
Secondary plot (kinetics)
Encyclopedia
In enzyme kinetics
, a secondary plot uses the intercept or slope from several Lineweaver-Burk plots to find additional kinetic constants.
For example, when a set of v by [S] curves from an enzyme
with a ping–pong mechanism (varying substrate A, fixed substrate B) are plotted in a Lineweaver–Burk plot, a set of parallel lines will be produced.
The following Michaelis–Menten equation
relates the initial reaction rate v0 to the substrate
concentrations [A] and [B]: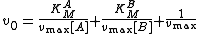
The y-intercept of this equation is equal to the following: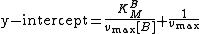
The y-intercept is determined at several different fixed concentrations of substrate B (and varying substrate A). The y-intercept values are then plotted versus 1/[B] to determine the Michaelis constant for substrate B,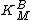 , as shown in the Figure to the right. The slope is equal to
, as shown in the Figure to the right. The slope is equal to 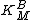 divided by
divided by 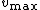 and the intercept is equal to 1 over
and the intercept is equal to 1 over 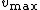 .
.
For a competitive enzyme inhibitor
, the apparent Michaelis constant is equal to the following:
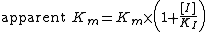
The slope of the Lineweaver-Burk plot is therefore equal to:
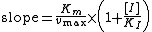
If one creates a secondary plot consisting of the slope values from several Lineweaver-Burk plots of varying inhibitor concentration [I], the competitive inhbition constant may be found. The slope of the secondary plot divided by the intercept is equal to 1/kI. This method allows one to find the kI constant, even when the Michaelis constant and vmax values are not known.
Enzyme kinetics
Enzyme kinetics is the study of the chemical reactions that are catalysed by enzymes. In enzyme kinetics, the reaction rate is measured and the effects of varying the conditions of the reaction investigated...
, a secondary plot uses the intercept or slope from several Lineweaver-Burk plots to find additional kinetic constants.
For example, when a set of v by [S] curves from an enzyme
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
with a ping–pong mechanism (varying substrate A, fixed substrate B) are plotted in a Lineweaver–Burk plot, a set of parallel lines will be produced.
The following Michaelis–Menten equation
Michaelis-Menten kinetics
In biochemistry, Michaelis–Menten kinetics is one of the simplest and best-known models of enzyme kinetics. It is named after German biochemist Leonor Michaelis and Canadian physician Maud Menten. The model takes the form of an equation describing the rate of enzymatic reactions, by relating...
relates the initial reaction rate v0 to the substrate
Substrate (biochemistry)
In biochemistry, a substrate is a molecule upon which an enzyme acts. Enzymes catalyze chemical reactions involving the substrate. In the case of a single substrate, the substrate binds with the enzyme active site, and an enzyme-substrate complex is formed. The substrate is transformed into one or...
concentrations [A] and [B]:
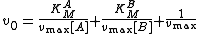
The y-intercept of this equation is equal to the following:
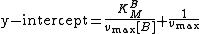
The y-intercept is determined at several different fixed concentrations of substrate B (and varying substrate A). The y-intercept values are then plotted versus 1/[B] to determine the Michaelis constant for substrate B,
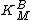 , as shown in the Figure to the right. The slope is equal to
, as shown in the Figure to the right. The slope is equal to 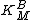 divided by
divided by 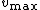 and the intercept is equal to 1 over
and the intercept is equal to 1 over 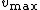 .
.Secondary Plot in Inhibition Studies
A secondary plot may also be used to find a specific inhbition constant, kI.For a competitive enzyme inhibitor
Enzyme inhibitor
An enzyme inhibitor is a molecule that binds to enzymes and decreases their activity. Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors. They are also used as herbicides and pesticides...
, the apparent Michaelis constant is equal to the following:
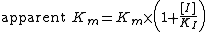
The slope of the Lineweaver-Burk plot is therefore equal to:
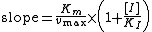
If one creates a secondary plot consisting of the slope values from several Lineweaver-Burk plots of varying inhibitor concentration [I], the competitive inhbition constant may be found. The slope of the secondary plot divided by the intercept is equal to 1/kI. This method allows one to find the kI constant, even when the Michaelis constant and vmax values are not known.

