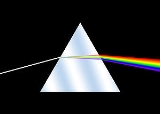
Spectrum analysis
Encyclopedia
Spectrum, also known as emission spectrochemical analysis, is the original scientific method of charting and analyzing the chemical properties of matter and gases by looking at the bands in their optical spectrum. The empirical laws of spectrum analysis are commonly known as Kirchhoff's Three Laws of Spectroscopy as follows:
 Anders Jonas Ångström
Anders Jonas Ångström
, Charles Wheatstone
, Gustav Kirchhoff
, Robert Bunsen
, William Crookes
, and others contributed to early spectroscopy through the discovery and exploitation of spectral emission lines.
In 1835, Charles Wheatstone
reported that different metals could be easily distinguished by the different bright lines in the emission spectra of their sparks, thereby launching the science of spectrum analysis.
In 1854, David Alter
, a scientist of Freeport, Pennsylvania
, published a work that included the spectral radiance
properties for twelve metals, titled On Certain Physical Properties of Light Produced by the Combustion of Different Metals in an Electric Spark Refracted by a Prism.
Dr. Alter began studying the optical properties of matter ever since finding a piece of melted, prism
atic glass in the debris of the great Pittsburgh fire of 1845. By 1855, Alter published another article that expanded his original theory by including six gases, including the first discovery of what came to be named the Balmer lines of hydrogen
. Alter's article contains a paragraph where he visualized the application of spectrum analysis to astronomy
, mentioning the study and detection of elements in the combustion of shooting stars or luminous meteors, and daguerreotyped
the dark lines of the solar spectrum. Alter's spectral discoveries were noted in various scientific publications in France, Germany, and Switzerland from 1854 to 1860.
In 1853, the Swedish
physisist Anders Jonas Ångström
presented similar theories about gases having spectra in his work: Optiska Undersökningar to the Royal Swedish Academy of Sciences
—pointing out that the electric spark yields two superposed spectra. Ångström postulated that an incandescent gas emits luminous rays of the same refrangibility as those it can absorb—a fundamental principle of spectrum analysis.
In 1860, German physicist Gustav Kirchhoff
and chemist Robert Bunsen
published their own findings on the spectra of eight metals and identified these metals in natural elements. Kirchoff went on to contribute fundamental research on the nature of spectral absorption
. Spectrum analysis was then grouped by Kirchhoff into the three fundamental laws commonly called Kirchoff's Laws, these laws integrated both Alter and Ångström's discoveries of radiance
and emission with Kirchhoff's fundamental discoveries of absorption.
Johann Balmer discovered in 1885 that the four visible lines of hydrogen were part of a series
that could be expressed in terms of integers. This was followed a few years later by the Rydberg formula
, which described additional series of lines.
In the 1860's William Huggins
and his wife Margaret used spectroscopy to determine that the stars were composed of the same elements as found on earth. They also used the non-relativistic Doppler shift
(redshift
) equation on the spectrum of the star Sirius
in 1868 to determine its axial speed. They were the first to take a spectrum of a planetary nebula when the Cat's Eye Nebula
(NGC 6543) was analyzed. Using spectral techniques, they were able to distinguish nebulae
from galaxies
.
In the early twentieth century, spectrum analysis led to "atomic spectroscopy
" and quantum mechanics
.
- An incandescent solid, liquid, or gas under high pressure emits a continuous spectrumContinuous spectrumThe spectrum of a linear operator is commonly divided into three parts: point spectrum, continuous spectrum, and residual spectrum.If H is a topological vector space and A:H \to H is a linear map, the spectrum of A is the set of complex numbers \lambda such that A - \lambda I : H \to H is not...
. - A hot gas under low pressure emits a "bright-line" or emission-line spectrumEmission spectrumThe emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted by the element's atoms or the compound's molecules when they are returned to a lower energy state....
. - A continuous spectrum source viewed through a cool, low-density gas produces an absorption-line spectrum.
Origins

Anders Jonas Ångström
Anders Jonas Ångström was a Swedish physicist and one of the founders of the science of spectroscopy.-Biography:...
, Charles Wheatstone
Charles Wheatstone
Sir Charles Wheatstone FRS , was an English scientist and inventor of many scientific breakthroughs of the Victorian era, including the English concertina, the stereoscope , and the Playfair cipher...
, Gustav Kirchhoff
Gustav Kirchhoff
Gustav Robert Kirchhoff was a German physicist who contributed to the fundamental understanding of electrical circuits, spectroscopy, and the emission of black-body radiation by heated objects...
, Robert Bunsen
Robert Bunsen
Robert Wilhelm Eberhard Bunsen was a German chemist. He investigated emission spectra of heated elements, and discovered caesium and rubidium with Gustav Kirchhoff. Bunsen developed several gas-analytical methods, was a pioneer in photochemistry, and did early work in the field of organoarsenic...
, William Crookes
William Crookes
Sir William Crookes, OM, FRS was a British chemist and physicist who attended the Royal College of Chemistry, London, and worked on spectroscopy...
