
Pickering emulsion
Encyclopedia
A Pickering emulsion is an emulsion
that is stabilized by solid particles (for example colloidal silica) which adsorb onto the interface
between the two phase
s. This type of emulsion
was named after S.U. Pickering
, who described the phenomenon in 1907, although the effect was first recognized by Walter Ramsden in 1903.
If oil and water are mixed and small oil droplets are formed and dispersed throughout the water, eventually the droplets will coalesce to decrease the amount of energy in the system. However, if solid particles are added to the mixture, they will bind to the surface of the interface and prevent the droplets from coalescing thus causing the emulsion to be more stable.
Properties such as hydrophobicity, shape, and size of the particle can have an effect on the stability of the emulsion. The particle’s contact angle
to the surface of the droplet is a characteristic of the hydrophobicity. If the contact angle of the particle to the interface is low, the particle will be mostly wetted by the droplet and therefore will not be likely to prevent coalescence of the droplets. Particles that are partially hydrophobic (i.e. contact angle of approximately 90°) are better stabilizers because they are partially wettable by both liquids and therefore bind better to the surface of the droplets. The stabilization energy is given by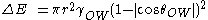
where r is the particle radius,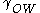 is the interfacial tension, and
is the interfacial tension, and 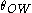 is the contact angle. When the contact angle is approximately 90°, the energy required to stabilize the system is at its minimum.
is the contact angle. When the contact angle is approximately 90°, the energy required to stabilize the system is at its minimum.
Generally the phase that preferentially wets the particle will be the continuous phase in the emulsion system.
Additionally, it has been demonstrated that the stability of the Pickering emulsions can be improved by the utilization of amphiphilic particles so-called "Janus Particles", due to the higher adsorption energy of the particles at the liquid-liquid interface.
Homogenised milk is an example of a Pickering-stabilized emulsion. Casein (protein) units are adsorbed at the surface of milk fat globules and act as a surfactant. The casein replaces the milkfat globule membrane (MFGM), which is damaged during homogenisation.
Recently, using latex particles for Pickering stabilization and then fusing these particles to form a permeable shell, a new form of particle called a colloidosome, has been developed for encapsulation.
Emulsion
An emulsion is a mixture of two or more liquids that are normally immiscible . Emulsions are part of a more general class of two-phase systems of matter called colloids. Although the terms colloid and emulsion are sometimes used interchangeably, emulsion is used when both the dispersed and the...
that is stabilized by solid particles (for example colloidal silica) which adsorb onto the interface
Interface (chemistry)
An interface is a surface forming a common boundary among two different phases, such as an insoluble solid and a liquid, two immiscible liquids or a liquid and an insoluble gas. The importance of the interface depends on which type of system is being treated: the bigger the quotient area/volume,...
between the two phase
Phase (matter)
In the physical sciences, a phase is a region of space , throughout which all physical properties of a material are essentially uniform. Examples of physical properties include density, index of refraction, and chemical composition...
s. This type of emulsion
Emulsion
An emulsion is a mixture of two or more liquids that are normally immiscible . Emulsions are part of a more general class of two-phase systems of matter called colloids. Although the terms colloid and emulsion are sometimes used interchangeably, emulsion is used when both the dispersed and the...
was named after S.U. Pickering
Percival Spencer Umfreville Pickering
Percival Spencer Umfreville Pickering was a British chemist and horticulturist.Pickering grew up in a wealthy family, and was able to start a career in science by building his own laboratory in his private house. In 1881, he took up a position as lecturer at Bedford College, where he stayed until...
, who described the phenomenon in 1907, although the effect was first recognized by Walter Ramsden in 1903.
If oil and water are mixed and small oil droplets are formed and dispersed throughout the water, eventually the droplets will coalesce to decrease the amount of energy in the system. However, if solid particles are added to the mixture, they will bind to the surface of the interface and prevent the droplets from coalescing thus causing the emulsion to be more stable.
Properties such as hydrophobicity, shape, and size of the particle can have an effect on the stability of the emulsion. The particle’s contact angle
Contact angle
The contact angle is the angle at which a liquid/vapor interface meets a solid surface. The contact angle is specific for any given system and is determined by the interactions across the three interfaces. Most often the concept is illustrated with a small liquid droplet resting on a flat...
to the surface of the droplet is a characteristic of the hydrophobicity. If the contact angle of the particle to the interface is low, the particle will be mostly wetted by the droplet and therefore will not be likely to prevent coalescence of the droplets. Particles that are partially hydrophobic (i.e. contact angle of approximately 90°) are better stabilizers because they are partially wettable by both liquids and therefore bind better to the surface of the droplets. The stabilization energy is given by
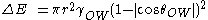
where r is the particle radius,
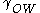 is the interfacial tension, and
is the interfacial tension, and 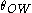 is the contact angle. When the contact angle is approximately 90°, the energy required to stabilize the system is at its minimum.
is the contact angle. When the contact angle is approximately 90°, the energy required to stabilize the system is at its minimum.Generally the phase that preferentially wets the particle will be the continuous phase in the emulsion system.
Additionally, it has been demonstrated that the stability of the Pickering emulsions can be improved by the utilization of amphiphilic particles so-called "Janus Particles", due to the higher adsorption energy of the particles at the liquid-liquid interface.
Homogenised milk is an example of a Pickering-stabilized emulsion. Casein (protein) units are adsorbed at the surface of milk fat globules and act as a surfactant. The casein replaces the milkfat globule membrane (MFGM), which is damaged during homogenisation.
Recently, using latex particles for Pickering stabilization and then fusing these particles to form a permeable shell, a new form of particle called a colloidosome, has been developed for encapsulation.

