Hydrogen selenide
Encyclopedia
Hydrogen selenide is the inorganic compound
with the formula H2Se. It is the simplest and virtually the only hydride
of selenium
. H2Se is a colorless, flammable gas under standard conditions. It is the most toxic selenium compound with an exposure limit: 0.05 ppm over an 8 hour period. This compound has a very irritating smell resembling that of decayed horseradish
but smells of rotten eggs at higher concentrations.
-active vibrational bands are observed: 2358, 2345, and 1034 cm−1.
The properties of H2S
and H2Se are similar, although the selenide is more acidic with pKa = 3.89, and the second pKa = 11.0 at 25 °C. Reflecting its acidity, H2Se is soluble in water.
at T > 300 °C with hydrogen gas. A number of routes to H2Se have been reported, which are suitable for both large and small scale preparations. In the laboratory, H2Se is usually prepared by the action of water on Al2Se3, concomitant with formation of hydrated alumina. A related reaction involves the acid hydrolysis of FeSe.
H2Se can also be prepared by means of different methods based on the in situ generation in aqueous solution using boron hydride, Marsh test
and Devarda's alloy
. According to the Sonoda method, H2Se is generated from the reaction of H2O and CO on Se in the presence of Et3N
. H2Se can be purchased in cylinders.
(SO2).
Its decomposition is used to prepare highly pure Se metal.
H2Se gas is used to dope
semiconductors with selenium.
. The threshold limit value
is 0.05 ppm. At high concentrations even exposure for less than a minute causes the gas to attack the eyes and mucous membranes causing cold-like symptoms for at least a few days afterwards. In Germany, the limit in drinking
water is 0.008 mg/L, and the US EPA recommends a maximum contamination of 0.01 mg/L.
Inorganic compound
Inorganic compounds have traditionally been considered to be of inanimate, non-biological origin. In contrast, organic compounds have an explicit biological origin. However, over the past century, the classification of inorganic vs organic compounds has become less important to scientists,...
with the formula H2Se. It is the simplest and virtually the only hydride
Hydride
In chemistry, a hydride is the anion of hydrogen, H−, or, more commonly, a compound in which one or more hydrogen centres have nucleophilic, reducing, or basic properties. In compounds that are regarded as hydrides, hydrogen is bonded to a more electropositive element or group...
of selenium
Selenium
Selenium is a chemical element with atomic number 34, chemical symbol Se, and an atomic mass of 78.96. It is a nonmetal, whose properties are intermediate between those of adjacent chalcogen elements sulfur and tellurium...
. H2Se is a colorless, flammable gas under standard conditions. It is the most toxic selenium compound with an exposure limit: 0.05 ppm over an 8 hour period. This compound has a very irritating smell resembling that of decayed horseradish
Horseradish
Horseradish is a perennial plant of the Brassicaceae family, which also includes mustard, wasabi, broccoli, and cabbages. The plant is probably native to south eastern Europe and the Arab World , but is popular around the world today...
but smells of rotten eggs at higher concentrations.
Structure and properties
H2Se adopts a "bent" structure with a H-Se-H bond angle of 91°. Consistent with this structure, three IRInfrared
Infrared light is electromagnetic radiation with a wavelength longer than that of visible light, measured from the nominal edge of visible red light at 0.74 micrometres , and extending conventionally to 300 µm...
-active vibrational bands are observed: 2358, 2345, and 1034 cm−1.
The properties of H2S
Hydrogen sulfide
Hydrogen sulfide is the chemical compound with the formula . It is a colorless, very poisonous, flammable gas with the characteristic foul odor of expired eggs perceptible at concentrations as low as 0.00047 parts per million...
and H2Se are similar, although the selenide is more acidic with pKa = 3.89, and the second pKa = 11.0 at 25 °C. Reflecting its acidity, H2Se is soluble in water.
Preparation
Industrially, it is produced by treating elemental seleniumSelenium
Selenium is a chemical element with atomic number 34, chemical symbol Se, and an atomic mass of 78.96. It is a nonmetal, whose properties are intermediate between those of adjacent chalcogen elements sulfur and tellurium...
at T > 300 °C with hydrogen gas. A number of routes to H2Se have been reported, which are suitable for both large and small scale preparations. In the laboratory, H2Se is usually prepared by the action of water on Al2Se3, concomitant with formation of hydrated alumina. A related reaction involves the acid hydrolysis of FeSe.
-
- Al2Se3 + 6 H2O 2 Al(OH)3 + 3 H2Se
H2Se can also be prepared by means of different methods based on the in situ generation in aqueous solution using boron hydride, Marsh test
Marsh test
The Marsh test is a highly sensitive method in the detection of arsenic, especially useful in the field of forensic toxicology when arsenic was used as a poison...
and Devarda's alloy
Devarda's alloy
Devarda's alloy , is an alloy of aluminium , copper and zinc ....
. According to the Sonoda method, H2Se is generated from the reaction of H2O and CO on Se in the presence of Et3N
Triethylamine
Triethylamine is the chemical compound with the formula N3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine, for which TEA is also a common abbreviation....
. H2Se can be purchased in cylinders.
Reactions
Elemental selenium can be recovered from H2Se through a reaction with aqueous sulfur dioxideSulfur dioxide
Sulfur dioxide is the chemical compound with the formula . It is released by volcanoes and in various industrial processes. Since coal and petroleum often contain sulfur compounds, their combustion generates sulfur dioxide unless the sulfur compounds are removed before burning the fuel...
(SO2).
-
- 2 H2Se + SO2 2 H2O + Se + S
Its decomposition is used to prepare highly pure Se metal.
Applications
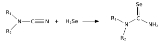
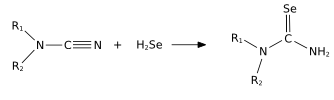
H2Se is commonly used in the synthesis of Se-containing compounds. It adds across alkenes. Illustrative is the synthesis of selenoureas from nitriles.
H2Se gas is used to dope
Doping (semiconductor)
In semiconductor production, doping intentionally introduces impurities into an extremely pure semiconductor for the purpose of modulating its electrical properties. The impurities are dependent upon the type of semiconductor. Lightly and moderately doped semiconductors are referred to as extrinsic...
semiconductors with selenium.
Safety
The gas is hazardous, being one of the most toxic compound of selenium and far more toxic than its congener hydrogen sulfideHydrogen sulfide
Hydrogen sulfide is the chemical compound with the formula . It is a colorless, very poisonous, flammable gas with the characteristic foul odor of expired eggs perceptible at concentrations as low as 0.00047 parts per million...
. The threshold limit value
Threshold Limit Value
The threshold limit value of a chemical substance is a level to which it is believed a worker can be exposed day after day for a working lifetime without adverse health effects. Strictly speaking, TLV is a reserved term of the American Conference of Governmental Industrial Hygienists...
is 0.05 ppm. At high concentrations even exposure for less than a minute causes the gas to attack the eyes and mucous membranes causing cold-like symptoms for at least a few days afterwards. In Germany, the limit in drinking
water is 0.008 mg/L, and the US EPA recommends a maximum contamination of 0.01 mg/L.

