
Hapticity
Encyclopedia
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
s of a ligand
Ligand
In coordination chemistry, a ligand is an ion or molecule that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from...
are coordinated to a central atom. Hapticity of a ligand is indicated by the Greek
Greek language
Greek is an independent branch of the Indo-European family of languages. Native to the southern Balkans, it has the longest documented history of any Indo-European language, spanning 34 centuries of written records. Its writing system has been the Greek alphabet for the majority of its history;...
character 'eta', η. A superscripted number following the η denotes the number of contiguous atoms of the ligand that are bound to the metal. In general the η-notation is only used when there is more than one atom coordinated (otherwise the κ-notation is used, see also hapticity vs. denticity).
History
The need for additional nomenclature for organometallic compounds became apparent in the mid-1950s when Dunitz, OrgelLeslie Orgel
Leslie Eleazer Orgel FRS was a British chemist.Born in London, England, Orgel received his B.A. in chemistry with first class honours from Oxford University in 1949...
, and Rich described the structure of the "sandwich complex
Sandwich compound
In organometallic chemistry, a sandwich compound is a chemical compound featuring a metal bound by haptic covalent bonds to two arene ligands. The arenes have the formula CnHn, substituted derivatives and heterocyclic derivatives...
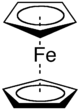
" ferrocene
Ferrocene
Ferrocene is an organometallic compound with the formula Fe2. It is the prototypical metallocene, a type of organometallic chemical compound consisting of two cyclopentadienyl rings bound on opposite sides of a central metal atom. Such organometallic compounds are also known as sandwich compounds...
by X-ray crystallography
X-ray crystallography
X-ray crystallography is a method of determining the arrangement of atoms within a crystal, in which a beam of X-rays strikes a crystal and causes the beam of light to spread into many specific directions. From the angles and intensities of these diffracted beams, a crystallographer can produce a...
where an iron
Iron
Iron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust...
atom is "sandwich
Sandwich
A sandwich is a food item, typically consisting of two or more slices of :bread with one or more fillings between them, or one slice of bread with a topping or toppings, commonly called an open sandwich. Sandwiches are a widely popular type of lunch food, typically taken to work or school, or...
ed" between two parallel cyclopentadienyl
Cyclopentadienyl
In organic chemistry, cyclopentadienyl is a cyclic group of atoms with the formula C5H5. Cyclopentadienyl are closely related to cyclopentadiene. Cyclopentadienyl have five carbon atoms bonded together in a pentagonal planar ring, all five of which are bonded to individual hydrogen atoms...
rings. Cotton
F. Albert Cotton
Frank Albert Cotton was the W.T. Doherty-Welch Foundation Chair and Distinguished Professor of Chemistry at Texas A&M University. He authored over 1700 scientific articles. Cotton was recognized for his research on the chemistry of the transition metals.-Education:Frank Albert Cotton was born on...
later proposed the term hapticity derived from the adjectival prefix hapto (from the Greek haptein, to fasten, denoting contact or combination) placed before the name of the olefin, where the Greek letter η (eta) is used to denote the number of contiguous atoms of a ligand that bind to a metal center. The term is usually employed to describe ligands containing extended π-systems or where agostic bonding is not obvious from the formula.
Historically important compounds where the ligands are described with hapticity
- FerroceneFerroceneFerrocene is an organometallic compound with the formula Fe2. It is the prototypical metallocene, a type of organometallic chemical compound consisting of two cyclopentadienyl rings bound on opposite sides of a central metal atom. Such organometallic compounds are also known as sandwich compounds...
- bis(η5-cyclopentadienylCyclopentadieneCyclopentadiene is an organic compound with the formula C5H6. This colorless liquid has a strong and unpleasant odor. At room temperature, this cyclic diene dimerizes over the course of hours to give dicyclopentadiene via a Diels–Alder reaction...
)ironIronIron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust... - UranoceneUranoceneUranocene U2 is the most notable cyclooctatetraenide of the f elements and one of the first organouranium compounds to be synthesized. Uranocene is a member of the actinocenes, a group of metallocenes incorporating elements from the actinide series...
- bis(η8-1,3,5,7-cyclooctatetraeneCyclooctatetraene1,3,5,7-Cyclooctatetraene is an unsaturated derivative of cyclooctane, with the formula C8H8. It is also known as [8]annulene. This polyunsaturated hydrocarbon is a colorless to light yellow flammable liquid at room temperature...
)uraniumUraniumUranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons... - W(CO)3(PPri3)2(η2-H2 ) - the first compound to be synthesized with a dihydrogen ligand.
- IrCl(CO)[P(C6H5)3]2(η2-O2) - the dioxygen derivative which forms reversibly upon oxygenation of Vaska's complexVaska's complexVaska's complex is the trivial name for the chemical compound trans-chlorocarbonylbisiridium, which has the formula IrCl[P3]2. This square planar diamagnetic organometallic complex consists of a central iridium atom bound to two mutually trans triphenylphosphine ligands, carbon monoxide, and a...
.
Examples
The η-notation is encountered in many coordination compounds:- Side-on bonding of molecules containing σ-bonds like H2Dihydrogen complexDihydrogen complexes are coordination complexes containing intact H2 as a ligand. The prototypical complex is W32. This class of compounds represent intermediates in metal-catalyzed reactions involving hydrogen. Hundreds of dihydrogen complexes have been reported...
:- W(CO)3(PiPr3)2(η2-H2)
- Side-on bonded ligands containing multiple bonded atoms, e.g. ethyleneEthyleneEthylene is a gaseous organic compound with the formula . It is the simplest alkene . Because it contains a carbon-carbon double bond, ethylene is classified as an unsaturated hydrocarbon. Ethylene is widely used in industry and is also a plant hormone...
in Zeise's saltZeise's saltZeise's salt, potassium trichloroplatinate, is the chemical compound with the formula KPtCl3]·H2O. The anion of this air-stable, yellow, coordination complex contains an η2-ethylene ligand. The anion features a platinum atom with a square planar geometry.-Preparation:This compound is commercially...
, which is bonded through donation of the π-bonding electrons:- K[PtCl3(η2-C2H4)].H2OWater of crystallizationIn crystallography, water of crystallization or water of hydration or crystallization water is water that occurs in crystals. Water of crystallization is necessary for the maintenance of crystalline properties, but capable of being removed by sufficient heat...
- K[PtCl3(η2-C2H4)].H2O
- Related complexes containing bridging π-ligands:
- (μ-η2:η2-C2H2AcetyleneAcetylene is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in pure form and thus is usually handled as a solution.As an alkyne, acetylene is unsaturated because...
)Co2(CO)6 and (Cp*2SmSamariumSamarium is a chemical element with the symbol Sm, atomic number 62 and atomic weight 150.36. It is a moderately hard silvery metal which readily oxidizes in air. Being a typical member of the lanthanide series, samarium usually assumes the oxidation state +3...
)2(μ-η2:η2- N2NitrogenNitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
) - DioxygenOxygenOxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
in bis{(trispyrazolylborato)copper(II)}(μ-η2:η2-O2),
- (μ-η2:η2-C2H2
-
- Note that with some bridging ligandBridging ligandA bridging ligand is a ligand that connects two or more atoms, usually metal ions. The ligand may be atomic or polyatomic. Virtually all complex organic compounds can serve as bridging ligands, so the term is usually restricted to small ligands such as pseudohalides or to ligands that are...
s, an alternative bridging mode is observed, e.g. κ1,κ1, like in (Me3SiCH2)3V(μ-N2-κ1(N),κ1(N'))V(CH2SiMe3)3 contains a bridging dinitrogen molecule, where the molecule is end-on coordinated to the two metal centers (see hapticity vs. denticity).- The bonding of π-bonded species can be extended over several atoms, e.g. in allylAllylAn allyl group is a substituent with the structural formula H2C=CH-CH2R, where R is the connection to the rest of the molecule. It is made up of a methylene , attached to a vinyl group . The name is derived from the Latin word for garlic, Allium sativum. Theodor Wertheim isolated an allyl...
, butadiene ligands, but also in cyclopentadienylCyclopentadienyl complexA cyclopentadienyl complex is a metal complex with one or more cyclopentadienyl groups . Based on the type of bonding between the metals and the cyclopentadienyl]] moieties, cyclopentadienyl complexes are classified into the following three categories: a) π-complexes, b) σ-complexes, and c) ionic...
or benzeneBenzeneBenzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6....
rings can share their electrons. - Apparent violations of the 18-electron rule18-Electron ruleThe 18-electron rule is a rule of thumb used primarily for predicting formulas for stable metal complexes. The rule rests on the fact that valence shells of a transition metal consists of nine valence orbitals, which collectively can accommodate 18 electrons either as nonbinding electron pairs or...
sometimes are explicable in compounds with unusual hapticities:- The 18-VE complex (η5-C5H5)Fe(η1-C5H5)(CO)2 contains one η5 bonded cyclopentadienyl, and one η1 bonded cyclopentadienyl.
