Grain boundary strengthening
Encyclopedia
Grain-boundary strengthening (or Hall–Petch strengthening) is a method of strengthening
materials by changing their average crystallite
(grain) size. It is based on the observation that grain boundaries impede dislocation movement and that the number of dislocation
s within a grain have an effect on how easily dislocations can traverse grain boundaries and travel from grain to grain. So, by changing grain size one can influence dislocation movement and yield strength. For example, heat treatment
after plastic deformation and changing the rate of solidification are ways to alter grain size.
act as pinning points
impeding further dislocation propagation. Since the lattice structure of adjacent grains differs in orientation, it requires more energy for a dislocation to change directions and move into the adjacent grain. The grain boundary is also much more disordered than inside the grain, which also prevents the dislocations from moving in a continuous slip plane. Impeding this dislocation movement will hinder the onset of plasticity and hence increase the yield strength of the material.
Under an applied stress, existing dislocations and dislocations generated by Frank–Read Sources will move through a crystalline lattice until encountering a grain boundary, where the large atomic mismatch between different grains creates a repulsive stress field to oppose continued dislocation motion. As more dislocations propagate to this boundary, dislocation 'pile up' occurs as a cluster of dislocations are unable to move past the boundary. As dislocations generate repulsive stress fields, each successive dislocation will apply a repulsive force to the dislocation incident with the grain boundary. These repulsive forces act as a driving force to reduce the energetic barrier for diffusion across the boundary, such that additional pile up causes dislocation diffusion across the grain boundary, allowing further deformation in the material. Decreasing grain size decreases the amount of possible pile up at the boundary, increasing the amount of applied stress necessary to move a dislocation across a grain boundary. The higher the applied stress to move the dislocation, the higher the yield strength. Thus, there is then an inverse relationship between grain size and yield strength, as demonstrated by the Hall–Petch equation. However, when there is a large direction change in the orientation of the two adjacent grains, the dislocation may not necessarily move from one grain to the other but instead create a new source of dislocation in the adjacent grain. The theory remains the same that more grain boundaries create more opposition to dislocation movement and in turn strengthens the material.
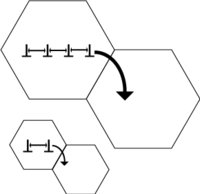
Obviously, there is a limit to this mode of strengthening, as infinitely strong materials do not exist. Grain sizes can range from about 100 um (large grains) to 1 um (small grains). Lower than this, the size of dislocations begins to approach the size of the grains. At a grain size of about 10 nm (1E-08 m), only one or two dislocations can fit inside of a grain (see Figure 1 above). This scheme prohibits dislocation pile-up and never results in grain boundary diffusion. The lattice resolves the applied stress by grain boundary sliding, resulting in a decrease in the material's yield strength.
To understand the mechanism of grain boundary strengthening one must understand the nature of dislocation-dislocation interactions. Dislocations create a stress field around them given by:
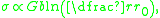
where G is the material's shear modulus, and b is the Burgers vector
. If the dislocations are in the right alignment with respect to each other, the local stress fields they create will repel each other. This helps dislocation movement along grains and across grain boundaries. Hence, the more dislocations are present in a grain, the greater the stress field felt by a dislocation near a grain boundary:
There is an inverse relationship between delta yield strength and grain size to some power, x.
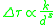
where k is the strengthening coefficient and both k and x are material specific. The smaller the grain size, the smaller the repulsion stress felt by a grain boundary dislocation and the higher the applied stress needed to propagate dislocations through the material.
The relation between yield stress and grain size is described mathematically by the Hall–Petch equation:
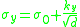
where σy is the yield stress, σo is a materials constant for the starting stress for dislocation movement (or the resistance of the lattice to dislocation motion), ky is the strengthening coefficient (a constant unique to each material), and d is the average grain diameter.
Theoretically, a material could be made infinitely strong if the grains are made infinitely small. This is impossible though, because the lower limit of grain size is a single unit cell of the material. Even then, if the grains of a material are the size of a single unit cell, then the material is in fact amorphous, not crystalline, since there is no long range order, and dislocations can not be defined in an amorphous material. It has been observed experimentally that the microstructure with the highest yield strength is a grain size of about 10 nm (1E-08 m), because grains smaller than this undergo another yielding mechanism, grain boundary sliding. Producing engineering materials with this ideal grain size is difficult because only thin films can be reliably produced with grains of this size.
In 1951, while at the University of Sheffield, E. O. Hall wrote three papers which appeared in volume 64 of the Proceedings of the Physical Society
. In his third paper, Hall showed that the length of slip bands or crack lengths correspond to grain sizes and thus a relationship could be established between the two. Hall concentrated on the yielding properties of mild steels.
