Radiometric dating
Encyclopedia
Radiometric dating is a technique used to date materials such as rocks, usually based on a comparison between the observed abundance of a naturally occurring radioactive isotope
and its decay products, using known decay rates. It is the principal source of information about the absolute age of rocks and other geological features, including the age of the Earth
itself, and can be used to date a wide range of natural and man-made materials. Together with stratigraphic principles
, radiometric dating methods are used in geochronology
to establish the geological time scale. Among the best-known techniques are radiocarbon dating
, potassium-argon dating
and uranium-lead dating
. By allowing the establishment of geological timescales, it provides a significant source of information about the ages of fossil
s and the deduced rates of evolution
ary change. Radiometric dating is also used to date archaeological
materials, including ancient artifacts.
Different methods of radiometric dating vary in the timescale over which they are accurate and the materials to which they can be applied.
 All ordinary matter
All ordinary matter
is made up of combinations of chemical element
s, each with its own atomic number
, indicating the number of proton
s in the atomic nucleus
. Additionally, elements may exist in different isotope
s, with each isotope of an element differing in the number of neutron
s in the nucleus. A particular isotope of a particular element is called a nuclide
. Some nuclides are inherently unstable. That is, at some point in time, an atom of such a nuclide will spontaneously transform into a different nuclide. This transformation may be accomplished in a number of different ways, including radioactive decay
, either by emission of particles (usually electron
s (beta decay
), positron
s or alpha particle
s) or by spontaneous fission
, and electron capture
.
While the moment in time at which a particular nucleus decays is unpredictable, a collection of atoms of a radioactive nuclide decays exponentially at a rate described by a parameter known as the half-life
, usually given in units of years when discussing dating techniques. After one half-life has elapsed, one half of the atoms of the nuclide in question will have decayed into a "daughter" nuclide or decay product
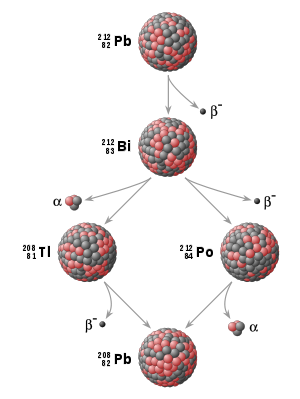
. In many cases, the daughter nuclide itself is radioactive, resulting in a decay chain
, eventually ending with the formation of a stable (nonradioactive) daughter nuclide; each step in such a chain is characterized by a distinct half-life. In these cases, usually the half-life of interest in radiometric dating is the longest one in the chain, which is the rate-limiting factor in the ultimate transformation of the radioactive nuclide into its stable daughter. Isotopic systems that have been exploited for radiometric dating have half-lives ranging from only about 10 years (e.g., tritium
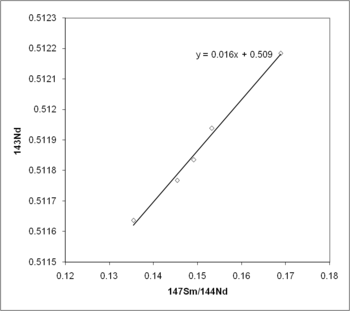
) to over 100 billion years (e.g., Samarium-147
).
In general, the half-life of a nuclide depends solely on its nuclear properties; it is not affected by external factors such as temperature
, pressure
, chemical environment, or presence of a magnetic
or electric field
. (For some nuclides which decay by the process of electron capture
, such as beryllium-7, strontium-85, and zirconium-89, the decay rate may be slightly affected by local electron density, therefore these isotopes may not be as suitable for radiometric dating.) But in general, the half-life of any nuclide is essentially a constant. Therefore, in any material containing a radioactive nuclide, the proportion of the original nuclide to its decay product(s) changes in a predictable way as the original nuclide decays over time. This predictability allows the relative abundances of related nuclides to be used as a clock
to measure the time from the incorporation of the original nuclide(s) into a material to the present.
 The basic equation of radiometric dating requires that neither the parent nuclide nor the daughter product can enter or leave the material after its formation. The possible confounding effects of contamination of parent and daughter isotopes have to be considered, as do the effects of any loss or gain of such isotopes since the sample was created. It is therefore essential to have as much information as possible about the material being dated and to check for possible signs of alteration
The basic equation of radiometric dating requires that neither the parent nuclide nor the daughter product can enter or leave the material after its formation. The possible confounding effects of contamination of parent and daughter isotopes have to be considered, as do the effects of any loss or gain of such isotopes since the sample was created. It is therefore essential to have as much information as possible about the material being dated and to check for possible signs of alteration
. Precision is enhanced if measurements are taken on multiple samples from different locations of the rock body. Alternatively, if several different minerals can be dated from the same sample and are assumed to be formed by the same event and were in equilibrium with the reservoir when they formed, they should form an isochron
. This can reduce the problem of contamination
. In uranium-lead dating
, the concordia diagram is used which also decreases the problem of nuclide loss. Finally, correlation between different isotopic dating methods may be required to confirm the age of a sample. For example, a study of the Amitsoq gneiss
es from western Greenland
used five different radiometric dating methods to examine twelve samples and achieved agreement to within 30 Ma on an age of 3,640 Ma.
Accurate radiometric dating generally requires that the parent has a long enough half-life that it will be present in significant amounts at the time of measurement (except as described below under "Dating with short-lived extinct radionuclides"), the half-life of the parent is accurately known, and enough of the daughter product is produced to be accurately measured and distinguished from the initial amount of the daughter present in the material. The procedures used to isolate and analyze the parent and daughter nuclides must be precise and accurate. This normally involves isotope ratio mass spectrometry.
The precision of a dating method depends in part on the half-life of the radioactive isotope involved. For instance, carbon-14 has a half-life of 5,730 years. After an organism has been dead for 60,000 years so little carbon-14 is left that accurate dating can not be established. On the other hand, the concentration of carbon-14 falls off so steeply that the age of relatively young remains can be determined precisely to within a few decades.
, setting the isotopic "clock" to zero. The temperature at which this happens is known as the closure temperature
or blocking temperature and is specific to a particular material and isotopic system. These temperatures are experimentally determined in the lab by artificially resetting sample minerals using a high-temperature furnace. As the mineral cools, the crystal structure begins to form and diffusion of isotopes is less easy. At a certain temperature, the crystal structure has formed sufficiently to prevent diffusion of isotopes. This temperature is what is known as closure temperature and represents the temperature below which the mineral is a closed system to isotopes. Thus an igneous or metamorphic rock or melt, which is slowly cooling, does not begin to exhibit measurable radioactive decay until it cools below the closure temperature. The age that can be calculated by radiometric dating is thus the time at which the rock or mineral cooled to closure temperature. Dating of different minerals and/or isotope systems (with differing closure temperatures) within the same rock can therefore enable the tracking of the thermal history of the rock in question with time, and thus the history of metamorphic events may become known in detail. This field is known as thermochronology
or thermochronometry.
