
On the Equilibrium of Heterogeneous Substances
Encyclopedia
In the history of thermodynamics
, On the Equilibrium of Heterogeneous Substances is a 300-page paper written by American mathematical-engineer Willard Gibbs. It is one of the founding papers in thermodynamics
, along with German physicist Hermann von Helmholtz
's 1882 paper "Thermodynamik chemischer Vorgänge." Together they form the foundation of chemical thermodynamics
as well as a large part of physical chemistry
.
Gibbs's Equilibrium marked the beginning of chemical thermodynamics by integrating chemical, physical, electrical, and electromagnetic
phenomena into a coherent system. It introduced concepts such as chemical potential
, phase rule, and others, which form the basis for modern physical chemistry. American writer Bill Bryson
describes Gibbs's Equilibrium paper as "the Principia
of thermodynamics
".
On the Equilibrium of Heterogeneous Substances, was originally published in a relatively obscure American journal, the Transactions of the Connecticut Academy, in several parts, during the years 1875 to 1878 (although most cite "1876" as the key year). It remained largely unknown until translated into German by Wilhelm Ostwald
and into French by Henry Louis Le Chatelier.
of Fluids," and "Method of Geometrical Representation of the Thermodynamic Properties of Substances by means of Surfaces." His subsequent and most important publication was "On the Equilibrium of Heterogeneous Substances" (in two parts, 1876 and 1878). In this monumental, densely woven, 300-page treatise, the first law of thermodynamics
, the second law of thermodynamics
, the fundamental thermodynamic relation, are applied to the predication and quantification of thermodynamic
reaction tendencies
in any thermodynamic system
in a visual, three-dimensional graphical language of Lagrangian
calculus and phase changes, among others. As stated by Henri Louis Le Chatelier
, it "founded a new department of chemical science that is becoming comparable in importance to that created by Lavoisier." This work was translated into German by W. Ostwald (who styled its author the "founder of chemical energetics") in 1891 and into French by H. le Chatelier in 1899.
Gibbs's "Equilibrium" paper is considered one of the greatest achievements in physical science in the 19th century and one of the foundations of the science of physical chemistry
. In these papers Gibbs applied thermodynamics
to the interpretation of physicochemical phenomena and showed the explanation and interrelationship of what had been known only as isolated, inexplicable facts.
Gibbs' papers on heterogeneous equilibria included:
(The energy
of the world
is constant).
(The entropy
of the world tends to a maximum)
The comprehension of the laws which govern any material system is greatly facilitated by considering the energy
and entropy
of the system in the various states of which it is capable. As the difference of the values of the energy for any two states represents the combined amount of work
and heat received or yielded by the system
when it is brought from one state
to the other, and the difference of entropy is the limit of all possible values of the integral:
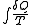
in which dQ denotes the element of heat received from external sources, and T is the temperature of the part of the system receiving it, the varying values of energy and entropy characterize in all that is essential the effect producible by the system in passing from one state to another. For by mechanical and thermodynamic contrivances, supposedly theoretically perfect, any supply of work and heat may be transformed into any other which does not differ from it either in the amount of work and heat taken together or in the value of the integral:
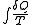
But it is not only in respect to the external relations of a system that its energy and entropy are of predominant importance. As in the case of simple mechanical systems, such as are discussed in theoretical mechanics, which are capable of only one kind of action upon external systems, namely the performance of mechanical work, the function which expresses the capability of the system of this kind of action also plays the leading part in the theory of equilibrium, the condition of equilibrium being that the variation of this function shall vanish, so in a thermodynamic system, such as all material systems are, which is capable of two different kinds of action upon external systems, the two functions which express the twofold capabilities of the system afford an almost equally simple criterion for equilibrium.
History of thermodynamics
The history of thermodynamics is a fundamental strand in the history of physics, the history of chemistry, and the history of science in general...
, On the Equilibrium of Heterogeneous Substances is a 300-page paper written by American mathematical-engineer Willard Gibbs. It is one of the founding papers in thermodynamics
Thermodynamics
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation...
, along with German physicist Hermann von Helmholtz
Hermann von Helmholtz
Hermann Ludwig Ferdinand von Helmholtz was a German physician and physicist who made significant contributions to several widely varied areas of modern science...
