
Meso compound
Encyclopedia
A meso compound or meso isomer is a non-optically active member of a set of stereoisomers, at least two of which are optically active. This means that despite containing two or more stereocenter
s (chiral centers) it is not chiral. A meso compound is superimposable on its mirror image, and it does not produce a "(+)" or "(-)" reading when analyzed with a polarimeter
. A meso compound need not even have a chiral center.
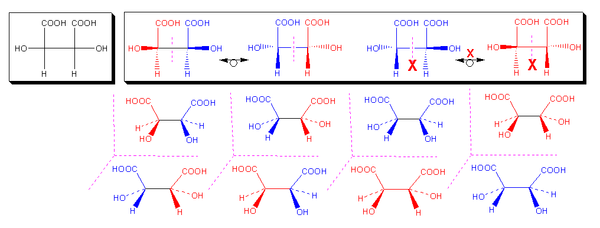
For example, there are 3 isomers of tartaric acid
(depicted below): There is a meso compound (the 2R,3S and 2S,3R isomers are equivalent) and the optically active pair of dextrotartaric acid
(L-(R,R)-(+)-tartaric acid) and levotartaric acid
(D-(S,S)-(-)-tartaric acid). In the meso compound an internal plane of symmetry exists, bisecting the molecule which is not present in the non-meso compounds. That is, on rotating the meso compound by 180° on a plane perpendicular to the screen, the same stereochemistry is obtained, again this is not seen in the non-meso tartaric acid. (see Fischer projection
).
It is a requirement for two of the stereocenters in a meso compound to have at least two substituents in common (though having this characteristic does not necessarily mean that the compound is meso). For example, in 2,4-pentanediol, both the second and fourth carbons, which are stereocenters, have all four substituents in common.
Since a meso isomer has a superposable mirror image, a compound with a total of n stereocenters cannot have 2n stereoisomers if at least one of the stereoisomers is meso.
has a meso cis-isomer (molecule has a mirror plane) and two trans-enantiomers:
The two cis stereoisomers of 1,2-substituted cyclohexane
s behave like meso compounds at room temperature in most cases. At room temperature, most 1,2-disubstituted cyclohexanes undergo rapid ring flipping (exceptions being rings with bulky substituents), and as a result, the two cis stereoisomers behave chemically identically with chiral reagents. At low temperatures, however, this is not the case, as the activation energy for the ring-flip cannot be overcome, and they therefore behave like enantiomers. A meso form is a type of stereoisomer which is optically inactive. In nearly all cases at room temperature, the two cis stereoisomers of 1,2-disubstituted cyclohexanes can be treated as chemically equivalent. Also noteworthy is the fact that when a cyclohexane undergoes a ring flip, the absolute configurations of the sterocenters do not change.
Stereocenter
A stereocenter or stereogenic center is an atom, bearing groups such that an interchanging of any two groups leads to a stereoisomer.A chirality center is a stereocenter consisting of an atom holding a set of ligands in a spatial arrangement which is not superposable on its mirror image...
s (chiral centers) it is not chiral. A meso compound is superimposable on its mirror image, and it does not produce a "(+)" or "(-)" reading when analyzed with a polarimeter
Polarimeter
A polarimeter is a scientific instrument used to measure the angle of rotation caused by passing polarized light through an optically active substance....
. A meso compound need not even have a chiral center.
For example, there are 3 isomers of tartaric acid
Tartaric acid
Tartaric acid is a white crystalline diprotic organic acid. It occurs naturally in many plants, particularly grapes, bananas, and tamarinds; is commonly combined with baking soda to function as a leavening agent in recipes, and is one of the main acids found in wine. It is added to other foods to...
(depicted below): There is a meso compound (the 2R,3S and 2S,3R isomers are equivalent) and the optically active pair of dextrotartaric acid
Tartaric acid
Tartaric acid is a white crystalline diprotic organic acid. It occurs naturally in many plants, particularly grapes, bananas, and tamarinds; is commonly combined with baking soda to function as a leavening agent in recipes, and is one of the main acids found in wine. It is added to other foods to...