 The mathematical expression that relates radioactive decay to geologic time, is
The mathematical expression that relates radioactive decay to geologic time, is
where
The equation is most conveniently expressed in terms of the measured quantity N(t) rather than the constant initial value No.
The above equation makes use of information on the composition of parent and daughter isotopes at the time the material being tested cooled below its closure temperature. This is well-established for most isotopic systems. However, construction of an isochron
does not require information on the original compositions, using merely the present ratios of the parent and daughter isotopes to a standard isotope. Plotting an isochron is used to solve the age equation graphically and calculate the age of the sample and the original composition.
as a method by which one might determine the age of the Earth
. In the century since then the techniques have been greatly improved and expanded. Dating can now be performed on samples as small as a nanogram using a mass spectrometer. The mass spectrometer was invented in the 1940s and began to be used in radiometric dating in the 1950s. The mass spectrometer operates by generating a beam of ionized atoms
from the sample under test. The ions then travel through a magnetic field, which diverts them into different sampling sensors, known as "Faraday cup
s", depending on their mass and level of ionization. On impact in the cups, the ions set up a very weak current that can be measured to determine the rate of impacts and the relative concentrations of different atoms in the beams.
 The uranium-lead radiometric dating
The uranium-lead radiometric dating
scheme has been refined to the point that the error margin in dates of rocks can be as low as less than two million years in two-and-a-half billion years. An error margin of 2–5 % has been achieved on younger Mesozoic
rocks.
Uranium-lead dating is often performed on the mineral
zircon
(ZrSiO4), though it can be used on other materials, such as baddeleyite
. Zircon and baddeleyite incorporate uranium atoms into their crystalline structure as substitutes for zirconium
, but strongly reject lead. It has a very high closure temperature, is resistant to mechanical weathering and is very chemically inert. Zircon also forms multiple crystal layers during metamorphic events, which each may record an isotopic age of the event. In situ micro-beam analysis can be achieved via laser ICP-MS
or SIMS
techniques.
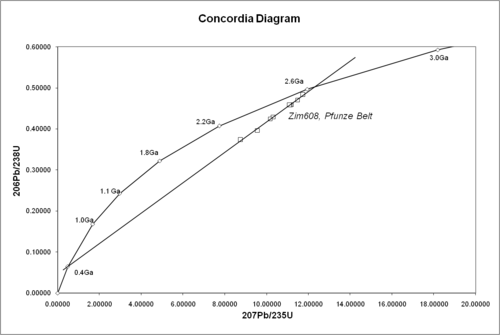
One of its great advantages is that any sample provides two clocks, one based on uranium-235's decay to lead-207 with a half-life of about 700 million years, and one based on uranium-238's decay to lead-206 with a half-life of about 4.5 billion years, providing a built-in crosscheck that allows accurate determination of the age of the sample even if some of the lead has been lost. This can be seen in the concordia diagram, where the samples plot along an errorchron (straight line) which intersects the concordia curve at the age of the sample.
of 1.06 x 1011 years. Accuracy levels of less than twenty million years in two-and-a-half billion years are achievable.
or positron
decay of potassium-40 to argon-40. Potassium-40 has a half-life of 1.3 billion years, and so this method is applicable to the oldest rocks. Radioactive potassium-40 is common in mica
s, feldspar
s, and hornblende
s, though the closure temperature is fairly low in these materials, about 125°C (mica) to 450°C (hornblende).
-87 to strontium
-87, with a half-life of 50 billion years. This scheme is used to date old igneous and metamorphic rocks, and has also been used to date lunar samples
. Closure temperatures are so high that they are not a concern. Rubidium-strontium dating is not as precise as the uranium-lead method, with errors of 30 to 50 million years for a 3-billion-year-old sample.
While uranium
is water-soluble, thorium
and protactinium
are not, and so they are selectively precipitated into ocean-floor sediment
s, from which their ratios are measured. The scheme has a range of several hundred thousand years.
 Carbon-14 is a radioactive isotope of carbon, with a half-life of 5,730 years, which is very short compared with the above isotopes. In other radiometric dating methods, the heavy parent isotopes were produced by nucleosynthesis
Carbon-14 is a radioactive isotope of carbon, with a half-life of 5,730 years, which is very short compared with the above isotopes. In other radiometric dating methods, the heavy parent isotopes were produced by nucleosynthesis
in supernovas, meaning that any parent isotope with a short half-life should be extinct by now. Carbon-14, though, is continuously created through collisions of neutrons generated by cosmic rays with nitrogen in the upper atmosphere
and thus remains at a near-constant level on Earth. The carbon-14 ends up as a trace component in atmospheric carbon dioxide
(CO2).
An organism acquires carbon during its lifetime. Plants acquire it through photosynthesis
, and animals acquire it from consumption of plants and other animals. When an organism dies, it ceases to take in new carbon-14, and the existing isotope decays with a characteristic half-life (5730 years). The proportion of carbon-14 left when the remains of the organism are examined provides an indication of the time elapsed since its death. The carbon-14 dating limit lies around 58,000 to 62,000 years.
The rate of creation of carbon-14 appears to be roughly constant, as cross-checks of carbon-14 dating with other dating methods show it gives consistent results. However, local eruptions of volcano
es or other events that give off large amounts of carbon dioxide can reduce local concentrations of carbon-14 and give inaccurate dates. The releases of carbon dioxide into the biosphere
as a consequence of industrialization have also depressed the proportion of carbon-14 by a few percent; conversely, the amount of carbon-14 was increased by above-ground nuclear bomb tests that were conducted into the early 1960s. Also, an increase in the solar wind
or the Earth's magnetic field
above the current value would depress the amount of carbon-14 created in the atmosphere. These effects are corrected for by the calibration of the radiocarbon dating scale.
 This involves inspection of a polished slice of a material to determine the density of "track" markings left in it by the spontaneous fission
This involves inspection of a polished slice of a material to determine the density of "track" markings left in it by the spontaneous fission
of uranium-238 impurities. The uranium content of the sample has to be known, but that can be determined by placing a plastic film over the polished slice of the material, and bombarding it with slow neutrons. This causes induced fission of 235U, as opposed to the spontaneous fission of 238U. The fission tracks produced by this process are recorded in the plastic film. The uranium content of the material can then be calculated from the number of tracks and the neutron flux
.
