
Magnetic structure
Encyclopedia
The term magnetic structure of a material pertains to the ordered arrangement of magnetic spins, typically within an ordered crystallographic lattice. Its study is a branch of solid-state chemistry
.
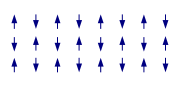
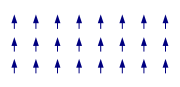 Most solid materials are Pauli-paramagnetic. These materials either do not have electron spins or these spins are not ordered unless an external field induces some non-random orientation. Such materials are not considered to 'have' a magnetic structure. This is different for ferro-
Most solid materials are Pauli-paramagnetic. These materials either do not have electron spins or these spins are not ordered unless an external field induces some non-random orientation. Such materials are not considered to 'have' a magnetic structure. This is different for ferro-
, ferri-
and antiferromagnetic
materials. They differ in the relative ordering of their spins within the lattice. In some ferromagnetic cases the structure may be relatively simple in that all spins point in the same direction, or at least that would be the case at very low temperatures. Towards higher temperatures there will be more and more 'rebellious' spins defying the order of the magnetic structure and at a certain temperature the order will break down and the spins will point in random directions. In some materials the pattern in which the spins order is much more complicated. In antiferromagnetic materials spins point in opposite directions so that the overall magnetic moment is zero. However, this is not necessarily achieved by a simple up and down pattern. Much more complicated structures can arise. At times one can recognize layers in which all spins point in one direction (as in a ferromagnet) but in the next layer they point in the opposite direction giving an overall antiferromagnetic arrangement. The possible number of arrangements is very large and can include spirals, clusters, tetrahedra etc.
. Neutrons are primarily scattered by the nuclei of the atoms in the structure. At a temperature above the ordering point of the magnetic moments, where the material behaves as a paramagnetic one, neutron diffraction will therefore give a picture of the crystallographic structure only. Below the ordering point, e.g. the Néel temperature
of an antiferromagnet
or the Curie-point of a ferromagnet the neutrons will also experience scattering from the magnetic moments because they themselves possess spin. The intensities of the Bragg reflections will therefore change. In fact in some cases entirely new Bragg-reflections will occur if the unit cell of the ordering is larger than that of the crystallographic structure. This is a form of superstructure formation. Thus the symmetry of the total structure may well differ from the crystallographic substructure. It needs to be described by one of the 1651 magnetic (Shubnikov) groups rather than one of the non-magnetic space groups.
Although ordinary X-ray diffraction is 'blind' to the arrangement of the spins, it has become possible to use a special form of X-ray diffraction to study magnetic structure. If a wavelength is selected that is close to an absorption edge
of one of elements contained in the materials the scattering becomes anomalous and this component to the scattering is (somewhat) sensitive to the non-spherical shape of the outer electrons of an atom with an unpaired spin. This means that this type of anomalous X-ray diffraction
does contain information of the desired type.
Solid-state chemistry
Solid-state chemistry, also sometimes referred to as materials chemistry, is the study of the synthesis, structure, and properties of solid phase materials, particularly, but not necessarily exclusively of, non-molecular solids...
.

Magnetic structures
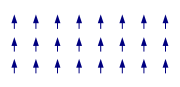
Ferromagnetism
Ferromagnetism is the basic mechanism by which certain materials form permanent magnets, or are attracted to magnets. In physics, several different types of magnetism are distinguished...
, ferri-
Ferrimagnetism
In physics, a ferrimagnetic material is one in which the magnetic moments of the atoms on different sublattices are opposed, as in antiferromagnetism; however, in ferrimagnetic materials, the opposing moments are unequal and a spontaneous magnetization remains...
and antiferromagnetic
Antiferromagnetism
In materials that exhibit antiferromagnetism, the magnetic moments of atoms or molecules, usuallyrelated to the spins of electrons, align in a regular pattern with neighboring spins pointing in opposite directions. This is, like ferromagnetism and ferrimagnetism, a manifestation of ordered magnetism...
materials. They differ in the relative ordering of their spins within the lattice. In some ferromagnetic cases the structure may be relatively simple in that all spins point in the same direction, or at least that would be the case at very low temperatures. Towards higher temperatures there will be more and more 'rebellious' spins defying the order of the magnetic structure and at a certain temperature the order will break down and the spins will point in random directions. In some materials the pattern in which the spins order is much more complicated. In antiferromagnetic materials spins point in opposite directions so that the overall magnetic moment is zero. However, this is not necessarily achieved by a simple up and down pattern. Much more complicated structures can arise. At times one can recognize layers in which all spins point in one direction (as in a ferromagnet) but in the next layer they point in the opposite direction giving an overall antiferromagnetic arrangement. The possible number of arrangements is very large and can include spirals, clusters, tetrahedra etc.
Techniques to study them
Such ordering can be studied by observing the magnetic susceptibility as a function of temperature and/or the size of the applied magnetic field, but a truly three-dimensional picture of the arrangement of the spins is best obtained by means of neutron diffractionNeutron diffraction
Neutron diffraction or elastic neutron scattering is the application of neutron scattering to the determination of the atomic and/or magnetic structure of a material: A sample to be examined is placed in a beam of thermal or cold neutrons to obtain a diffraction pattern that provides information of...
. Neutrons are primarily scattered by the nuclei of the atoms in the structure. At a temperature above the ordering point of the magnetic moments, where the material behaves as a paramagnetic one, neutron diffraction will therefore give a picture of the crystallographic structure only. Below the ordering point, e.g. the Néel temperature
Néel temperature
The Néel temperature or magnetic ordering temperature , TN, is the temperature above which an antiferromagnetic material becomes paramagnetic—that is, the thermal energy becomes large enough to destroy the macroscopic magnetic ordering within the material....
of an antiferromagnet
Antiferromagnetism
In materials that exhibit antiferromagnetism, the magnetic moments of atoms or molecules, usuallyrelated to the spins of electrons, align in a regular pattern with neighboring spins pointing in opposite directions. This is, like ferromagnetism and ferrimagnetism, a manifestation of ordered magnetism...
or the Curie-point of a ferromagnet the neutrons will also experience scattering from the magnetic moments because they themselves possess spin. The intensities of the Bragg reflections will therefore change. In fact in some cases entirely new Bragg-reflections will occur if the unit cell of the ordering is larger than that of the crystallographic structure. This is a form of superstructure formation. Thus the symmetry of the total structure may well differ from the crystallographic substructure. It needs to be described by one of the 1651 magnetic (Shubnikov) groups rather than one of the non-magnetic space groups.
Although ordinary X-ray diffraction is 'blind' to the arrangement of the spins, it has become possible to use a special form of X-ray diffraction to study magnetic structure. If a wavelength is selected that is close to an absorption edge
Absorption edge
An absorption edge is a sharp discontinuity in the absorption spectrum of a material. These discontinuities occur at wavelengths at which the energy of an absorbed photon corresponds to an electronic transition or ionisation potential....
of one of elements contained in the materials the scattering becomes anomalous and this component to the scattering is (somewhat) sensitive to the non-spherical shape of the outer electrons of an atom with an unpaired spin. This means that this type of anomalous X-ray diffraction
Anomalous X-ray scattering
Anomalous X-ray scattering AXRS or XRAS is a technique within X-ray diffraction that makes use of the anomalous dispersion that occurs when a wavelength is selected that is in the vicinity of an absorption edge of one of the constituent elements of the sample.-Atomic scattering factors:In X-ray...
does contain information of the desired type.

