum oxide is more stable than chromium oxide.
Equation (2) - Equation (1) gives
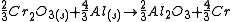
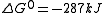
Since Gibb's free energy is negative, aluminium acts as the reducing agent.
In pyrometallurgy, Al is used as a reducing agent in the alumino-thermic process or thermite
process to extract Cr and Mn by reduction of their oxides.
Oxide
An oxide is a chemical compound that contains at least one oxygen atom in its chemical formula. Metal oxides typically contain an anion of oxygen in the oxidation state of −2....
s and sulphides
Sulfide
A sulfide is an anion of sulfur in its lowest oxidation state of 2-. Sulfide is also a slightly archaic term for thioethers, a common type of organosulfur compound that are well known for their bad odors.- Properties :...
. These diagrams were first constructed by Harold Ellingham
Harold Ellingham
Harold Johann Thomas Ellingham, OBE, was a British physical chemist, best known for his Ellingham diagrams, which summarize a large amount of information concerning extractive metallurgy....
in 1944. In metallurgy
Metallurgy
Metallurgy is a domain of materials science that studies the physical and chemical behavior of metallic elements, their intermetallic compounds, and their mixtures, which are called alloys. It is also the technology of metals: the way in which science is applied to their practical use...
, the Ellingham diagram is used to predict the equilibrium temperature between a metal
Metal
A metal , is an element, compound, or alloy that is a good conductor of both electricity and heat. Metals are usually malleable and shiny, that is they reflect most of incident light...
, its oxide
Oxide
An oxide is a chemical compound that contains at least one oxygen atom in its chemical formula. Metal oxides typically contain an anion of oxygen in the oxidation state of −2....
and oxygen
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
, and by extension, reactions of a metal with sulphur, nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
, and other non-metals. The diagrams are useful in predicting the conditions under which a metal ore
Ore
An ore is a type of rock that contains minerals with important elements including metals. The ores are extracted through mining; these are then refined to extract the valuable element....
will be reduced to the metal. The analysis is thermodynamic in nature and ignores reaction kinetics. Thus, processes that are predicted to be favourable by the Ellingham diagram can still be slow.
Thermodynamics
Ellingham diagrams follow from the Second Law of ThermodynamicsSecond law of thermodynamics
The second law of thermodynamics is an expression of the tendency that over time, differences in temperature, pressure, and chemical potential equilibrate in an isolated physical system. From the state of thermodynamic equilibrium, the law deduced the principle of the increase of entropy and...
[ΔG = ΔH - TΔS]
and are a particular graphical form of it. ΔG is the Gibbs Free Energy Change,ΔH is the Enthalpy Change and ΔS is the Entropy Change]
The Ellingham diagram plots the Gibbs free energy
Gibbs free energy
In thermodynamics, the Gibbs free energy is a thermodynamic potential that measures the "useful" or process-initiating work obtainable from a thermodynamic system at a constant temperature and pressure...
change (ΔG) for the oxidation reaction versus the temperature. In the temperature ranges commonly used, the metal and the oxide are in a condensed state (liquid or solid) with the oxygen gaseous, the reactions may be exothermic or endothermic, but the ΔG of the oxidation always becomes more negative with lower temperature, and thus the reaction becomes more probable statistically. At a sufficiently high temperature, the sign of ΔG may invert (becoming negative) and the oxide can spontaneously reduce to the metal.
As with any chemical reaction prediction based on purely energetic grounds the reaction may or may not take place spontaneously on kinetic grounds if one or more stages in the reaction pathway have very high Activation Energies EA.
If two metals are present, two equilibriums have to be considered, so that the metal with the more negative ΔG reduces, the other oxidizes.
Salient Features
- Curves in the Ellingham diagrams for the formation of metallic oxides are straight lines with a positive slope.
- The lower the position of a metal in the Ellingham diagram more is the stability of its oxide. For example, the Ellingham diagram for Al is found to be below Fe2O3.
- A metal found in the Ellingham diagram can act as a reducing agent for a metallic oxide found above it. Thus, Al can act as a reducing agent to Cr which is above it in the diagram.
- The greater the gap between any two lines, the greater the efficiency of the reducing agent.
- Stability of metallic oxides decrease with increase in temperature. Highly unstable oxides like Ag2O and HgO easily undergo thermal decomposition.
- The formation enthalpy of carbon dioxideCarbon dioxideCarbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
(CO2) is almost a temperature-independent constant, while that of carbon monoxideCarbon monoxideCarbon monoxide , also called carbonous oxide, is a colorless, odorless, and tasteless gas that is slightly lighter than air. It is highly toxic to humans and animals in higher quantities, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal...
(CO) has negative slope. According to Boudouard reactionBoudouard reactionThe Boudouard reaction is the redox reaction of a chemical equilibrium mixture of carbon monoxide and carbon dioxide at a given temperature. It is the disproportionation of carbon monoxide into carbon dioxide and graphite or its reverse:...
, carbon monoxide is the dominant compound in higher temperatures, and the higher the temperature, the more efficient reductant carbon monoxide also is. - A substance whose formation enthalpy is lower (ΔG line lower on diagram) at given temperature, will reduce one whose formation enthalpy is higher on the diagram. Hence metallic aluminum can reduce iron from iron oxide into metallic iron, aluminum itself oxidizing into aluminum oxide. (This reaction is employed in thermiteThermiteThermite is a pyrotechnic composition of a metal powder and a metal oxide that produces an exothermic oxidation-reduction reaction known as a thermite reaction. If aluminium is the reducing agent it is called an aluminothermic reaction...