Based on his experimental work carried out in 1946–1949, N. J. Petch of the University of Leeds
, England
published a paper in 1953 independent from Hall's. Petch's paper concentrated more on brittle fracture. By measuring the variation in cleavage strength with respect to ferritic
grain size at very low temperatures, Petch found a relationship exact to that of Hall's. Thus this important relationship is named after both Hall and Petch.
Other explanations that have been proposed to rationalize the apparent softening of metals with nanosized grains include poor sample quality and the suppression of dislocation pileups.
Many of the early measurements of a reverse Hall–Petch effect were likely the result of unrecognized pores in samples. The presence of voids in nanocrystalline metals would undoubtedly lead to their having weaker mechanical properties.
The pileup of dislocations at grain boundaries is a hallmark mechanism of the Hall–Petch relationship. Once grain sizes drop below the equilibrium distance between dislocations, though, this relationship should no longer be valid. Nevertheless, it is not entirely clear what exactly the dependency of yield stress should be on grain sizes below this point.
. The specific techniques and corresponding mechanisms will vary based on what materials are being considered.
One method for controlling grain size in aluminum alloys is by introducing particles to serve as nucleants, such as Al–5%Ti. Grains will grow via heterogeneous nucleation; that is, for a given degree of undercooling beneath the melting temperature, aluminum particles in the melt will nucleate on the surface of the added particles. Grains will grow in the form of dendrites
growing radially away from the surface of the nucleant. Solute particles can then be added (called grain refiners) which limit the growth of dendrites, leading to grain refinement. TiB2 is a common grain refiner for Al alloys; however, novel refiners such as Al3Sc have been suggested.
One common technique is to induce a very small fraction of the melt to solidify at a much higher temperature than the rest; this will generate seed crystal
s that act as a template when the rest of the material falls to its (lower) melting temperature
and begins to solidify. Since a huge number of minuscule seed crystals are present, a nearly equal number of crystallites result, and the size of any one grain is limited.
Strength of materials
In materials science, the strength of a material is its ability to withstand an applied stress without failure. The applied stress may be tensile, compressive, or shear. Strength of materials is a subject which deals with loads, deformations and the forces acting on a material. A load applied to a...
materials by changing their average crystallite
Crystallite
Crystallites are small, often microscopic crystals that, held together through highly defective boundaries, constitute a polycrystalline solid. Metallurgists often refer to crystallites as grains.- Details :...
(grain) size. It is based on the observation that grain boundaries impede dislocation movement and that the number of dislocation
Dislocation
In materials science, a dislocation is a crystallographic defect, or irregularity, within a crystal structure. The presence of dislocations strongly influences many of the properties of materials...
s within a grain have an effect on how easily dislocations can traverse grain boundaries and travel from grain to grain. So, by changing grain size one can influence dislocation movement and yield strength. For example, heat treatment
Heat treatment
Heat treating is a group of industrial and metalworking processes used to alter the physical, and sometimes chemical, properties of a material. The most common application is metallurgical. Heat treatments are also used in the manufacture of many other materials, such as glass...
after plastic deformation and changing the rate of solidification are ways to alter grain size.
Theory
In grain-boundary strengthening the grain boundariesGrain boundary
A grain boundary is the interface between two grains, or crystallites, in a polycrystalline material. Grain boundaries are defects in the crystal structure, and tend to decrease the electrical and thermal conductivity of the material...
act as pinning points
Pinning points
In a crystalline material, a dislocation is capable of traveling throughout the lattice when relatively small stresses are applied. This movement of dislocations results in the material plastically deforming. Pinning points in the material act to halt a dislocation's movement, requiring a greater...
impeding further dislocation propagation. Since the lattice structure of adjacent grains differs in orientation, it requires more energy for a dislocation to change directions and move into the adjacent grain. The grain boundary is also much more disordered than inside the grain, which also prevents the dislocations from moving in a continuous slip plane. Impeding this dislocation movement will hinder the onset of plasticity and hence increase the yield strength of the material.
Under an applied stress, existing dislocations and dislocations generated by Frank–Read Sources will move through a crystalline lattice until encountering a grain boundary, where the large atomic mismatch between different grains creates a repulsive stress field to oppose continued dislocation motion. As more dislocations propagate to this boundary, dislocation 'pile up' occurs as a cluster of dislocations are unable to move past the boundary. As dislocations generate repulsive stress fields, each successive dislocation will apply a repulsive force to the dislocation incident with the grain boundary. These repulsive forces act as a driving force to reduce the energetic barrier for diffusion across the boundary, such that additional pile up causes dislocation diffusion across the grain boundary, allowing further deformation in the material. Decreasing grain size decreases the amount of possible pile up at the boundary, increasing the amount of applied stress necessary to move a dislocation across a grain boundary. The higher the applied stress to move the dislocation, the higher the yield strength. Thus, there is then an inverse relationship between grain size and yield strength, as demonstrated by the Hall–Petch equation. However, when there is a large direction change in the orientation of the two adjacent grains, the dislocation may not necessarily move from one grain to the other but instead create a new source of dislocation in the adjacent grain. The theory remains the same that more grain boundaries create more opposition to dislocation movement and in turn strengthens the material.