, and others contributed to early spectroscopy through the discovery and exploitation of spectral emission lines.
In 1835, Charles Wheatstone
Charles Wheatstone
Sir Charles Wheatstone FRS , was an English scientist and inventor of many scientific breakthroughs of the Victorian era, including the English concertina, the stereoscope , and the Playfair cipher...
reported that different metals could be easily distinguished by the different bright lines in the emission spectra of their sparks, thereby launching the science of spectrum analysis.
In 1854, David Alter
David Alter
David Alter was a prominent American inventor and scientist of the 19th century. He was born in Westmoreland County, Pennsylvania and graduated from the Reformed Medical School in New York City. He had German and Swiss ancestry.-Inventions:Dr...
, a scientist of Freeport, Pennsylvania
Freeport, Pennsylvania
Freeport is a borough in Armstrong County, Pennsylvania, United States; it is situated along the Allegheny River in the southwest corner of the county. The population was 1,962 at the 2000 Census. Freeport received its name from David Todd, who declared it to be a free-port where no man would be...
, published a work that included the spectral radiance
Radiance
Radiance and spectral radiance are radiometric measures that describe the amount of radiation such as light or radiant heat that passes through or is emitted from a particular area, and falls within a given solid angle in a specified direction. They are used to characterize both emission from...
properties for twelve metals, titled On Certain Physical Properties of Light Produced by the Combustion of Different Metals in an Electric Spark Refracted by a Prism.
Dr. Alter began studying the optical properties of matter ever since finding a piece of melted, prism
Prism (optics)
In optics, a prism is a transparent optical element with flat, polished surfaces that refract light. The exact angles between the surfaces depend on the application. The traditional geometrical shape is that of a triangular prism with a triangular base and rectangular sides, and in colloquial use...
atic glass in the debris of the great Pittsburgh fire of 1845. By 1855, Alter published another article that expanded his original theory by including six gases, including the first discovery of what came to be named the Balmer lines of hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
. Alter's article contains a paragraph where he visualized the application of spectrum analysis to astronomy
Astronomy
Astronomy is a natural science that deals with the study of celestial objects and phenomena that originate outside the atmosphere of Earth...
, mentioning the study and detection of elements in the combustion of shooting stars or luminous meteors, and daguerreotyped
Daguerreotype
The daguerreotype was the first commercially successful photographic process. The image is a direct positive made in the camera on a silvered copper plate....
the dark lines of the solar spectrum. Alter's spectral discoveries were noted in various scientific publications in France, Germany, and Switzerland from 1854 to 1860.
In 1853, the Swedish
Sweden
Sweden , officially the Kingdom of Sweden , is a Nordic country on the Scandinavian Peninsula in Northern Europe. Sweden borders with Norway and Finland and is connected to Denmark by a bridge-tunnel across the Öresund....
physisist Anders Jonas Ångström
Anders Jonas Ångström
Anders Jonas Ångström was a Swedish physicist and one of the founders of the science of spectroscopy.-Biography:...
presented similar theories about gases having spectra in his work: Optiska Undersökningar to the Royal Swedish Academy of Sciences
Royal Swedish Academy of Sciences
The Royal Swedish Academy of Sciences or Kungliga Vetenskapsakademien is one of the Royal Academies of Sweden. The Academy is an independent, non-governmental scientific organization which acts to promote the sciences, primarily the natural sciences and mathematics.The Academy was founded on 2...
—pointing out that the electric spark yields two superposed spectra. Ångström postulated that an incandescent gas emits luminous rays of the same refrangibility as those it can absorb—a fundamental principle of spectrum analysis.
In 1860, German physicist Gustav Kirchhoff
Gustav Kirchhoff
Gustav Robert Kirchhoff was a German physicist who contributed to the fundamental understanding of electrical circuits, spectroscopy, and the emission of black-body radiation by heated objects...
and chemist Robert Bunsen
Robert Bunsen
Robert Wilhelm Eberhard Bunsen was a German chemist. He investigated emission spectra of heated elements, and discovered caesium and rubidium with Gustav Kirchhoff. Bunsen developed several gas-analytical methods, was a pioneer in photochemistry, and did early work in the field of organoarsenic...
published their own findings on the spectra of eight metals and identified these metals in natural elements. Kirchoff went on to contribute fundamental research on the nature of spectral absorption
Absorption (electromagnetic radiation)
In physics, absorption of electromagnetic radiation is the way by which the energy of a photon is taken up by matter, typically the electrons of an atom. Thus, the electromagnetic energy is transformed to other forms of energy for example, to heat. The absorption of light during wave propagation is...
. Spectrum analysis was then grouped by Kirchhoff into the three fundamental laws commonly called Kirchoff's Laws, these laws integrated both Alter and Ångström's discoveries of radiance
Radiance
Radiance and spectral radiance are radiometric measures that describe the amount of radiation such as light or radiant heat that passes through or is emitted from a particular area, and falls within a given solid angle in a specified direction. They are used to characterize both emission from...
and emission with Kirchhoff's fundamental discoveries of absorption.
