Regioselectivity
Encyclopedia
In chemistry
, regioselectivity is the preference of one direction of chemical bond
making or breaking over all other possible directions. It can often apply to which of many possible positions a reagent will affect, such as which proton
a strong base
will abstract from an organic molecule, or where on a substituted benzene
ring a further substituent
will add.
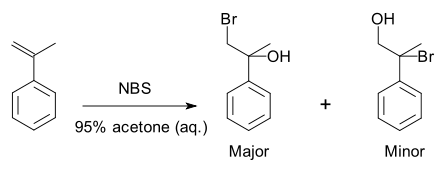
A specific example is a halohydrin formation reaction
with 2-propenylbenzene :
Because of the preference for the formation of one product over another, the reaction is selective. This reaction is regioselective because it selectively generates one constitutional isomer rather than the other.
Certain examples of regioselectivity have been formulated as rules for certain classes of compounds under certain conditions, many of which are named. Among the first introduced to chemistry students are Markovnikov's rule
for the addition of protic acids
to alkenes
, and the Fürst-Plattner rule
for the addition of nucleophiles
to derivatives of cyclohexene
, especially epoxide
derivatives.
Regioselectivity in ring-closure reactions is subject to Baldwin's rules
.
Chemistry
Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry is concerned with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds....
, regioselectivity is the preference of one direction of chemical bond
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electromagnetic force attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction...
making or breaking over all other possible directions. It can often apply to which of many possible positions a reagent will affect, such as which proton
Proton
The proton is a subatomic particle with the symbol or and a positive electric charge of 1 elementary charge. One or more protons are present in the nucleus of each atom, along with neutrons. The number of protons in each atom is its atomic number....
a strong base
Base (chemistry)
For the term in genetics, see base A base in chemistry is a substance that can accept hydrogen ions or more generally, donate electron pairs. A soluble base is referred to as an alkali if it contains and releases hydroxide ions quantitatively...
will abstract from an organic molecule, or where on a substituted benzene
Benzene
Benzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6....
ring a further substituent
Substituent
In organic chemistry and biochemistry, a substituent is an atom or group of atoms substituted in place of a hydrogen atom on the parent chain of a hydrocarbon...
will add.
A specific example is a halohydrin formation reaction
Halohydrin formation reaction
The halohydrin formation reaction is a chemical reaction in which a halogen is added to an alkene in aqueous solution to form a halohydrin. The reaction is a form of electrophilic addition; it is similar to the halogen addition reaction....
with 2-propenylbenzene :
Because of the preference for the formation of one product over another, the reaction is selective. This reaction is regioselective because it selectively generates one constitutional isomer rather than the other.
Certain examples of regioselectivity have been formulated as rules for certain classes of compounds under certain conditions, many of which are named. Among the first introduced to chemistry students are Markovnikov's rule
Markovnikov's rule
In organic chemistry, Markovnikov's rule or Markownikoff's rule is an observation based on Zaitsev's rule. It was formulated by the Russian chemist Vladimir Vasilevich Markovnikov in 1870....
for the addition of protic acids
Acid
An acid is a substance which reacts with a base. Commonly, acids can be identified as tasting sour, reacting with metals such as calcium, and bases like sodium carbonate. Aqueous acids have a pH of less than 7, where an acid of lower pH is typically stronger, and turn blue litmus paper red...
to alkenes
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
, and the Fürst-Plattner rule
Fürst-Plattner Rule
The Fürst-Plattner rule describes the addition of nucleophiles to cyclohexene derivatives.Epoxidation of the substituted cyclohexene gives the product where the R group is in the pseudo-equatorial position. Nucleophilic opening of the epoxide can occur by attack at either the 1 or 2 position...
for the addition of nucleophiles
Nucleophile
A nucleophile is a species that donates an electron-pair to an electrophile to form a chemical bond in a reaction. All molecules or ions with a free pair of electrons can act as nucleophiles. Because nucleophiles donate electrons, they are by definition Lewis bases.Nucleophilic describes the...
to derivatives of cyclohexene
Cyclohexene
Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorless liquid with a sharp smell. It is an intermediate in various industrial processes...
, especially epoxide
Epoxide
An epoxide is a cyclic ether with three ring atoms. This ring approximately defines an equilateral triangle, which makes it highly strained. The strained ring makes epoxides more reactive than other ethers. Simple epoxides are named from the parent compound ethylene oxide or oxirane, such as in...
derivatives.
Regioselectivity in ring-closure reactions is subject to Baldwin's rules
Baldwin's rules
Baldwin's Rules in organic chemistry are a series of guidelines outlining the relative favourabilities of ring closure reactions in alicyclic compounds. They were first proposed by Jack Baldwin in 1976...
.