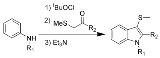
Gassman indole synthesis
Encyclopedia
The Gassman indole synthesis is a series of chemical reaction
s used to synthesize substituted indole
s from aniline
.
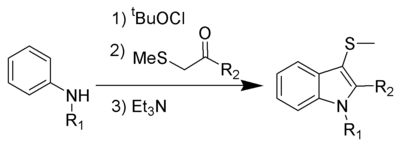 This is a one-pot chemical reaction, and none of the intermediates are isolated. R1 can be hydrogen
This is a one-pot chemical reaction, and none of the intermediates are isolated. R1 can be hydrogen
or alkyl, while R2 works best with aryl
, but can also be alkyl. Electron-rich anilines, such as 4-methoxyaniline, tend to fail in this reaction.
The 3-position thiomethyl group is often removed using Raney nickel
to give the 3-H-indole.
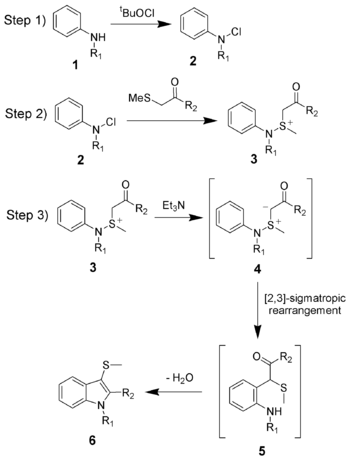 The reaction mechanism of the Gassman indole synthesis is divided among three steps.
The reaction mechanism of the Gassman indole synthesis is divided among three steps.
The first step is the oxidation of the aniline 1 using tert-butyl hypochlorite (tBuOCl) to give the chloramine
2.
The second step is the addition of the ketone
to give the sulfonium
ion
3, and is typically done at low temperatures (-78 °C).
The third and final step is the addition of a base
, which in this case is triethylamine
. Upon warming to room temperature, the base will deprotonate
the sulfonium ion creating the sulfonium ylide
4, which quickly undergoes a [2,3]-sigmatropic rearrangement
to give the ketone
5. The ketone 5 will undergo a facile condensation
to give the desired 3-thiomethylindole 6.
Chemical reaction
A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Chemical reactions can be either spontaneous, requiring no input of energy, or non-spontaneous, typically following the input of some type of energy, such as heat, light or electricity...
s used to synthesize substituted indole
Indole
Indole is an aromatic heterocyclic organic compound. It has a bicyclic structure, consisting of a six-membered benzene ring fused to a five-membered nitrogen-containing pyrrole ring. Indole is a popular component of fragrances and the precursor to many pharmaceuticals. Compounds that contain an...
s from aniline
Aniline
Aniline, phenylamine or aminobenzene is an organic compound with the formula C6H5NH2. Consisting of a phenyl group attached to an amino group, aniline is the prototypical aromatic amine. Being a precursor to many industrial chemicals, its main use is in the manufacture of precursors to polyurethane...
.

Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
or alkyl, while R2 works best with aryl
Aryl
In the context of organic molecules, aryl refers to any functional group or substituent derived from an aromatic ring, be it phenyl, naphthyl, thienyl, indolyl, etc....
, but can also be alkyl. Electron-rich anilines, such as 4-methoxyaniline, tend to fail in this reaction.
The 3-position thiomethyl group is often removed using Raney nickel
Raney nickel
Raney nickel is a solid catalyst composed of fine grains of a nickel-aluminium alloy, used in many industrial processes. It was developed in 1926 by American]] engineer Murray Raney as an alternative catalyst for the hydrogenation of vegetable oils in industrial processes...
to give the 3-H-indole.
Reaction mechanism

The first step is the oxidation of the aniline 1 using tert-butyl hypochlorite (tBuOCl) to give the chloramine
Chloramine
Chloramines are derivatives of ammonia by substitution of one, two or three hydrogen atoms with chlorine atoms. Monochloramine is an inorganic compound with the formula NH2Cl. It is an unstable colourless liquid at its melting point of -66° temperature, but it is usually handled as a dilute...
2.
The second step is the addition of the ketone
Ketone
In organic chemistry, a ketone is an organic compound with the structure RCR', where R and R' can be a variety of atoms and groups of atoms. It features a carbonyl group bonded to two other carbon atoms. Many ketones are known and many are of great importance in industry and in biology...
to give the sulfonium
Sulfonium
A sulfonium ion, also known as sulphonium ion or sulfanium ion, is a positively charged ion featuring three organic substituents attached to sulfur. They have the formula [SR3]+...
ion
Ion
An ion is an atom or molecule in which the total number of electrons is not equal to the total number of protons, giving it a net positive or negative electrical charge. The name was given by physicist Michael Faraday for the substances that allow a current to pass between electrodes in a...
3, and is typically done at low temperatures (-78 °C).
The third and final step is the addition of a base
Base (chemistry)
For the term in genetics, see base A base in chemistry is a substance that can accept hydrogen ions or more generally, donate electron pairs. A soluble base is referred to as an alkali if it contains and releases hydroxide ions quantitatively...
, which in this case is triethylamine
Triethylamine
Triethylamine is the chemical compound with the formula N3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine, for which TEA is also a common abbreviation....
. Upon warming to room temperature, the base will deprotonate
Deprotonation
Deprotonation is the removal of a proton from a molecule, forming the conjugate base.The relative ability of a molecule to give up a proton is measured by its pKa value. A low pKa value indicates that the compound is acidic and will easily give up its proton to a base...
the sulfonium ion creating the sulfonium ylide
Ylide
An ylide or ylid is a neutral dipolar molecule containing a formally negatively charged atom directly attached to a hetero atom with a formal positive charge , and in which both atoms have full octets of electrons. Ylides are thus 1,2-dipolar compounds...
4, which quickly undergoes a [2,3]-sigmatropic rearrangement
Sigmatropic reaction
A sigmatropic reaction in organic chemistry is a pericyclic reaction wherein the net result is one σ-bond is changed to another σ-bond in an uncatalyzed intramolecular process. The name sigmatropic is the result of a compounding of the long-established sigma designation from single carbon-carbon...
to give the ketone
Ketone
In organic chemistry, a ketone is an organic compound with the structure RCR', where R and R' can be a variety of atoms and groups of atoms. It features a carbonyl group bonded to two other carbon atoms. Many ketones are known and many are of great importance in industry and in biology...
5. The ketone 5 will undergo a facile condensation
Condensation reaction
A condensation reaction is a chemical reaction in which two molecules or moieties combine to form one single molecule, together with the loss of a small molecule. When this small molecule is water, it is known as a dehydration reaction; other possible small molecules lost are hydrogen chloride,...
to give the desired 3-thiomethylindole 6.