This scheme has application over a wide range of geologic dates. For dates up to a few million years mica
s, tektite
s (glass fragments from volcanic eruptions), and meteorites are best used. Older materials can be dated using zircon
, apatite
, titanite
, epidote
and garnet
which have a variable amount of uranium content. Because the fission tracks are healed by temperatures over about 200°C the technique has limitations as well as benefits. The technique has potential applications for detailing the thermal history of a deposit.
were produced by irradiation of seawater during atmospheric detonations of nuclear weapon
s between 1952 and 1958. The residence time of 36Cl in the atmosphere is about 1 week. Thus, as an event marker of 1950s water in soil
and ground water, 36Cl is also useful for dating waters less than 50 years before the present. 36Cl has seen use in other areas of the geological sciences, including dating ice and sediments.
.
At the beginning of the solar system, there were several relatively short-lived radionuclides like 26Al, 60Fe, 53Mn, and 129I present within the solar nebula. These radionuclides—possibly produced by the explosion of a supernova—are extinct today, but their decay products can be detected in very old material, such as that which constitutes meteorite
s. By measuring the decay products of extinct radionuclides with a mass spectrometer and using isochronplots, it is possible to determine relative ages of different events in the early history of the solar system. Dating methods based on extinct radionuclides can also be calibrated with the U-Pb method to give absolute ages. Thus both the approximate age and a high time resolution can be obtained. Generally a shorter half-life leads to a higher time resolution at the expense of timescale.
Since xenon is a volatile noble gas it can be assumed that there wasn't much of it in the rock to begin with. Since it is much rarer than iodine, it can be assumed that most of the 129Xe present in the rock is a by-product of 129I decay. By using the solar system's average xenon content as the natural abundance
, the excess of 129Xe to the abundance of 129I ratio can be derived.
of 720 000 years. The dating is simply a question of finding the deviation from the natural abundance
of 26Mg (the product of 26Al decay) in comparison with the ratio of the stable isotopes 27Al/24Mg.
The excess of 26Mg (often designated 26Mg* ) is found by comparing the 26Mg/27Mg ratio to that of other Solar System materials.
The 129I - 129Xe chronometer gives an estimate of the time period for formation of primitive meteorites of about 20 million years. Since some xenon might have escaped the rocks this formation period might be even shorter.
The 26Al - 26Mg chronometer, on the other hand, gives an estimate of the time period for formation of primitive meteorites of only a few million years (1.4 million years for Chondrule formation).
Isotope
Isotopes are variants of atoms of a particular chemical element, which have differing numbers of neutrons. Atoms of a particular element by definition must contain the same number of protons but may have a distinct number of neutrons which differs from atom to atom, without changing the designation...
and its decay products, using known decay rates. It is the principal source of information about the absolute age of rocks and other geological features, including the age of the Earth
Age of the Earth
The age of the Earth is 4.54 billion years This age is based on evidence from radiometric age dating of meteorite material and is consistent with the ages of the oldest-known terrestrial and lunar samples...
itself, and can be used to date a wide range of natural and man-made materials. Together with stratigraphic principles
Stratigraphy
Stratigraphy, a branch of geology, studies rock layers and layering . It is primarily used in the study of sedimentary and layered volcanic rocks....
, radiometric dating methods are used in geochronology
Geochronology
Geochronology is the science of determining the age of rocks, fossils, and sediments, within a certain degree of uncertainty inherent to the method used. A variety of dating methods are used by geologists to achieve this, and schemes of classification and terminology have been proposed...
to establish the geological time scale. Among the best-known techniques are radiocarbon dating
Radiocarbon dating
Radiocarbon dating is a radiometric dating method that uses the naturally occurring radioisotope carbon-14 to estimate the age of carbon-bearing materials up to about 58,000 to 62,000 years. Raw, i.e. uncalibrated, radiocarbon ages are usually reported in radiocarbon years "Before Present" ,...
, potassium-argon dating
Potassium-argon dating
Potassium–argon dating or K–Ar dating is a radiometric dating method used in geochronology and archeology. It is based on measurement of the product of the radioactive decay of an isotope of potassium into argon . Potassium is a common element found in many materials, such as micas, clay minerals,...
and uranium-lead dating
Uranium-lead dating
Uranium-lead is one of the oldest and most refined of the radiometric dating schemes, with a routine age range of about 1 million years to over 4.5 billion years, and with routine precisions in the 0.1-1 percent range...
. By allowing the establishment of geological timescales, it provides a significant source of information about the ages of fossil
Fossil
Fossils are the preserved remains or traces of animals , plants, and other organisms from the remote past...
s and the deduced rates of evolution
Evolution
Evolution is any change across successive generations in the heritable characteristics of biological populations. Evolutionary processes give rise to diversity at every level of biological organisation, including species, individual organisms and molecules such as DNA and proteins.Life on Earth...
ary change. Radiometric dating is also used to date archaeological
Archaeology
Archaeology, or archeology , is the study of human society, primarily through the recovery and analysis of the material culture and environmental data that they have left behind, which includes artifacts, architecture, biofacts and cultural landscapes...
materials, including ancient artifacts.
Different methods of radiometric dating vary in the timescale over which they are accurate and the materials to which they can be applied.
Radioactive decay

Matter
Matter is a general term for the substance of which all physical objects consist. Typically, matter includes atoms and other particles which have mass. A common way of defining matter is as anything that has mass and occupies volume...
is made up of combinations of chemical element
Chemical element
A chemical element is a pure chemical substance consisting of one type of atom distinguished by its atomic number, which is the number of protons in its nucleus. Familiar examples of elements include carbon, oxygen, aluminum, iron, copper, gold, mercury, and lead.As of November 2011, 118 elements...
s, each with its own atomic number
Atomic number
In chemistry and physics, the atomic number is the number of protons found in the nucleus of an atom and therefore identical to the charge number of the nucleus. It is conventionally represented by the symbol Z. The atomic number uniquely identifies a chemical element...
, indicating the number of proton
Proton
The proton is a subatomic particle with the symbol or and a positive electric charge of 1 elementary charge. One or more protons are present in the nucleus of each atom, along with neutrons. The number of protons in each atom is its atomic number....
s in the atomic nucleus
Atomic nucleus
The nucleus is the very dense region consisting of protons and neutrons at the center of an atom. It was discovered in 1911, as a result of Ernest Rutherford's interpretation of the famous 1909 Rutherford experiment performed by Hans Geiger and Ernest Marsden, under the direction of Rutherford. The...
. Additionally, elements may exist in different isotope
Isotope
Isotopes are variants of atoms of a particular chemical element, which have differing numbers of neutrons. Atoms of a particular element by definition must contain the same number of protons but may have a distinct number of neutrons which differs from atom to atom, without changing the designation...
s, with each isotope of an element differing in the number of neutron
Neutron
The neutron is a subatomic hadron particle which has the symbol or , no net electric charge and a mass slightly larger than that of a proton. With the exception of hydrogen, nuclei of atoms consist of protons and neutrons, which are therefore collectively referred to as nucleons. The number of...
s in the nucleus. A particular isotope of a particular element is called a nuclide
Nuclide
A nuclide is an atomic species characterized by the specific constitution of its nucleus, i.e., by its number of protons Z, its number of neutrons N, and its nuclear energy state....