- ReductionRedoxRedox reactions describe all chemical reactions in which atoms have their oxidation state changed....
of the 18-VE compound [Ru(η6-C6Me6)2]2+ (where both aromatic rings are bonded in an η6-coordination), results in another 18VE compound: [Ru(η6-C6Me6)(η4-C6Me6)].
- Examples of polyhapto coordinated heterocyclic and inorganic rings: Cr(η5-C4H4S)(CO)3 contains the sulfurSulfurSulfur or sulphur is the chemical element with atomic number 16. In the periodic table it is represented by the symbol S. It is an abundant, multivalent non-metal. Under normal conditions, sulfur atoms form cyclic octatomic molecules with chemical formula S8. Elemental sulfur is a bright yellow...
heterocycle thiopheneThiopheneThiophene is a heterocyclic compound with the formula C4H4S. Consisting of a flat five-membered ring, it is aromatic as indicated by its extensive substitution reactions. Related to thiophene are benzothiophene and dibenzothiophene, containing the thiophene ring fused with one and two benzene...
and Cr(η6-B3N3Me6)(CO)3 contains a coordinated inorganic ring (B3N3 ring).
- The bonding of π-bonded species can be extended over several atoms, e.g. in allyl
- Note that with some bridging ligand
Electrons donated by "π- ligands" vs. hapticity
| Ligand | Electrons contributed (neutral counting) | Electrons contributed (ionic counting) |
|---|---|---|
| η1 Allyl Allyl An allyl group is a substituent with the structural formula H2C=CH-CH2R, where R is the connection to the rest of the molecule. It is made up of a methylene , attached to a vinyl group . The name is derived from the Latin word for garlic, Allium sativum. Theodor Wertheim isolated an allyl... |
1 | 2 |
| η3-Allyl Allyl An allyl group is a substituent with the structural formula H2C=CH-CH2R, where R is the connection to the rest of the molecule. It is made up of a methylene , attached to a vinyl group . The name is derived from the Latin word for garlic, Allium sativum. Theodor Wertheim isolated an allyl... cyclopropenyl |
3 | 4 |
| η3-Allenyl | 3 | 4 |
| η2-Butadiene | 2 | 2 |
| η4-Butadiene | 4 | 4 |
| η1-cyclopentadienyl Cyclopentadienyl complex A cyclopentadienyl complex is a metal complex with one or more cyclopentadienyl groups . Based on the type of bonding between the metals and the cyclopentadienyl]] moieties, cyclopentadienyl complexes are classified into the following three categories: a) π-complexes, b) σ-complexes, and c) ionic... |
1 | 2 |
| η3-cyclopentadienyl Cyclopentadienyl complex A cyclopentadienyl complex is a metal complex with one or more cyclopentadienyl groups . Based on the type of bonding between the metals and the cyclopentadienyl]] moieties, cyclopentadienyl complexes are classified into the following three categories: a) π-complexes, b) σ-complexes, and c) ionic... |
3 | 4 |
| η5-cyclopentadienyl Cyclopentadienyl complex A cyclopentadienyl complex is a metal complex with one or more cyclopentadienyl groups . Based on the type of bonding between the metals and the cyclopentadienyl]] moieties, cyclopentadienyl complexes are classified into the following three categories: a) π-complexes, b) σ-complexes, and c) ionic... pentadienyl cyclohexadienyl |
5 | 6 |
| η2-Benzene Benzene Benzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6.... |
2 | 2 |
| η4-Benzene Benzene Benzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6.... |
4 | 4 |
| η6-Benzene Benzene Benzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6.... |
6 | 6 |
| η7-Cycloheptatrienyl | 7 | 6 |
| η8-Cyclooctatetraenyl | 8 | 10 |
Changes in hapticity
The hapticity of a ligand can change in the course of a reaction. E.g. in a redox reaction:Here one of the η6-benzene rings changes to a η4-benzene.