(L-(R,R)-(+)-tartaric acid) and levotartaric acid
Tartaric acid
Tartaric acid is a white crystalline diprotic organic acid. It occurs naturally in many plants, particularly grapes, bananas, and tamarinds; is commonly combined with baking soda to function as a leavening agent in recipes, and is one of the main acids found in wine. It is added to other foods to...
(D-(S,S)-(-)-tartaric acid). In the meso compound an internal plane of symmetry exists, bisecting the molecule which is not present in the non-meso compounds. That is, on rotating the meso compound by 180° on a plane perpendicular to the screen, the same stereochemistry is obtained, again this is not seen in the non-meso tartaric acid. (see Fischer projection
Fischer projection
The Fischer projection, devised by Hermann Emil Fischer in 1891, is a two-dimensional representation of a three-dimensional organic molecule by projection. Fischer projections were originally proposed for the depiction of carbohydrates and used by chemists, particularly in organic chemistry and...
).

It is a requirement for two of the stereocenters in a meso compound to have at least two substituents in common (though having this characteristic does not necessarily mean that the compound is meso). For example, in 2,4-pentanediol, both the second and fourth carbons, which are stereocenters, have all four substituents in common.
Since a meso isomer has a superposable mirror image, a compound with a total of n stereocenters cannot have 2n stereoisomers if at least one of the stereoisomers is meso.
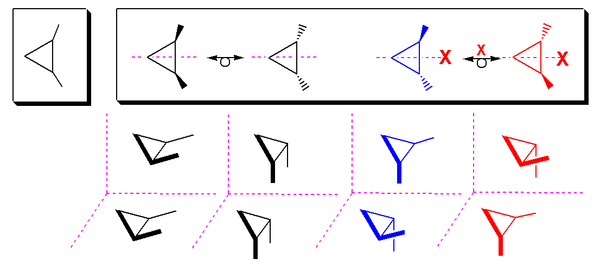
Cyclic meso compounds
1,2-substituted cyclopropaneCyclopropane
Cyclopropane is a cycloalkane molecule with the molecular formula C3H6, consisting of three carbon atoms linked to each other to form a ring, with each carbon atom bearing two hydrogen atoms...
has a meso cis-isomer (molecule has a mirror plane) and two trans-enantiomers:

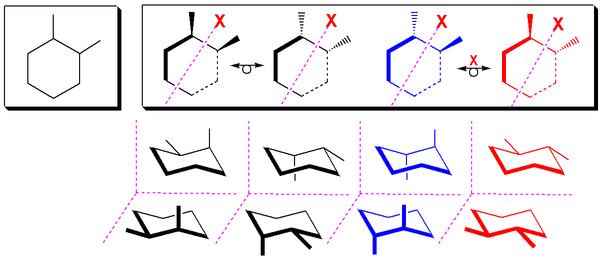
The two cis stereoisomers of 1,2-substituted cyclohexane
Cyclohexane
Cyclohexane is a cycloalkane with the molecular formula C6H12. Cyclohexane is used as a nonpolar solvent for the chemical industry, and also as a raw material for the industrial production of adipic acid and caprolactam, both of which being intermediates used in the production of nylon...
s behave like meso compounds at room temperature in most cases. At room temperature, most 1,2-disubstituted cyclohexanes undergo rapid ring flipping (exceptions being rings with bulky substituents), and as a result, the two cis stereoisomers behave chemically identically with chiral reagents. At low temperatures, however, this is not the case, as the activation energy for the ring-flip cannot be overcome, and they therefore behave like enantiomers. A meso form is a type of stereoisomer which is optically inactive. In nearly all cases at room temperature, the two cis stereoisomers of 1,2-disubstituted cyclohexanes can be treated as chemically equivalent. Also noteworthy is the fact that when a cyclohexane undergoes a ring flip, the absolute configurations of the sterocenters do not change.