Obviously, there is a limit to this mode of strengthening, as infinitely strong materials do not exist. Grain sizes can range from about 100 um (large grains) to 1 um (small grains). Lower than this, the size of dislocations begins to approach the size of the grains. At a grain size of about 10 nm (1E-08 m), only one or two dislocations can fit inside of a grain (see Figure 1 above). This scheme prohibits dislocation pile-up and never results in grain boundary diffusion. The lattice resolves the applied stress by grain boundary sliding, resulting in a decrease in the material's yield strength.
To understand the mechanism of grain boundary strengthening one must understand the nature of dislocation-dislocation interactions. Dislocations create a stress field around them given by:
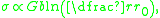
where G is the material's shear modulus, and b is the Burgers vector
Burgers vector
The Burgers vector, named after Dutch physicist Jan Burgers, is a vector, often denoted b, that represents the magnitude and direction of the lattice distortion of dislocation in a crystal lattice....
. If the dislocations are in the right alignment with respect to each other, the local stress fields they create will repel each other. This helps dislocation movement along grains and across grain boundaries. Hence, the more dislocations are present in a grain, the greater the stress field felt by a dislocation near a grain boundary:

Subgrain strengthening
A subgrain is a part of the grain that is only slightly disoriented from other parts of the grain. Current research is being done to see the effect of subgrain strengthening in materials. Depending on the processing of the material, subgrains can form within the grains of the material. For example, when Fe-based material is ball-milled for long periods of time (e.g. 100+ hours), subgrains of 60–90 nm are formed. It has been shown that the higher the density of the subgrains, the higher the yield stress of the material due to the increased subgrain boundary. The strength of the metal was found to vary reciprocally with the size of the subgrain, which is analogous to the Hall–Petch equation. The subgrain boundary strengthening also has a breakdown point of around a subgrain size of 0.1 nm, which is the size where any subgrains smaller than that size would decrease yield strength. http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6TXD-4MRFCBJ-7&_user=501045&_coverDate=08%2F15%2F2007&_alid=654367194&_rdoc=3&_fmt=full&_orig=search&_cdi=5588&_sort=d&_docanchor=&view=c&_ct=86&_acct=C000022659&_version=1&_urlVersion=0&_userid=501045&md5=66df0d7de746d813ad753d4b2e261581.Hall–Petch relationship
| Material | σo [MPa] | k [MPa m1/2] |
|---|---|---|
| Copper | 25 | 0.11 |
| Titanium | 80 | 0.40 |
| Mild steel | 70 | 0.74 |
| Ni3Al | 300 | 1.70 |
There is an inverse relationship between delta yield strength and grain size to some power, x.
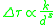
where k is the strengthening coefficient and both k and x are material specific. The smaller the grain size, the smaller the repulsion stress felt by a grain boundary dislocation and the higher the applied stress needed to propagate dislocations through the material.
The relation between yield stress and grain size is described mathematically by the Hall–Petch equation:
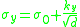
where σy is the yield stress, σo is a materials constant for the starting stress for dislocation movement (or the resistance of the lattice to dislocation motion), ky is the strengthening coefficient (a constant unique to each material), and d is the average grain diameter.
Theoretically, a material could be made infinitely strong if the grains are made infinitely small. This is impossible though, because the lower limit of grain size is a single unit cell of the material. Even then, if the grains of a material are the size of a single unit cell, then the material is in fact amorphous, not crystalline, since there is no long range order, and dislocations can not be defined in an amorphous material. It has been observed experimentally that the microstructure with the highest yield strength is a grain size of about 10 nm (1E-08 m), because grains smaller than this undergo another yielding mechanism, grain boundary sliding. Producing engineering materials with this ideal grain size is difficult because only thin films can be reliably produced with grains of this size.
History
In the early 1950s two groundbreaking series of papers were written independently on the relationship between grain boundaries and strength.In 1951, while at the University of Sheffield, E. O. Hall wrote three papers which appeared in volume 64 of the Proceedings of the Physical Society
Proceedings of the Physical Society
The Proceedings of the Physical Society is a journal on the subject of physics, originally associated with the Physical Society of London, England. - Journal history :* 1874–1925: Proceedings of the Physical Society of London...