Similarly hapticity can change during a substitution reaction:
Here the η5-cyclopentadienyl changes to an η3-cyclopentadienyl, giving room on the metal for an extra 2-electron donating ligand 'L'. Removal of one molecule of CO and again donation of two more electrons by the cyclopentadienyl ligand restores the η5-cyclopentadienyl. The so-called indenyl effect
Indenyl effect
In organometallic chemistry, the indenyl effect refers to the enhanced rates of substitution displayed by η5-indenyl complexes vs the related η5-cyclopentadienyl complexes.-Mechanism:...
also describes changes in hapticity in a substitution reaction.
Hapticity vs. denticity
Hapticity must be distinguished from denticityDenticity
Denticity refers to the number of atoms in a single ligand that bind to a central atom in a coordination complex. In many cases, only one atom in the ligand binds to the metal, so the denticity equals one, and the ligand is said to be monodentate...
. Polydentate ligands coordinate via multiple coordination sites within the ligand. In this case the coordinating atoms are identified using the κ-notation, as for example seen in coordination of 1,2-bis(diphenylphosphino)ethane
1,2-Bis(diphenylphosphino)ethane
1,2-Bisethane is a commonly used bidentate ligand in coordination chemistry. Dppe is almost invariably chelated, although there are examples of unidentate and of bridging behavior.-Preparation:...
(Ph2PCH2CH2PPh2), to NiCl2
Nickel(II) chloride
Nickel chloride , is the chemical compound NiCl2. The anhydrous salt is yellow, but the more familiar hydrate NiCl2·6H2O is green. It is very rarely found in nature as mineral nickelbischofite. A dihydrate is also known. In general nickel chloride, in various forms, is the most important source of...
as dichloro[ethane-1,2-diylbis(diphenylphosphane)-κ2P]nickel(II). If the coordinating atoms are contiguous (connected to each other), the η-notation is used, as e.g. in titanocene dichloride
Titanocene dichloride
Titanocene dichloride is the organotitanium compound with the formula 2TiCl2, commonly abbreviated as Cp2TiCl2. This metallocene is a common reagent in organometallic and organic synthesis. It exists as a bright red solid that slowly hydrolyzes in air...
: dichlorobis(η5-2,4-cyclopentadien-1-yl)titanium.
Hapticity and fluxionality
Molecules with polyhapto ligands are often "fluxional", also known as stereochemically non-rigid. Two classes of fluxionality are prevalent for organometallic complexes of polyhapto ligands:- Case 1, typically: when the hapticity value is less than the number of sp2 carbon atoms. In such situations, the metal will often migrate from carbon to carbon, maintaining the same net hapticity. The η1-C5H5 ligand in (η5-C5H5)Fe( η1-C5H5)(CO)2 rearranges rapidly in solution such that Fe binds alternatingly to each carbon atom in the η1-C5H5 ligand. This reaction is degenerateDegenerate energy levelIn physics, two or more different quantum states are said to be degenerate if they are all at the same energy level. Statistically this means that they are all equally probable of being filled, and in Quantum Mechanics it is represented mathematically by the Hamiltonian for the system having more...
and, in the jargon of organic chemistryOrganic chemistryOrganic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, composition, reactions, and preparation of carbon-based compounds, hydrocarbons, and their derivatives...
, it is an example of a sigmatropic rearrangement. - Case 2, typically: complexes containing cyclic polyhapto ligands with maximized hapticity. Such ligands tend to rotate. A famous example is ferroceneFerroceneFerrocene is an organometallic compound with the formula Fe2. It is the prototypical metallocene, a type of organometallic chemical compound consisting of two cyclopentadienyl rings bound on opposite sides of a central metal atom. Such organometallic compounds are also known as sandwich compounds...
, Fe(η5-C5H5)2, wherein the Cp rings rotate with a low energy barrier about the principal axis of the molecule that "skewers" each ring (see rotational symmetryRotational symmetryGenerally speaking, an object with rotational symmetry is an object that looks the same after a certain amount of rotation. An object may have more than one rotational symmetry; for instance, if reflections or turning it over are not counted, the triskelion appearing on the Isle of Man's flag has...
). This "ring whizzing" explains, inter alia, why only one isomer can be isolated for Fe(η5-C5H4Br)2. In this case, the rotamers are not necessarily degenerate, but the rotational barriers have low energies of activation.


2.png)
.png)