
Thiele tube
Encyclopedia
Johannes Thiele (chemist)
Friedrich Karl Johannes Thiele was a German chemist and a prominent professor at several universities, including those in Munich and Strasbourg. He developed many laboratory techniques related to isolation of organic compounds...
, is a laboratory glassware
Laboratory glassware
Laboratory glassware refers to a variety of equipment, traditionally made of glass, used for scientific experiments and other work in science, especially in chemistry and biology laboratories...
designed to contain and heat
Heat
In physics and thermodynamics, heat is energy transferred from one body, region, or thermodynamic system to another due to thermal contact or thermal radiation when the systems are at different temperatures. It is often described as one of the fundamental processes of energy transfer between...
an oil bath
Oil bath
An oil bath is a laboratory heating device which uses boiling oil as the temperature regulator. Since different oils have different boiling points it is possible to obtain a temperature near to the desired temperature by selecting an oil with a boiling point as close as possible to the desired...
. Such a setup is commonly used in the determination of the melting point
Melting point
The melting point of a solid is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depends on pressure and is usually specified at standard atmospheric pressure...
of a substance
Chemical substance
In chemistry, a chemical substance is a form of matter that has constant chemical composition and characteristic properties. It cannot be separated into components by physical separation methods, i.e. without breaking chemical bonds. They can be solids, liquids or gases.Chemical substances are...
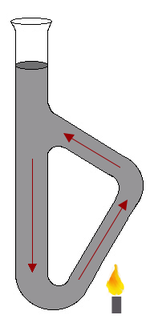
. The apparatus itself resembles a glass test tube
Test tube
A test tube, also known as a culture tube or sample tube, is a common piece of laboratory glassware consisting of a finger-like length of glass or clear plastic tubing, open at the top, usually with a rounded U-shaped bottom....
with an attached handle.
Operation
Oil is poured into the tube, and then the "handle" is heated, either by a small flame or some other heating element.The shape of the Thiele tube allows for formation of convectionConvection
Convection is the movement of molecules within fluids and rheids. It cannot take place in solids, since neither bulk current flows nor significant diffusion can take place in solids....
currents in the oil when it is heated. These currents maintain a fairly uniform temperature distribution throughout the oil in the tube. The side arm of the tube is designed to generate these convection currents and thus transfer the heat from the flame evenly and rapidly throughout the heating oil. The sample, packed in a capillary tube, is attached to the thermometer, and held by means of a rubber band or a small slice of rubber tubing.The Thiele tube is usually heated using a microburner with a small flame.
Melting point determination
A sample in a sealed capillary, attached to a thermometerThermometer
Developed during the 16th and 17th centuries, a thermometer is a device that measures temperature or temperature gradient using a variety of different principles. A thermometer has two important elements: the temperature sensor Developed during the 16th and 17th centuries, a thermometer (from the...
with a rubber band, is immersed in the tube. Heating is commenced, and the temperature ranges at which the sample melts can be then be observed. During heating, the point at which melting is observed and the temperature constant is the melting point of the sample. A more modern method utilizes dedicated equipment, known as a melting point apparatus
Melting point apparatus
A melting point apparatus is a scientific instrument used to determine the melting point of a substance. Some types of melting point apparatuses include the Thiele tube, Fisher-Johns apparatus, Gallenkamp melting point apparatus and automatic melting point apparatus.- Design :While the outward...
. A slow heating rate at the melting point is needed in order to get an accurate measurement. Record the temperature on the thermometer when the sample starts to melt and record the temperature again when all of the sample has melted (this gives you the melting point range). The Thiele tube in itself has many applications such as analyzing and synthesizing organic compounds such as aspirin
Aspirin
Aspirin , also known as acetylsalicylic acid , is a salicylate drug, often used as an analgesic to relieve minor aches and pains, as an antipyretic to reduce fever, and as an anti-inflammatory medication. It was discovered by Arthur Eichengrun, a chemist with the German company Bayer...
.