. Some nuclides are inherently unstable. That is, at some point in time, an atom of such a nuclide will spontaneously transform into a different nuclide. This transformation may be accomplished in a number of different ways, including radioactive decay
Radioactive decay
Radioactive decay is the process by which an atomic nucleus of an unstable atom loses energy by emitting ionizing particles . The emission is spontaneous, in that the atom decays without any physical interaction with another particle from outside the atom...
, either by emission of particles (usually electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
s (beta decay
Beta decay
In nuclear physics, beta decay is a type of radioactive decay in which a beta particle is emitted from an atom. There are two types of beta decay: beta minus and beta plus. In the case of beta decay that produces an electron emission, it is referred to as beta minus , while in the case of a...
), positron
Positron
The positron or antielectron is the antiparticle or the antimatter counterpart of the electron. The positron has an electric charge of +1e, a spin of ½, and has the same mass as an electron...
s or alpha particle
Alpha particle
Alpha particles consist of two protons and two neutrons bound together into a particle identical to a helium nucleus, which is classically produced in the process of alpha decay, but may be produced also in other ways and given the same name...
s) or by spontaneous fission
Spontaneous fission
Spontaneous fission is a form of radioactive decay characteristic of very heavy isotopes. Because the nuclear binding energy reaches a maximum at a nuclear mass greater than about 60 atomic mass units , spontaneous breakdown into smaller nuclei and single particles becomes possible at heavier masses...
, and electron capture
Electron capture
Electron capture is a process in which a proton-rich nuclide absorbs an inner atomic electron and simultaneously emits a neutrino...
.
While the moment in time at which a particular nucleus decays is unpredictable, a collection of atoms of a radioactive nuclide decays exponentially at a rate described by a parameter known as the half-life
Half-life
Half-life, abbreviated t½, is the period of time it takes for the amount of a substance undergoing decay to decrease by half. The name was originally used to describe a characteristic of unstable atoms , but it may apply to any quantity which follows a set-rate decay.The original term, dating to...
, usually given in units of years when discussing dating techniques. After one half-life has elapsed, one half of the atoms of the nuclide in question will have decayed into a "daughter" nuclide or decay product
Decay product
In nuclear physics, a decay product is the remaining nuclide left over from radioactive decay. Radioactive decay often involves a sequence of steps...
. In many cases, the daughter nuclide itself is radioactive, resulting in a decay chain
Decay chain
In nuclear science, the decay chain refers to the radioactive decay of different discrete radioactive decay products as a chained series of transformations...
, eventually ending with the formation of a stable (nonradioactive) daughter nuclide; each step in such a chain is characterized by a distinct half-life. In these cases, usually the half-life of interest in radiometric dating is the longest one in the chain, which is the rate-limiting factor in the ultimate transformation of the radioactive nuclide into its stable daughter. Isotopic systems that have been exploited for radiometric dating have half-lives ranging from only about 10 years (e.g., tritium
Tritium
Tritium is a radioactive isotope of hydrogen. The nucleus of tritium contains one proton and two neutrons, whereas the nucleus of protium contains one proton and no neutrons...
) to over 100 billion years (e.g., Samarium-147
Isotopes of samarium
Naturally occurring samarium is composed of five stable isotopes, 144Sm, 149Sm, 150Sm, 152Sm and 154Sm, and two extremely long-lived radioisotopes, 147Sm and 148Sm , with 152Sm being the most abundant...
).
In general, the half-life of a nuclide depends solely on its nuclear properties; it is not affected by external factors such as temperature
Temperature
Temperature is a physical property of matter that quantitatively expresses the common notions of hot and cold. Objects of low temperature are cold, while various degrees of higher temperatures are referred to as warm or hot...
, pressure
Pressure
Pressure is the force per unit area applied in a direction perpendicular to the surface of an object. Gauge pressure is the pressure relative to the local atmospheric or ambient pressure.- Definition :...
, chemical environment, or presence of a magnetic
Magnetic field
A magnetic field is a mathematical description of the magnetic influence of electric currents and magnetic materials. The magnetic field at any given point is specified by both a direction and a magnitude ; as such it is a vector field.Technically, a magnetic field is a pseudo vector;...
or electric field
Electric field
In physics, an electric field surrounds electrically charged particles and time-varying magnetic fields. The electric field depicts the force exerted on other electrically charged objects by the electrically charged particle the field is surrounding...
. (For some nuclides which decay by the process of electron capture
Electron capture
Electron capture is a process in which a proton-rich nuclide absorbs an inner atomic electron and simultaneously emits a neutrino...
, such as beryllium-7, strontium-85, and zirconium-89, the decay rate may be slightly affected by local electron density, therefore these isotopes may not be as suitable for radiometric dating.) But in general, the half-life of any nuclide is essentially a constant. Therefore, in any material containing a radioactive nuclide, the proportion of the original nuclide to its decay product(s) changes in a predictable way as the original nuclide decays over time. This predictability allows the relative abundances of related nuclides to be used as a clock
Clock
A clock is an instrument used to indicate, keep, and co-ordinate time. The word clock is derived ultimately from the Celtic words clagan and clocca meaning "bell". A silent instrument missing such a mechanism has traditionally been known as a timepiece...
to measure the time from the incorporation of the original nuclide(s) into a material to the present.
Preconditions

Metasomatism
Metasomatism is the chemical alteration of a rock by hydrothermal and other fluids.Metasomatism can occur via the action of hydrothermal fluids from an igneous or metamorphic source. In the igneous environment, metasomatism creates skarns, greisen, and may affect hornfels in the contact...
. Precision is enhanced if measurements are taken on multiple samples from different locations of the rock body. Alternatively, if several different minerals can be dated from the same sample and are assumed to be formed by the same event and were in equilibrium with the reservoir when they formed, they should form an isochron
Isochron dating
Isochron dating is a common technique of radiometric dating and is applied to date certain events, such as crystallization, metamorphism, shock events, and differentiation of precursor melts, in the history of rocks...
. This can reduce the problem of contamination
Contamination
Contamination is the presence of a minor and unwanted constituent in material, physical body, natural environment, at a workplace, etc.-Specifics:"Contamination" also has more specific meanings in science:...
. In uranium-lead dating
Uranium-lead dating
Uranium-lead is one of the oldest and most refined of the radiometric dating schemes, with a routine age range of about 1 million years to over 4.5 billion years, and with routine precisions in the 0.1-1 percent range...
