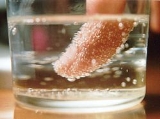
Nucleation
Overview
Liquid bubble
A bubble is a globule of one substance in another, usually gas in a liquid.Due to the Marangoni effect, bubbles may remain intact when they reach the surface of the immersive substance.-Common examples:...
s, crystals or glassy
Glass
Glass is an amorphous solid material. Glasses are typically brittle and optically transparent.The most familiar type of glass, used for centuries in windows and drinking vessels, is soda-lime glass, composed of about 75% silica plus Na2O, CaO, and several minor additives...
regions. Creation of liquid droplets in saturated vapor
Vapor
A vapor or vapour is a substance in the gas phase at a temperature lower than its critical point....
is also characterized by nucleation (see Cloud condensation nuclei
Cloud condensation nuclei
Cloud condensation nuclei or CCNs are small particles typically 0.2 µm, or 1/100 th the size of a cloud droplet ) about which cloud droplets coalesce. Water requires a non-gaseous surface to make the transition from a vapour to a liquid. In the atmosphere, this surface presents itself as tiny...
). Nucleation of crystalline, amorphous and even vacancy clusters solid materials is also important, for example to the semiconductor industry.

