
Dinitrogen complex
Encyclopedia
2n2-3d-balls.png)
Ligand
In coordination chemistry, a ligand is an ion or molecule that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from...
. In the area of coordination chemistry, the atomic and diatomic forms of nitrogen are distinguished, although otherwise "nitrogen" refers to N2.
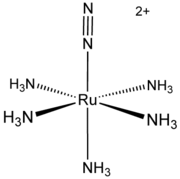
Metal complexes of N2 have been studied since 1965 when the first complex was reported by Allen and Senoff. This complex, [Ru(NH3)5(N2)]2+consists of a [Ru(NH3)5]2+ centre attached to one end of N2. Interest in such complexes arises because N2 comprises the majority of the atmosphere and because many useful compounds contain nitrogen atoms. Biological nitrogen fixation
Nitrogen fixation
Nitrogen fixation is the natural process, either biological or abiotic, by which nitrogen in the atmosphere is converted into ammonia . This process is essential for life because fixed nitrogen is required to biosynthesize the basic building blocks of life, e.g., nucleotides for DNA and RNA and...
probably occurs via the binding of N2 to a metal center in the enzyme nitrogenase
Nitrogenase
Nitrogenases are enzymes used by some organisms to fix atmospheric nitrogen gas . It is the only known family of enzymes that accomplish this process. Dinitrogen is quite inert because of the strength of its N-N triple bond...
, followed by a series of steps that involve electron transfer
Electron transfer
Electron transfer is the process by which an electron moves from an atom or a chemical species to another atom or chemical species...
and protonation
Protonation
In chemistry, protonation is the addition of a proton to an atom, molecule, or ion. Some classic examples include*the protonation of water by sulfuric acid:*the protonation of isobutene in the formation of a carbocation:2C=CH2 + HBF4 → 3C+ + BF4−*the protonation of ammonia in the...
. The hydrogenation of N2 is only weakly exothermic, hence the industrial hydrogenation of nitrogen via the Haber-Bosch Process employs high pressures and high temperatures.
Bonding modes
In terms of its bonding to transition metals, N2 is related to COCarbon monoxide
Carbon monoxide , also called carbonous oxide, is a colorless, odorless, and tasteless gas that is slightly lighter than air. It is highly toxic to humans and animals in higher quantities, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal...
and acetylene
Acetylene
Acetylene is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in pure form and thus is usually handled as a solution.As an alkyne, acetylene is unsaturated because...
as all three species have triple bond
Triple bond
A triple bond in chemistry is a chemical bond between two chemical elements involving six bonding electrons instead of the usual two in a covalent single bond. The most common triple bond, that between two carbon atoms, can be found in alkynes. Other functional groups containing a triple bond are...
s. A variety of bonding modes have been characterized.
End-on
As a ligand, N2 usually binds to metals as an "end-on" ligand, as illustrated by Allen and Senoff's complex. Such complexes are usually analgous to related CO derivatives. A good example of this relationship are the complexes IrCl(CO)(PPh3)2Vaska's complex
Vaska's complex is the trivial name for the chemical compound trans-chlorocarbonylbisiridium, which has the formula IrCl[P3]2. This square planar diamagnetic organometallic complex consists of a central iridium atom bound to two mutually trans triphenylphosphine ligands, carbon monoxide, and a...
and IrCl(N2)(PPh3)2.
Few complexes contain more than one N2 ligand, and no example features three (in contrast metal hexacarbonyls are common). The dinitrogen ligand in W(N2)2(Ph2CH2CH2PPh2)2 can be reduced to produce ammonia.
Bridging, end-on
N2 also serves as a bridging ligand, as illustrated by {[Ru(NH3)5]2(μ-N2)}4+.A study in 2006 of iron-dinitrogen complexes showed that the N–N bond is significantly weakened upon complexation with iron atoms with a low coordination number. The complex involved bidentate chelating ligands attached to the iron atoms in the Fe–N–N–Fe core, in which acts as a bridging ligand between the iron atoms. Increasing the coordination number of iron by modifying the chelating ligands and adding an additional ligand per iron atom showed an increase in the strength of the N–N bond in the resulting complex. It is thus suspected that Fe in a low-coordination environment is a key factor to the fixation of nitrogen by the nitrogenase
Nitrogenase
Nitrogenases are enzymes used by some organisms to fix atmospheric nitrogen gas . It is the only known family of enzymes that accomplish this process. Dinitrogen is quite inert because of the strength of its N-N triple bond...
enzyme, since its Fe–Mo cofactor also features Fe with low coordination numbers.
Side-on, bridging
In a second mode of bridging, bimetallic complexes are known wherein the N-N vector is perpendicular to the M-M vector. One example is [(η5-C5Me4H)2Zr]2(μBridging ligand
A bridging ligand is a ligand that connects two or more atoms, usually metal ions. The ligand may be atomic or polyatomic. Virtually all complex organic compounds can serve as bridging ligands, so the term is usually restricted to small ligands such as pseudohalides or to ligands that are...
2,η
Hapticity
The term hapticity is used to describe how a group of contiguous atoms of a ligand are coordinated to a central atom. Hapticity of a ligand is indicated by the Greek character 'eta', η. A superscripted number following the η denotes the number of contiguous atoms of the ligand that are bound to...
2,η2-N2).