.) - The intersection of two lines imply the equilibrium of oxidation and reduction reaction between two substances. Reduction with using a certain reductant is possible at the intersection point and higher temperatures where the ΔG line of the reductant is lower on diagram than the metallic oxide to be reduced. At the point of intersection the Gibbs energy is 0(zero), below this point the Gibbs energy is <0 and the oxides are stable,while above the point of intersection the Gibbs energy is >0 and so, the oxides are unstable.
Reducing agents
In industrial processes, the reduction of metal oxides is effected using carbon. Carbon is available cheaply as coal, which can be rendered to cokeCoke (fuel)
Coke is the solid carbonaceous material derived from destructive distillation of low-ash, low-sulfur bituminous coal. Cokes from coal are grey, hard, and porous. While coke can be formed naturally, the commonly used form is man-made.- History :...
). Moreover, when carbon reacts with oxygen it forms gaseous oxides carbon monoxide
Carbon monoxide
Carbon monoxide , also called carbonous oxide, is a colorless, odorless, and tasteless gas that is slightly lighter than air. It is highly toxic to humans and animals in higher quantities, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal...
and carbon dioxide
Carbon dioxide
Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
, therefore the dynamics of its oxidation is different from that for metals: its oxidation has a more negative ΔG with higher temperatures. Carbon can thus serve as reducing agent
Reducing agent
A reducing agent is the element or compound in a reduction-oxidation reaction that donates an electron to another species; however, since the reducer loses an electron we say it is "oxidized"...
. Using this property, reduction of metals may be performed as a double redox
Redox
Redox reactions describe all chemical reactions in which atoms have their oxidation state changed....
reaction at relatively low temperature.
Use of Ellingham Diagrams
The main application of Ellingham diagrams is in the extractive metallurgyExtractive metallurgy
Extractive metallurgy is the study of the processes used in the separation and concentration of raw materials. The field is an applied science, covering all aspects of the physical and chemical processes used to produce mineral-containing and metallic materials, sometimes for direct use as a...
industry, where it helps to select the best reducing agent for various ores in the extraction process. It also helps to guide the purification of metals, especially the removal of tramp elements. The direct reduction process for making iron rests firmly on the guidance of Ellingham diagrams, which show that hydrogen can alone reduce iron oxides to the metal.
Reducing agent for hematite
In iron ore smelting, hematite reduces at the top of the furnace, where temperature is in the range 600 - 700 °C. The Ellingham diagram indicates that in this range carbon monoxide will reducing agent since the process- 2 CO + O2 → 2 CO2
Is subject to a less negative free energy change than the process:
- 2 C + O2 →--> 2 CO
Thus, in the blast furnace haematite is reduced by CO even in the presence of carbon. The overall equation for this carbothermic reaction
Carbothermic reaction
Carbothermic reactions use carbon as reducing agent, usually for metal oxides. These chemical reactions are usually conducted at several hundreds of degree Celsius. Such processes are applied for production of the elemental forms of many elements. Carbothermic reactions are not however useful for...
is:
- 2 Fe2O3 + 3 C → 4 Fe + 3 CO2
Reducing agent for chromic oxide-carbon cannot be used
At high temperature the Ellingham curve for the reaction 2C(s) + O2(g) --> 2CO(g) slopes down and falls below the curves for all the metals. Hence, carbon can normally act as a reducing agent for all metal oxides at very high temperatures. But chromium formed at these temperatures react with carbon to form its carbide which gives undesirable properties to chromium metal obtained. Hence, for high temperature reduction of chromic oxide, carbon cannot be used.Alumino thermic process

Aluminium
Aluminium or aluminum is a silvery white member of the boron group of chemical elements. It has the symbol Al, and its atomic number is 13. It is not soluble in water under normal circumstances....
lies below the curves of most metals such chromium
Chromium
Chromium is a chemical element which has the symbol Cr and atomic number 24. It is the first element in Group 6. It is a steely-gray, lustrous, hard metal that takes a high polish and has a high melting point. It is also odorless, tasteless, and malleable...
, iron
Iron
Iron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust...
, etc. This fact indicates that aluminum can be used as the reducing agent for oxides of all these metals. This result is illustrated as below:
The free energies of formation of chromium oxide and aluminum oxide per mole of oxygen consumed are -540kJ and -827kJ respectively. The processes are:
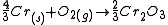 (1)
(1)
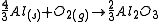 (2)
(2)
So Aluminum oxide is more stable than chromium oxide.
Equation (2) - Equation (1) gives
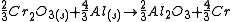
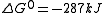
Since Gibb's free energy is negative, aluminium acts as the reducing agent.
In pyrometallurgy, Al is used as a reducing agent in the alumino-thermic process or thermite
Thermite
Thermite is a pyrotechnic composition of a metal powder and a metal oxide that produces an exothermic oxidation-reduction reaction known as a thermite reaction. If aluminium is the reducing agent it is called an aluminothermic reaction...
process to extract Cr and Mn by reduction of their oxides.
External links
- Interactive Ellingham diagrams at San José State UniversitySan José State UniversitySan Jose State University is a public university located in San Jose, California, United States...
- Ellingham diagram tutorial and interactive diagram (University of CambridgeUniversity of CambridgeThe University of Cambridge is a public research university located in Cambridge, United Kingdom. It is the second-oldest university in both the United Kingdom and the English-speaking world , and the seventh-oldest globally...
)