. In his third paper, Hall showed that the length of slip bands or crack lengths correspond to grain sizes and thus a relationship could be established between the two. Hall concentrated on the yielding properties of mild steels.
Based on his experimental work carried out in 1946–1949, N. J. Petch of the University of Leeds
University of Leeds
The University of Leeds is a British Redbrick university located in the city of Leeds, West Yorkshire, England...
, England
England
England is a country that is part of the United Kingdom. It shares land borders with Scotland to the north and Wales to the west; the Irish Sea is to the north west, the Celtic Sea to the south west, with the North Sea to the east and the English Channel to the south separating it from continental...
published a paper in 1953 independent from Hall's. Petch's paper concentrated more on brittle fracture. By measuring the variation in cleavage strength with respect to ferritic
Ferrite (iron)
Ferrite or alpha iron is a materials science term for iron, or a solid solution with iron as the main constituent, with a body centred cubic crystal structure. It is the component which gives steel and cast iron their magnetic properties, and is the classic example of a ferromagnetic material...
grain size at very low temperatures, Petch found a relationship exact to that of Hall's. Thus this important relationship is named after both Hall and Petch.
Reverse or inverse Hall–Petch relation
The Hall–Petch relation predicts that as the grain size decreases the yield strength increases. The Hall–Petch relation was experimentally found to be an effective model for materials with grain sizes ranging from 1 millimeter to 1 micrometre. Consequently it was believed that if average grain size could be decreased even further to the nanometer length scale the yield strength would increase as well. However, experiments on many nanocrystalline materials demonstrated that if the grains reached a small enough size, the critical grain size which is typically less than 100 nm (1E-07 m), the yield strength would either remain constant or decrease with decreasing grains size. This phenomenon has been termed the reverse or inverse Hall–Petch relation. A number of different mechanisms have been proposed for this relation. As suggested by Carlton et al. they fall into four categories: (1) Dislocation based (2) Diffusion based (3) Grain boundary shearing based (4) Two phase based.Other explanations that have been proposed to rationalize the apparent softening of metals with nanosized grains include poor sample quality and the suppression of dislocation pileups.
Many of the early measurements of a reverse Hall–Petch effect were likely the result of unrecognized pores in samples. The presence of voids in nanocrystalline metals would undoubtedly lead to their having weaker mechanical properties.
The pileup of dislocations at grain boundaries is a hallmark mechanism of the Hall–Petch relationship. Once grain sizes drop below the equilibrium distance between dislocations, though, this relationship should no longer be valid. Nevertheless, it is not entirely clear what exactly the dependency of yield stress should be on grain sizes below this point.
Grain refinement
Grain refinement, also known as inoculation, is the set of techniques used to implement grain boundary strengthening in metallurgyMetallurgy
Metallurgy is a domain of materials science that studies the physical and chemical behavior of metallic elements, their intermetallic compounds, and their mixtures, which are called alloys. It is also the technology of metals: the way in which science is applied to their practical use...
. The specific techniques and corresponding mechanisms will vary based on what materials are being considered.
One method for controlling grain size in aluminum alloys is by introducing particles to serve as nucleants, such as Al–5%Ti. Grains will grow via heterogeneous nucleation; that is, for a given degree of undercooling beneath the melting temperature, aluminum particles in the melt will nucleate on the surface of the added particles. Grains will grow in the form of dendrites
Dendrite (crystal)
A crystal dendrite is a crystal that develops with a typical multi-branching tree-like form. Dendritic crystal growth is very common and illustrated by snowflake formation and frost patterns on a window. Dendritic crystallization forms a natural fractal pattern...
growing radially away from the surface of the nucleant. Solute particles can then be added (called grain refiners) which limit the growth of dendrites, leading to grain refinement. TiB2 is a common grain refiner for Al alloys; however, novel refiners such as Al3Sc have been suggested.
One common technique is to induce a very small fraction of the melt to solidify at a much higher temperature than the rest; this will generate seed crystal
Seed crystal
A seed crystal is a small piece of single crystal/polycrystal material from which a large crystal of the same material typically is to be grown...
s that act as a template when the rest of the material falls to its (lower) melting temperature
Melting temperature
Melting temperature may refer to:* Melting point, the temperature at which a substance changes from solid to liquid state.* DNA melting temperature, the temperature at which a DNA double helix dissociates into single strands....
and begins to solidify. Since a huge number of minuscule seed crystals are present, a nearly equal number of crystallites result, and the size of any one grain is limited.
| Metal | Inoculant |
|---|---|
| Cast iron | FeSi, SiCa, graphite |
| Mg alloys | Zr, C |
| Cu alloys | Fe, Co, Zr |
| Al–Si alloys | P, Ti, B |
| Pb alloys | As, Te |
| Zn alloys | Ti |
| Ti alloys | – intermetallics |