's 1882 paper "Thermodynamik chemischer Vorgänge." Together they form the foundation of chemical thermodynamics
Chemical thermodynamics
Chemical thermodynamics is the study of the interrelation of heat and work with chemical reactions or with physical changes of state within the confines of the laws of thermodynamics...
as well as a large part of physical chemistry
Physical chemistry
Physical chemistry is the study of macroscopic, atomic, subatomic, and particulate phenomena in chemical systems in terms of physical laws and concepts...
.
Gibbs's Equilibrium marked the beginning of chemical thermodynamics by integrating chemical, physical, electrical, and electromagnetic
Electromagnetism
Electromagnetism is one of the four fundamental interactions in nature. The other three are the strong interaction, the weak interaction and gravitation...
phenomena into a coherent system. It introduced concepts such as chemical potential
Chemical potential
Chemical potential, symbolized by μ, is a measure first described by the American engineer, chemist and mathematical physicist Josiah Willard Gibbs. It is the potential that a substance has to produce in order to alter a system...
, phase rule, and others, which form the basis for modern physical chemistry. American writer Bill Bryson
Bill Bryson
William McGuire "Bill" Bryson, OBE, is a best-selling American author of humorous books on travel, as well as books on the English language and on science. Born an American, he was a resident of Britain for most of his adult life before moving back to the US in 1995...
describes Gibbs's Equilibrium paper as "the Principia
Philosophiae Naturalis Principia Mathematica
Philosophiæ Naturalis Principia Mathematica, Latin for "Mathematical Principles of Natural Philosophy", often referred to as simply the Principia, is a work in three books by Sir Isaac Newton, first published 5 July 1687. Newton also published two further editions, in 1713 and 1726...
of thermodynamics
Thermodynamics
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation...
".
On the Equilibrium of Heterogeneous Substances, was originally published in a relatively obscure American journal, the Transactions of the Connecticut Academy, in several parts, during the years 1875 to 1878 (although most cite "1876" as the key year). It remained largely unknown until translated into German by Wilhelm Ostwald
Wilhelm Ostwald
Friedrich Wilhelm Ostwald was a Baltic German chemist. He received the Nobel Prize in Chemistry in 1909 for his work on catalysis, chemical equilibria and reaction velocities...
and into French by Henry Louis Le Chatelier.
Overview
Gibbs first contributed to mathematical physics with two papers published in 1873 in the Transactions of the Connecticut Academy on "Graphical Methods in the ThermodynamicsThermodynamics
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation...
of Fluids," and "Method of Geometrical Representation of the Thermodynamic Properties of Substances by means of Surfaces." His subsequent and most important publication was "On the Equilibrium of Heterogeneous Substances" (in two parts, 1876 and 1878). In this monumental, densely woven, 300-page treatise, the first law of thermodynamics
First law of thermodynamics
The first law of thermodynamics is an expression of the principle of conservation of work.The law states that energy can be transformed, i.e. changed from one form to another, but cannot be created nor destroyed...
, the second law of thermodynamics
Second law of thermodynamics
The second law of thermodynamics is an expression of the tendency that over time, differences in temperature, pressure, and chemical potential equilibrate in an isolated physical system. From the state of thermodynamic equilibrium, the law deduced the principle of the increase of entropy and...
, the fundamental thermodynamic relation, are applied to the predication and quantification of thermodynamic
Thermodynamics
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation...
reaction tendencies
Spontaneous process
A spontaneous process is the time-evolution of a system in which it releases free energy and moves to a lower, more thermodynamically stable energy state...
in any thermodynamic system
Thermodynamic system
A thermodynamic system is a precisely defined macroscopic region of the universe, often called a physical system, that is studied using the principles of thermodynamics....
in a visual, three-dimensional graphical language of Lagrangian
Lagrangian
The Lagrangian, L, of a dynamical system is a function that summarizes the dynamics of the system. It is named after Joseph Louis Lagrange. The concept of a Lagrangian was originally introduced in a reformulation of classical mechanics by Irish mathematician William Rowan Hamilton known as...
calculus and phase changes, among others. As stated by Henri Louis Le Chatelier
Henri Louis Le Chatelier
Henri Louis Le Châtelier was an influential French chemist of the late 19th and early 20th centuries. He is most famous for devising Le Châtelier's principle, used by chemists to predict the effect a changing condition has on a system in chemical equilibrium...
, it "founded a new department of chemical science that is becoming comparable in importance to that created by Lavoisier." This work was translated into German by W. Ostwald (who styled its author the "founder of chemical energetics") in 1891 and into French by H. le Chatelier in 1899.
