Hydroamination
Encyclopedia
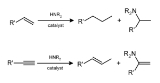
The hydroamination reaction is the addition of an N-H bond across the C=C or C≡C bonds of an alkene
or alkyne
. This is a highly atom economical
method of preparing substituted amines that are attractive targets for organic synthesis and the pharmaceutical industry.
The hydroamination reaction is approximately thermodynamically neutral; there is a high activation barrier due to the repulsion of the electron-rich substrate and the amine nucleophile. The reaction also has a high negative entropy, making it unfavorable at high temperatures. As a result, catalysts are necessary for this reaction to proceed.
Despite substantial effort, the development of a general catalytic process for this reaction remains elusive. Progress has been reported on the hydroamination of alkynes and alkenes using lanthanide
s and late transition metal
s. Although there have been many reports of the catalytic hydroamination reaction with group IV metals, there are far fewer describing enantioselective catalysis.
Group (IV) Hydroamination
Titanium
and zirconium
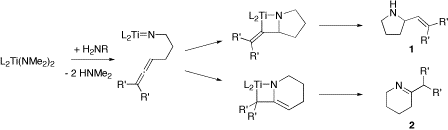
complexes catalyze inter-molecular hydroamination of alkynes and allenes. Both stoichiometric and catalytic variants were initially examined with zirconocene bis(amido) complexes. Titanocene amido and sulfonamido complexes catalyze the intra-molecular hydroamination of aminoalkenes via a [2+2] cycloaddition that forms the corresponding azametallacyclobutane, as illustrated in Figure 1. Subsequent protonolysis by incoming substrate gives the α-vinyl-pyrrolidine (1) or tetrahydropyridine (2) product. There is substantial experimental and theoretical evidence for the proposed imido intermediate and mechanism with neutral group IV catalysts.
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
or alkyne
Alkyne
Alkynes are hydrocarbons that have a triple bond between two carbon atoms, with the formula CnH2n-2. Alkynes are traditionally known as acetylenes, although the name acetylene also refers specifically to C2H2, known formally as ethyne using IUPAC nomenclature...
. This is a highly atom economical
Atom economy
Atom economy describes the conversion efficiency of a chemical process in terms of all atoms involved . In an ideal chemical process, the amount of starting materials or reactants equals the amount of all products generated and no atom is wasted...
method of preparing substituted amines that are attractive targets for organic synthesis and the pharmaceutical industry.
The hydroamination reaction is approximately thermodynamically neutral; there is a high activation barrier due to the repulsion of the electron-rich substrate and the amine nucleophile. The reaction also has a high negative entropy, making it unfavorable at high temperatures. As a result, catalysts are necessary for this reaction to proceed.
Despite substantial effort, the development of a general catalytic process for this reaction remains elusive. Progress has been reported on the hydroamination of alkynes and alkenes using lanthanide
Lanthanide
The lanthanide or lanthanoid series comprises the fifteen metallic chemical elements with atomic numbers 57 through 71, from lanthanum through lutetium...
s and late transition metal
Transition metal
The term transition metal has two possible meanings:*The IUPAC definition states that a transition metal is "an element whose atom has an incomplete d sub-shell, or which can give rise to cations with an incomplete d sub-shell." Group 12 elements are not transition metals in this definition.*Some...
s. Although there have been many reports of the catalytic hydroamination reaction with group IV metals, there are far fewer describing enantioselective catalysis.
Group (IV) Hydroamination
Titanium
Titanium
Titanium is a chemical element with the symbol Ti and atomic number 22. It has a low density and is a strong, lustrous, corrosion-resistant transition metal with a silver color....
and zirconium
Zirconium
Zirconium is a chemical element with the symbol Zr and atomic number 40. The name of zirconium is taken from the mineral zircon. Its atomic mass is 91.224. It is a lustrous, grey-white, strong transition metal that resembles titanium...
complexes catalyze inter-molecular hydroamination of alkynes and allenes. Both stoichiometric and catalytic variants were initially examined with zirconocene bis(amido) complexes. Titanocene amido and sulfonamido complexes catalyze the intra-molecular hydroamination of aminoalkenes via a [2+2] cycloaddition that forms the corresponding azametallacyclobutane, as illustrated in Figure 1. Subsequent protonolysis by incoming substrate gives the α-vinyl-pyrrolidine (1) or tetrahydropyridine (2) product. There is substantial experimental and theoretical evidence for the proposed imido intermediate and mechanism with neutral group IV catalysts.