Johann Balmer discovered in 1885 that the four visible lines of hydrogen were part of a series
Balmer series
The Balmer series or Balmer lines in atomic physics, is the designation of one of a set of six different named series describing the spectral line emissions of the hydrogen atom....
that could be expressed in terms of integers. This was followed a few years later by the Rydberg formula
Rydberg formula
The Rydberg formula is used in atomic physics to describe the wavelengths of spectral lines of many chemical elements. It was formulated by the Swedish physicist Johannes Rydberg, and presented on November 5, 1888.-History:...
, which described additional series of lines.
In the 1860's William Huggins
William Huggins
Sir William Huggins, OM, KCB, FRS was an English amateur astronomer best known for his pioneering work in astronomical spectroscopy.-Biography:...
and his wife Margaret used spectroscopy to determine that the stars were composed of the same elements as found on earth. They also used the non-relativistic Doppler shift
Doppler effect
The Doppler effect , named after Austrian physicist Christian Doppler who proposed it in 1842 in Prague, is the change in frequency of a wave for an observer moving relative to the source of the wave. It is commonly heard when a vehicle sounding a siren or horn approaches, passes, and recedes from...
(redshift
Redshift
In physics , redshift happens when light seen coming from an object is proportionally increased in wavelength, or shifted to the red end of the spectrum...
) equation on the spectrum of the star Sirius
Sirius
Sirius is the brightest star in the night sky. With a visual apparent magnitude of −1.46, it is almost twice as bright as Canopus, the next brightest star. The name "Sirius" is derived from the Ancient Greek: Seirios . The star has the Bayer designation Alpha Canis Majoris...
in 1868 to determine its axial speed. They were the first to take a spectrum of a planetary nebula when the Cat's Eye Nebula
Cat's Eye Nebula
The Cat's Eye Nebula is a planetary nebula in the constellation of Draco. Structurally, it is one of the most complex nebulae known, with high-resolution Hubble Space Telescope observations revealing remarkable structures such as knots, jets, bubbles and sinewy arc-like features...
(NGC 6543) was analyzed. Using spectral techniques, they were able to distinguish nebulae
Nebula
A nebula is an interstellar cloud of dust, hydrogen gas, helium gas and other ionized gases...
from galaxies
Galaxy
A galaxy is a massive, gravitationally bound system that consists of stars and stellar remnants, an interstellar medium of gas and dust, and an important but poorly understood component tentatively dubbed dark matter. The word galaxy is derived from the Greek galaxias , literally "milky", a...
.
In the early twentieth century, spectrum analysis led to "atomic spectroscopy
Atomic spectroscopy
Atomic spectroscopy is the determination of elemental composition by its electromagnetic or mass spectrum. Atomic spectroscopy is closely related to other forms of spectroscopy. It can be divided by atomization source or by the type of spectroscopy used. In the latter case, the main division is...
" and quantum mechanics
Quantum mechanics
Quantum mechanics, also known as quantum physics or quantum theory, is a branch of physics providing a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter. It departs from classical mechanics primarily at the atomic and subatomic...
.
See also
- Alpha-particle spectroscopyAlpha-particle spectroscopyOne method for testing for many alpha emitters is to use alpha-particle spectroscopy. For methods for gamma rays and beta particles, please see gamma spectroscopy and liquid scintillation counting respectively....
- Electromagnetic spectrumElectromagnetic spectrumThe electromagnetic spectrum is the range of all possible frequencies of electromagnetic radiation. The "electromagnetic spectrum" of an object is the characteristic distribution of electromagnetic radiation emitted or absorbed by that particular object....
- Fast Fourier transformFast Fourier transformA fast Fourier transform is an efficient algorithm to compute the discrete Fourier transform and its inverse. "The FFT has been called the most important numerical algorithm of our lifetime ." There are many distinct FFT algorithms involving a wide range of mathematics, from simple...
- Fraunhofer LinesFraunhofer linesIn physics and optics, the Fraunhofer lines are a set of spectral lines named for the German physicist Joseph von Fraunhofer . The lines were originally observed as dark features in the optical spectrum of the Sun....
- Mass spectrometryMass spectrometryMass spectrometry is an analytical technique that measures the mass-to-charge ratio of charged particles.It is used for determining masses of particles, for determining the elemental composition of a sample or molecule, and for elucidating the chemical structures of molecules, such as peptides and...
- Optical spectrum
- SpectroscopySpectroscopySpectroscopy is the study of the interaction between matter and radiated energy. Historically, spectroscopy originated through the study of visible light dispersed according to its wavelength, e.g., by a prism. Later the concept was expanded greatly to comprise any interaction with radiative...
- SpectrumSpectrumA spectrum is a condition that is not limited to a specific set of values but can vary infinitely within a continuum. The word saw its first scientific use within the field of optics to describe the rainbow of colors in visible light when separated using a prism; it has since been applied by...
- Quantum mechanicsQuantum mechanicsQuantum mechanics, also known as quantum physics or quantum theory, is a branch of physics providing a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter. It departs from classical mechanics primarily at the atomic and subatomic...