Gibbs's "Equilibrium" paper is considered one of the greatest achievements in physical science in the 19th century and one of the foundations of the science of physical chemistry
Physical chemistry
Physical chemistry is the study of macroscopic, atomic, subatomic, and particulate phenomena in chemical systems in terms of physical laws and concepts...
. In these papers Gibbs applied thermodynamics
Thermodynamics
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation...
to the interpretation of physicochemical phenomena and showed the explanation and interrelationship of what had been known only as isolated, inexplicable facts.
Gibbs' papers on heterogeneous equilibria included:
- Some chemical potentialChemical potentialChemical potential, symbolized by μ, is a measure first described by the American engineer, chemist and mathematical physicist Josiah Willard Gibbs. It is the potential that a substance has to produce in order to alter a system...
concepts - Some free energyThermodynamic free energyThe thermodynamic free energy is the amount of work that a thermodynamic system can perform. The concept is useful in the thermodynamics of chemical or thermal processes in engineering and science. The free energy is the internal energy of a system less the amount of energy that cannot be used to...
concepts - A Gibbsian ensemble ideal (basis of the statistical mechanicsStatistical mechanicsStatistical mechanics or statistical thermodynamicsThe terms statistical mechanics and statistical thermodynamics are used interchangeably...
field) - A phase rule
Opening section
Energy
In physics, energy is an indirectly observed quantity. It is often understood as the ability a physical system has to do work on other physical systems...
of the world
World
World is a common name for the whole of human civilization, specifically human experience, history, or the human condition in general, worldwide, i.e. anywhere on Earth....
is constant).
Entropy
Entropy is a thermodynamic property that can be used to determine the energy available for useful work in a thermodynamic process, such as in energy conversion devices, engines, or machines. Such devices can only be driven by convertible energy, and have a theoretical maximum efficiency when...
of the world tends to a maximum)
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- ClausiusRudolf ClausiusRudolf Julius Emanuel Clausius , was a German physicist and mathematician and is considered one of the central founders of the science of thermodynamics. By his restatement of Sadi Carnot's principle known as the Carnot cycle, he put the theory of heat on a truer and sounder basis...
- Clausius
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
The comprehension of the laws which govern any material system is greatly facilitated by considering the energy
Energy
In physics, energy is an indirectly observed quantity. It is often understood as the ability a physical system has to do work on other physical systems...
and entropy
Entropy
Entropy is a thermodynamic property that can be used to determine the energy available for useful work in a thermodynamic process, such as in energy conversion devices, engines, or machines. Such devices can only be driven by convertible energy, and have a theoretical maximum efficiency when...
of the system in the various states of which it is capable. As the difference of the values of the energy for any two states represents the combined amount of work
Work (thermodynamics)
In thermodynamics, work performed by a system is the energy transferred to another system that is measured by the external generalized mechanical constraints on the system. As such, thermodynamic work is a generalization of the concept of mechanical work in mechanics. Thermodynamic work encompasses...
and heat received or yielded by the system
Thermodynamic system
A thermodynamic system is a precisely defined macroscopic region of the universe, often called a physical system, that is studied using the principles of thermodynamics....
when it is brought from one state
Thermodynamic state
A thermodynamic state is a set of values of properties of a thermodynamic system that must be specified to reproduce the system. The individual parameters are known as state variables, state parameters or thermodynamic variables. Once a sufficient set of thermodynamic variables have been...
to the other, and the difference of entropy is the limit of all possible values of the integral:
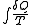
in which dQ denotes the element of heat received from external sources, and T is the temperature of the part of the system receiving it, the varying values of energy and entropy characterize in all that is essential the effect producible by the system in passing from one state to another. For by mechanical and thermodynamic contrivances, supposedly theoretically perfect, any supply of work and heat may be transformed into any other which does not differ from it either in the amount of work and heat taken together or in the value of the integral:
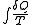
But it is not only in respect to the external relations of a system that its energy and entropy are of predominant importance. As in the case of simple mechanical systems, such as are discussed in theoretical mechanics, which are capable of only one kind of action upon external systems, namely the performance of mechanical work, the function which expresses the capability of the system of this kind of action also plays the leading part in the theory of equilibrium, the condition of equilibrium being that the variation of this function shall vanish, so in a thermodynamic system, such as all material systems are, which is capable of two different kinds of action upon external systems, the two functions which express the twofold capabilities of the system afford an almost equally simple criterion for equilibrium.
External Links
- Gibbs's paper "On the Equilibrium of Heterogeneous Substances"