, the concordia diagram is used which also decreases the problem of nuclide loss. Finally, correlation between different isotopic dating methods may be required to confirm the age of a sample. For example, a study of the Amitsoq gneiss
Gneiss
Gneiss is a common and widely distributed type of rock formed by high-grade regional metamorphic processes from pre-existing formations that were originally either igneous or sedimentary rocks.-Etymology:...
es from western Greenland
Greenland
Greenland is an autonomous country within the Kingdom of Denmark, located between the Arctic and Atlantic Oceans, east of the Canadian Arctic Archipelago. Though physiographically a part of the continent of North America, Greenland has been politically and culturally associated with Europe for...
used five different radiometric dating methods to examine twelve samples and achieved agreement to within 30 Ma on an age of 3,640 Ma.
Accurate radiometric dating generally requires that the parent has a long enough half-life that it will be present in significant amounts at the time of measurement (except as described below under "Dating with short-lived extinct radionuclides"), the half-life of the parent is accurately known, and enough of the daughter product is produced to be accurately measured and distinguished from the initial amount of the daughter present in the material. The procedures used to isolate and analyze the parent and daughter nuclides must be precise and accurate. This normally involves isotope ratio mass spectrometry.
The precision of a dating method depends in part on the half-life of the radioactive isotope involved. For instance, carbon-14 has a half-life of 5,730 years. After an organism has been dead for 60,000 years so little carbon-14 is left that accurate dating can not be established. On the other hand, the concentration of carbon-14 falls off so steeply that the age of relatively young remains can be determined precisely to within a few decades.
Closure temperature
If a material that selectively rejects the daughter nuclide is heated, any daughter nuclides that have been accumulated over time will be lost through diffusionDiffusion
Molecular diffusion, often called simply diffusion, is the thermal motion of all particles at temperatures above absolute zero. The rate of this movement is a function of temperature, viscosity of the fluid and the size of the particles...
, setting the isotopic "clock" to zero. The temperature at which this happens is known as the closure temperature
Closure temperature
In radiometric dating, closure temperature or blocking temperature refers to the temperature of a system, such as a mineral, at the time given by its radiometric date. In physical terms, the closure temperature at which a system has cooled so that there is no longer any exchange of parent or...
or blocking temperature and is specific to a particular material and isotopic system. These temperatures are experimentally determined in the lab by artificially resetting sample minerals using a high-temperature furnace. As the mineral cools, the crystal structure begins to form and diffusion of isotopes is less easy. At a certain temperature, the crystal structure has formed sufficiently to prevent diffusion of isotopes. This temperature is what is known as closure temperature and represents the temperature below which the mineral is a closed system to isotopes. Thus an igneous or metamorphic rock or melt, which is slowly cooling, does not begin to exhibit measurable radioactive decay until it cools below the closure temperature. The age that can be calculated by radiometric dating is thus the time at which the rock or mineral cooled to closure temperature. Dating of different minerals and/or isotope systems (with differing closure temperatures) within the same rock can therefore enable the tracking of the thermal history of the rock in question with time, and thus the history of metamorphic events may become known in detail. This field is known as thermochronology
Thermochronology
Thermochronology is the study of the thermal evolution of a region of a planet. Thermochronologists use radiometric dating along with the closure temperatures that represent the temperature of the mineral being studied at the time given by the date recorded, to understand the thermal history of a...
or thermochronometry.
The age equation

- D = D0 + N(t) (eλt − 1)
where
- t is age of the sample,
- D is number of atoms of the daughter isotope in the sample,
- D0 is number of atoms of the daughter isotope in the original composition,
- N is number of atoms of the parent isotope in the sample at time t (the present), given by N(t) = Noe-λt, and
- λ is the decay constant of the parent isotope, equal to the inverse of the radioactive half-lifeHalf-lifeHalf-life, abbreviated t½, is the period of time it takes for the amount of a substance undergoing decay to decrease by half. The name was originally used to describe a characteristic of unstable atoms , but it may apply to any quantity which follows a set-rate decay.The original term, dating to...
of the parent isotope times the natural logarithm of 2.
The equation is most conveniently expressed in terms of the measured quantity N(t) rather than the constant initial value No.
The above equation makes use of information on the composition of parent and daughter isotopes at the time the material being tested cooled below its closure temperature. This is well-established for most isotopic systems. However, construction of an isochron
Isochron dating
Isochron dating is a common technique of radiometric dating and is applied to date certain events, such as crystallization, metamorphism, shock events, and differentiation of precursor melts, in the history of rocks...
does not require information on the original compositions, using merely the present ratios of the parent and daughter isotopes to a standard isotope. Plotting an isochron is used to solve the age equation graphically and calculate the age of the sample and the original composition.
Modern dating methods
Radiometric dating has been carried out since 1905 when it was invented by Ernest RutherfordErnest Rutherford
Ernest Rutherford, 1st Baron Rutherford of Nelson OM, FRS was a New Zealand-born British chemist and physicist who became known as the father of nuclear physics...
as a method by which one might determine the age of the Earth
Age of the Earth
The age of the Earth is 4.54 billion years This age is based on evidence from radiometric age dating of meteorite material and is consistent with the ages of the oldest-known terrestrial and lunar samples...
. In the century since then the techniques have been greatly improved and expanded. Dating can now be performed on samples as small as a nanogram using a mass spectrometer. The mass spectrometer was invented in the 1940s and began to be used in radiometric dating in the 1950s. The mass spectrometer operates by generating a beam of ionized atoms
Ion
An ion is an atom or molecule in which the total number of electrons is not equal to the total number of protons, giving it a net positive or negative electrical charge. The name was given by physicist Michael Faraday for the substances that allow a current to pass between electrodes in a...
from the sample under test. The ions then travel through a magnetic field, which diverts them into different sampling sensors, known as "Faraday cup
Faraday cup
A Faraday cup is a metal cup designed to catch charged particles in vacuum. The resulting current can be measured and used to determine the number of ions or electrons hitting the cup...
s", depending on their mass and level of ionization. On impact in the cups, the ions set up a very weak current that can be measured to determine the rate of impacts and the relative concentrations of different atoms in the beams.
Uranium-lead dating method

Uranium-lead dating
Uranium-lead is one of the oldest and most refined of the radiometric dating schemes, with a routine age range of about 1 million years to over 4.5 billion years, and with routine precisions in the 0.1-1 percent range...
scheme has been refined to the point that the error margin in dates of rocks can be as low as less than two million years in two-and-a-half billion years. An error margin of 2–5 % has been achieved on younger Mesozoic
Mesozoic
The Mesozoic era is an interval of geological time from about 250 million years ago to about 65 million years ago. It is often referred to as the age of reptiles because reptiles, namely dinosaurs, were the dominant terrestrial and marine vertebrates of the time...
rocks.
Uranium-lead dating is often performed on the mineral
Mineral
A mineral is a naturally occurring solid chemical substance formed through biogeochemical processes, having characteristic chemical composition, highly ordered atomic structure, and specific physical properties. By comparison, a rock is an aggregate of minerals and/or mineraloids and does not...
zircon
Zircon
Zircon is a mineral belonging to the group of nesosilicates. Its chemical name is zirconium silicate and its corresponding chemical formula is ZrSiO4. A common empirical formula showing some of the range of substitution in zircon is 1–x4x–y...
(ZrSiO4), though it can be used on other materials, such as baddeleyite
Baddeleyite
Baddeleyite is a rare zirconium oxide mineral , occurring in a variety of monoclinic prismatic crystal forms. It is transparent to translucent, has high indices of refraction, and ranges from colorless to yellow, green, and dark brown. Baddeleyite is a refractory mineral, with a melting point of...
. Zircon and baddeleyite incorporate uranium atoms into their crystalline structure as substitutes for zirconium
Zirconium
Zirconium is a chemical element with the symbol Zr and atomic number 40. The name of zirconium is taken from the mineral zircon. Its atomic mass is 91.224. It is a lustrous, grey-white, strong transition metal that resembles titanium...
, but strongly reject lead. It has a very high closure temperature, is resistant to mechanical weathering and is very chemically inert. Zircon also forms multiple crystal layers during metamorphic events, which each may record an isotopic age of the event. In situ micro-beam analysis can be achieved via laser ICP-MS
ICP-MS
Inductively coupled plasma mass spectrometry is a type of mass spectrometry that is highly sensitive and capable of the determination of a range of metals and several non-metals at concentrations below one part in 1012...
or SIMS
Secondary ion mass spectrometry
Secondary ion mass spectrometry is a technique used in materials science and surface science to analyze the composition of solid surfaces and thin films by sputtering the surface of the specimen with a focused primary ion beam and collecting and analyzing ejected secondary ions...
techniques.
One of its great advantages is that any sample provides two clocks, one based on uranium-235's decay to lead-207 with a half-life of about 700 million years, and one based on uranium-238's decay to lead-206 with a half-life of about 4.5 billion years, providing a built-in crosscheck that allows accurate determination of the age of the sample even if some of the lead has been lost. This can be seen in the concordia diagram, where the samples plot along an errorchron (straight line) which intersects the concordia curve at the age of the sample.
Samarium-neodymium dating method
This involves the alpha-decay of 147Sm to 143Nd with a half-lifeHalf-life
Half-life, abbreviated t½, is the period of time it takes for the amount of a substance undergoing decay to decrease by half. The name was originally used to describe a characteristic of unstable atoms , but it may apply to any quantity which follows a set-rate decay.The original term, dating to...
of 1.06 x 1011 years. Accuracy levels of less than twenty million years in two-and-a-half billion years are achievable.
Potassium-argon dating method
This involves electron captureElectron capture
Electron capture is a process in which a proton-rich nuclide absorbs an inner atomic electron and simultaneously emits a neutrino...
or positron
Positron
The positron or antielectron is the antiparticle or the antimatter counterpart of the electron. The positron has an electric charge of +1e, a spin of ½, and has the same mass as an electron...
decay of potassium-40 to argon-40. Potassium-40 has a half-life of 1.3 billion years, and so this method is applicable to the oldest rocks. Radioactive potassium-40 is common in mica
Mica
The mica group of sheet silicate minerals includes several closely related materials having highly perfect basal cleavage. All are monoclinic, with a tendency towards pseudohexagonal crystals, and are similar in chemical composition...
s, feldspar
Feldspar
Feldspars are a group of rock-forming tectosilicate minerals which make up as much as 60% of the Earth's crust....
s, and hornblende
Hornblende
Hornblende is a complex inosilicate series of minerals .It is not a recognized mineral in its own right, but the name is used as a general or field term, to refer to a dark amphibole....
s, though the closure temperature is fairly low in these materials, about 125°C (mica) to 450°C (hornblende).
Rubidium-strontium dating method
This is based on the beta decay of rubidiumRubidium
Rubidium is a chemical element with the symbol Rb and atomic number 37. Rubidium is a soft, silvery-white metallic element of the alkali metal group. Its atomic mass is 85.4678. Elemental rubidium is highly reactive, with properties similar to those of other elements in group 1, such as very rapid...
-87 to strontium
Strontium
Strontium is a chemical element with the symbol Sr and the atomic number 38. An alkaline earth metal, strontium is a soft silver-white or yellowish metallic element that is highly reactive chemically. The metal turns yellow when exposed to air. It occurs naturally in the minerals celestine and...
-87, with a half-life of 50 billion years. This scheme is used to date old igneous and metamorphic rocks, and has also been used to date lunar samples
Moon rock
Moon rock describes rock that formed on the Earth's moon. The term is also loosely applied to other lunar materials collected during the course of human exploration of the Moon.The rocks collected from the Moon are measured by radiometric dating techniques...
. Closure temperatures are so high that they are not a concern. Rubidium-strontium dating is not as precise as the uranium-lead method, with errors of 30 to 50 million years for a 3-billion-year-old sample.
Uranium-thorium dating method
A relatively short-range dating technique is based on the decay of uranium-234 into thorium-230, a substance with a half-life of about 80,000 years. It is accompanied by a sister process, in which uranium-235 decays into protactinium-231, which has a half-life of 34,300 years.While uranium
Uranium
Uranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons...
is water-soluble, thorium
Thorium
Thorium is a natural radioactive chemical element with the symbol Th and atomic number 90. It was discovered in 1828 and named after Thor, the Norse god of thunder....
and protactinium
Protactinium
Protactinium is a chemical element with the symbol Pa and atomic number 91. It is a dense, silvery-gray metal which readily reacts with oxygen, water vapor and inorganic acids. It forms various chemical compounds where protactinium is usually present in the oxidation state +5, but can also assume...
are not, and so they are selectively precipitated into ocean-floor sediment
Sediment
Sediment is naturally occurring material that is broken down by processes of weathering and erosion, and is subsequently transported by the action of fluids such as wind, water, or ice, and/or by the force of gravity acting on the particle itself....
s, from which their ratios are measured. The scheme has a range of several hundred thousand years.
Radiocarbon dating method

Nucleosynthesis
Nucleosynthesis is the process of creating new atomic nuclei from pre-existing nucleons . It is thought that the primordial nucleons themselves were formed from the quark–gluon plasma from the Big Bang as it cooled below two trillion degrees...
in supernovas, meaning that any parent isotope with a short half-life should be extinct by now. Carbon-14, though, is continuously created through collisions of neutrons generated by cosmic rays with nitrogen in the upper atmosphere
Atmosphere
An atmosphere is a layer of gases that may surround a material body of sufficient mass, and that is held in place by the gravity of the body. An atmosphere may be retained for a longer duration, if the gravity is high and the atmosphere's temperature is low...
and thus remains at a near-constant level on Earth. The carbon-14 ends up as a trace component in atmospheric carbon dioxide
Carbon dioxide
Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
(CO2).
An organism acquires carbon during its lifetime. Plants acquire it through photosynthesis
Photosynthesis
Photosynthesis is a chemical process that converts carbon dioxide into organic compounds, especially sugars, using the energy from sunlight. Photosynthesis occurs in plants, algae, and many species of bacteria, but not in archaea. Photosynthetic organisms are called photoautotrophs, since they can...
, and animals acquire it from consumption of plants and other animals. When an organism dies, it ceases to take in new carbon-14, and the existing isotope decays with a characteristic half-life (5730 years). The proportion of carbon-14 left when the remains of the organism are examined provides an indication of the time elapsed since its death. The carbon-14 dating limit lies around 58,000 to 62,000 years.
The rate of creation of carbon-14 appears to be roughly constant, as cross-checks of carbon-14 dating with other dating methods show it gives consistent results. However, local eruptions of volcano
Volcano
2. Bedrock3. Conduit 4. Base5. Sill6. Dike7. Layers of ash emitted by the volcano8. Flank| 9. Layers of lava emitted by the volcano10. Throat11. Parasitic cone12. Lava flow13. Vent14. Crater15...
es or other events that give off large amounts of carbon dioxide can reduce local concentrations of carbon-14 and give inaccurate dates. The releases of carbon dioxide into the biosphere
Biosphere
The biosphere is the global sum of all ecosystems. It can also be called the zone of life on Earth, a closed and self-regulating system...
as a consequence of industrialization have also depressed the proportion of carbon-14 by a few percent; conversely, the amount of carbon-14 was increased by above-ground nuclear bomb tests that were conducted into the early 1960s. Also, an increase in the solar wind
Solar wind
The solar wind is a stream of charged particles ejected from the upper atmosphere of the Sun. It mostly consists of electrons and protons with energies usually between 1.5 and 10 keV. The stream of particles varies in temperature and speed over time...
or the Earth's magnetic field
Magnetic field
A magnetic field is a mathematical description of the magnetic influence of electric currents and magnetic materials. The magnetic field at any given point is specified by both a direction and a magnitude ; as such it is a vector field.Technically, a magnetic field is a pseudo vector;...
above the current value would depress the amount of carbon-14 created in the atmosphere. These effects are corrected for by the calibration of the radiocarbon dating scale.
Fission track dating method

Spontaneous fission
Spontaneous fission is a form of radioactive decay characteristic of very heavy isotopes. Because the nuclear binding energy reaches a maximum at a nuclear mass greater than about 60 atomic mass units , spontaneous breakdown into smaller nuclei and single particles becomes possible at heavier masses...
of uranium-238 impurities. The uranium content of the sample has to be known, but that can be determined by placing a plastic film over the polished slice of the material, and bombarding it with slow neutrons. This causes induced fission of 235U, as opposed to the spontaneous fission of 238U. The fission tracks produced by this process are recorded in the plastic film. The uranium content of the material can then be calculated from the number of tracks and the neutron flux
Flux
In the various subfields of physics, there exist two common usages of the term flux, both with rigorous mathematical frameworks.* In the study of transport phenomena , flux is defined as flow per unit area, where flow is the movement of some quantity per time...
.
This scheme has application over a wide range of geologic dates. For dates up to a few million years mica
Mica
The mica group of sheet silicate minerals includes several closely related materials having highly perfect basal cleavage. All are monoclinic, with a tendency towards pseudohexagonal crystals, and are similar in chemical composition...
s, tektite
Tektite
Tektites are natural glass rocks up to a few centimeters in size, which most scientists argue were formed by the impact of large meteorites on Earth's surface. Tektites are typically black or olive-green, and their shape varies from rounded to irregular.Tektites are among the "driest" rocks, with...
s (glass fragments from volcanic eruptions), and meteorites are best used. Older materials can be dated using zircon
Zircon
Zircon is a mineral belonging to the group of nesosilicates. Its chemical name is zirconium silicate and its corresponding chemical formula is ZrSiO4. A common empirical formula showing some of the range of substitution in zircon is 1–x4x–y...
, apatite
Apatite
Apatite is a group of phosphate minerals, usually referring to hydroxylapatite, fluorapatite, chlorapatite and bromapatite, named for high concentrations of OH−, F−, Cl− or Br− ions, respectively, in the crystal...
, titanite
Titanite
Titanite, or sphene , is a calcium titanium nesosilicate mineral, CaTiSiO5. Trace impurities of iron and aluminium are typically present...
, epidote
Epidote
Epidote is a calcium aluminium iron sorosilicate mineral, Ca2Al2O, crystallizing in the monoclinic system. Well-developed crystals are of frequent occurrence: they are commonly prismatic in habit, the direction of elongation being perpendicular to the single plane of symmetry. The faces are often...
and garnet
Garnet
The garnet group includes a group of minerals that have been used since the Bronze Age as gemstones and abrasives. The name "garnet" may come from either the Middle English word gernet meaning 'dark red', or the Latin granatus , possibly a reference to the Punica granatum , a plant with red seeds...
which have a variable amount of uranium content. Because the fission tracks are healed by temperatures over about 200°C the technique has limitations as well as benefits. The technique has potential applications for detailing the thermal history of a deposit.
Chlorine-36 dating method
Large amounts of otherwise rare 36ClChlorine-36
Chlorine-36 is an isotope of chlorine. Chlorine has two stable isotopes and one radioactive environmental isotope: the cosmogenic isotope 36Cl. The ratio of 36Cl to stable 37Cl in the environment is ~700 × 10−15. Its half-life is 301,000 ± 4,000 years. The long half-life of 36Cl makes it...
were produced by irradiation of seawater during atmospheric detonations of nuclear weapon
Nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission or a combination of fission and fusion. Both reactions release vast quantities of energy from relatively small amounts of matter. The first fission bomb test released the same amount...
s between 1952 and 1958. The residence time of 36Cl in the atmosphere is about 1 week. Thus, as an event marker of 1950s water in soil
Soil
Soil is a natural body consisting of layers of mineral constituents of variable thicknesses, which differ from the parent materials in their morphological, physical, chemical, and mineralogical characteristics...
and ground water, 36Cl is also useful for dating waters less than 50 years before the present. 36Cl has seen use in other areas of the geological sciences, including dating ice and sediments.
Luminescence dating methods
Natural sources of radiation in the environment knock loose electrons in, say, a piece of pottery, and these electrons accumulate in defects in the material's crystal lattice structure. Heating or illuminating the object will release the captured electrons, producing a luminescence. When the sample is heated, at a certain temperature it will glow from the emission of electrons released from the defects, and this glow can be used to estimate the age of the sample to a threshold of approximately 15 percent of its true age. The date of a rock is reset when volcanic activity remelts it. The date of a piece of pottery is reset by the heat of the kiln. Typically temperatures greater than 400 degrees Celsius will reset the "clock". This is termed thermoluminescenceThermoluminescence
Thermoluminescence is a form of luminescence that is exhibited by certain crystalline materials, such as some minerals, when previously absorbed energy from electromagnetic radiation or other ionizing radiation is re-emitted as light upon heating of the material...
.
Other methods
Other methods include:- argon-argonArgon-argon datingArgon-argon dating is a radiometric dating method invented to supersede potassium-argon dating in accuracy. The older method required two samples for dating while the newer method requires only one...
(Ar-Ar) - iodine-xenon (I-Xe)
- lanthanum-barium (La-Ba)
- lead-leadLead-lead datingLead-lead dating is a method for dating geological samples, normally based on 'whole-rock' samples of material such as granite. For most dating requirements it has been superseded by uranium-lead dating , but in certain specialized situations it is more important than U-Pb dating.-Decay equations...
(Pb-Pb) - lutetium-hafnium (Lu-Hf)
- neon-neon (Ne-Ne)
- rhenium-osmiumRhenium-osmium datingRhenium-Osmium dating is a form of radiometric dating based on the beta decay of the isotope 187Re to 187Os. This normally occurs with a half-life of 41.6 × 109 y, but studies using fully ionised 187Re atoms have found that this can decrease to only 33 y...
(Re-Os) - uranium-lead-helium (U-Pb-He)
- uranium-uraniumUranium-uranium datingUranium-uranium dating is a radiometric dating technique which compares two isotopes of uranium in a sample: 234U and 238U. 234U/238U dating is one of several radiometric dating techniques exploiting the uranium radioactive decay series, in which 238U undergoes 14 alpha and beta decay events...
(U-U)
Dating with short-lived extinct radionuclides
Absolute radiometric dating requires a measurable fraction of parent nucleus to remain in the sample rock. For rocks dating back to the beginning of the solar system, this requires extremely long-lived parent isotopes, making measurement of such rocks' exact ages imprecise. To be able to distinguish the relative ages of rocks from such old material, and to get a better time resolution than that available from long-lived isotopes, short-lived isotopes that are no longer present in the rock can be used.At the beginning of the solar system, there were several relatively short-lived radionuclides like 26Al, 60Fe, 53Mn, and 129I present within the solar nebula. These radionuclides—possibly produced by the explosion of a supernova—are extinct today, but their decay products can be detected in very old material, such as that which constitutes meteorite
Meteorite
A meteorite is a natural object originating in outer space that survives impact with the Earth's surface. Meteorites can be big or small. Most meteorites derive from small astronomical objects called meteoroids, but they are also sometimes produced by impacts of asteroids...
s. By measuring the decay products of extinct radionuclides with a mass spectrometer and using isochronplots, it is possible to determine relative ages of different events in the early history of the solar system. Dating methods based on extinct radionuclides can also be calibrated with the U-Pb method to give absolute ages. Thus both the approximate age and a high time resolution can be obtained. Generally a shorter half-life leads to a higher time resolution at the expense of timescale.
The 129I - 129Xe chronometer
129I beta-decays to 129Xe with a half-life of 16 million years.Since xenon is a volatile noble gas it can be assumed that there wasn't much of it in the rock to begin with. Since it is much rarer than iodine, it can be assumed that most of the 129Xe present in the rock is a by-product of 129I decay. By using the solar system's average xenon content as the natural abundance
Natural abundance
In chemistry, natural abundance refers to the abundance of isotopes of a chemical element as naturally found on a planet. The relative atomic mass of these isotopes is the atomic weight listed for the element in the periodic table...
, the excess of 129Xe to the abundance of 129I ratio can be derived.
The 26Al - 26Mg chronometer
Another example of short-lived extinct radionuclide dating is the 26Al - 26Mg chronometer, which can be used to estimate the relative ages of chondrules. 26Al decays to 26Mg with a half-lifeHalf-life
Half-life, abbreviated t½, is the period of time it takes for the amount of a substance undergoing decay to decrease by half. The name was originally used to describe a characteristic of unstable atoms , but it may apply to any quantity which follows a set-rate decay.The original term, dating to...
of 720 000 years. The dating is simply a question of finding the deviation from the natural abundance
Natural abundance
In chemistry, natural abundance refers to the abundance of isotopes of a chemical element as naturally found on a planet. The relative atomic mass of these isotopes is the atomic weight listed for the element in the periodic table...
of 26Mg (the product of 26Al decay) in comparison with the ratio of the stable isotopes 27Al/24Mg.
The excess of 26Mg (often designated 26Mg* ) is found by comparing the 26Mg/27Mg ratio to that of other Solar System materials.
The 129I - 129Xe chronometer gives an estimate of the time period for formation of primitive meteorites of about 20 million years. Since some xenon might have escaped the rocks this formation period might be even shorter.
The 26Al - 26Mg chronometer, on the other hand, gives an estimate of the time period for formation of primitive meteorites of only a few million years (1.4 million years for Chondrule formation).
See also
- Isochron datingIsochron datingIsochron dating is a common technique of radiometric dating and is applied to date certain events, such as crystallization, metamorphism, shock events, and differentiation of precursor melts, in the history of rocks...
- Isotope geochemistryIsotope geochemistryIsotope geochemistry is an aspect of geology based upon study of the relative and absolute concentrations of the elements and their isotopes in the Earth. Variations in the abundance of these isotopes, typically measured with an isotope ratio mass spectrometer or an accelerator mass spectrometer,...
- Isotopic signatureIsotopic signatureAn isotopic signature is a ratio of stable or unstable isotopes of particular elements found in an investigated material...
- Paleopedological recordPaleopedological recordThe paleopedological record is, essentially, the fossil record of soils. The paleopedological record consists chiefly of paleosols buried by flood sediments, or preserved at geological unconformities, especially plateau escarpments or sides of river valleys...
- Radioactivity
- RadiohaloRadiohaloRadiohalos or pleochroic halos are microscopic, spherical shells of discolouration within minerals such as biotite that occur in granite and other igneous rocks. The shells are zones of radiation damage caused by the inclusion of minute radioactive crystals within the host crystal structure...
- Sensitive High Resolution Ion Microprobe (SHRIMP)SHRIMPThe sensitive high resolution ion microprobe is a large-diameter, double-focusing secondary ion mass spectrometer sector instrument produced by Australian Scientific Instruments in Canberra, Australia...

