
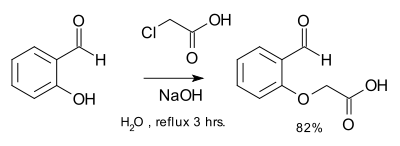
Williamson ether synthesis
Encyclopedia
Organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, photochemical reactions and redox reactions. In organic synthesis,...
, forming an ether
Ether
Ethers are a class of organic compounds that contain an ether group — an oxygen atom connected to two alkyl or aryl groups — of general formula R–O–R'. A typical example is the solvent and anesthetic diethyl ether, commonly referred to simply as "ether"...
from an organohalide and an alcohol
Alcohol
In chemistry, an alcohol is an organic compound in which the hydroxy functional group is bound to a carbon atom. In particular, this carbon center should be saturated, having single bonds to three other atoms....
. This reaction was developed by Alexander Williamson
Alexander William Williamson
Alexander William Williamson FRS was an English chemist of Scottish descent. He is best known today for the Williamson ether synthesis.-Biography:...
in 1850. Typically it involves the reaction of an alkoxide ion with a primary alkyl halide
Halide
A halide is a binary compound, of which one part is a halogen atom and the other part is an element or radical that is less electronegative than the halogen, to make a fluoride, chloride, bromide, iodide, or astatide compound. Many salts are halides...
via an SN2 reaction
SN2 reaction
The SN2 reaction is a type of nucleophilic substitution, where a lone pair from a nucleophile attacks an electron deficient electrophilic center and bonds to it, expelling another group called a leaving group. Thus the incoming group replaces the leaving group in one step...
. This reaction is important in the history of organic chemistry because it helped prove the structure of ether
Ether
Ethers are a class of organic compounds that contain an ether group — an oxygen atom connected to two alkyl or aryl groups — of general formula R–O–R'. A typical example is the solvent and anesthetic diethyl ether, commonly referred to simply as "ether"...
s.
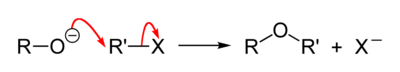
The general reaction mechanism is as follows:
Sodium ethoxide
Sodium ethoxide is an alkoxide salt with the chemical formula C2H5ONa.-Preparation:It is commercially available as a white solid, or as a solution in ethanol. It is easily prepared in the laboratory by reacting sodium metal with ethanol:...
with chloroethane to form diethyl ether
Diethyl ether
Diethyl ether, also known as ethyl ether, simply ether, or ethoxyethane, is an organic compound in the ether class with the formula . It is a colorless, highly volatile flammable liquid with a characteristic odor...
and sodium chloride:
- Na+C2H5O− + C2H5Cl C2H5OC2H5 + Na+Cl−
Scope
The Williamson reaction is of broad scope, is widely used in both laboratory and industrial synthesis, and remains the simplest and most popular method of preparing ethers. Both symmetrical and asymmetrical ethers are easily prepared. The intramolecular reaction of halohydrinHalohydrin
A halohydrin or a haloalcohol is a type of organic compound or functional group in which one carbon atom has a halogen substituent, and an adjacent carbon atom has a hydroxyl substituent. They are derived from alcohols are therefore characterized by the presence of both the hydroxyl functional...
s in particular, gives epoxide
Epoxide
An epoxide is a cyclic ether with three ring atoms. This ring approximately defines an equilateral triangle, which makes it highly strained. The strained ring makes epoxides more reactive than other ethers. Simple epoxides are named from the parent compound ethylene oxide or oxirane, such as in...
s.
In the case of asymmetrical ethers there are two possibilities for the choice of reactants, and one is usually preferable either on the basis of availability or reactivity. The Williamson reaction is also frequently used to prepare an ether indirectly from two alcohols. One of the alcohols is first converted to a leaving group (usually tosylate), then the two are reacted together.
The alkoxide (or aroxide) may be primary, secondary or tertiary. The alkylating agent, on the other hand is most preferably primary. Secondary alkylating agents also react, but tertiary ones are usually too prone to side reactions to be of practical use. The leaving group is most often a halide or a sulfonate ester synthesized for the purpose of the reaction. Since the conditions of the reaction are rather forcing, protecting group
Protecting group
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group in order to obtain chemoselectivity in a subsequent chemical reaction...
s are often used to pacify other parts of the reacting molecules (e.g. other alcohols, amine
Amine
Amines are organic compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group. Important amines include amino acids, biogenic amines,...
s, etc.)
Conditions
Since alkoxide ions are highly reactive, they are usually prepared immediately prior to the reaction, or are generated in situ. In laboratory chemistry, in situ generation is most often accomplished by the use of a carbonateCarbonate
In chemistry, a carbonate is a salt of carbonic acid, characterized by the presence of the carbonate ion, . The name may also mean an ester of carbonic acid, an organic compound containing the carbonate group C2....
base or potassium hydroxide
Potassium hydroxide
Potassium hydroxide is an inorganic compound with the formula KOH, commonly called caustic potash.Along with sodium hydroxide , this colorless solid is a prototypical strong base. It has many industrial and niche applications. Most applications exploit its reactivity toward acids and its corrosive...
, while in industrial syntheses phase transfer catalysis is very common. A wide range of solvents can be used, but protic solvents and apolar solvents tend to slow the reaction rate strongly, as a result of lowering the availability of the free nucleophile. For this reason, acetonitrile
Acetonitrile
Acetonitrile is the chemical compound with formula . This colourless liquid is the simplest organic nitrile. It is produced mainly as a byproduct of acrylonitrile manufacture...
and N,N-dimethylformamide are particularly commonly used.
A typical Williamson reaction is conducted at 50–100°C and is complete in 1–8 hours. Often the complete disappearance of the starting material is difficult to achieve, and side reactions are common. Yields of 50–95% are generally achieved in laboratory syntheses, while near-quantitative conversion can be achieved in industrial procedures.
Catalysis is not usually necessary in laboratory syntheses. However, if an unreactive alkylating agent is used (e.g. an alkyl chloride) then the rate of reaction can be greatly improved by the addition of a catalytic quantity of a soluble iodide salt (which undergoes halide exchange with the chloride to yield a much more reactive iodide, a variant of the Finkelstein reaction
Finkelstein reaction
The Finkelstein reaction, named for the German chemist Hans Finkelstein , is an SN2 reaction that involves the exchange of one halogen atom for another...
). In extreme cases, silver salts may be added for example silver oxide :
The silver ion coordinates with the halide leaving group to make its departure more facile. Finally, phase transfer catalysts are sometimes used (e.g. tetrabutylammonium bromide or 18-crown-6
18-Crown-6
18-Crown-6 is an organic compound with the formula [C2H4O]6 and the IUPAC name of 1,4,7,10,13,16-hexaoxacyclooctadecane. The compound is a crown ether. Crown ethers coordinate some metal cations in their central cavity; 18-crown-6 displays a particular affinity for potassium cations. The synthesis...
) in order to increase the solubility of the alkoxide by offering a softer counter-ion
Counterion
A counterion is the ion that accompanies an ionic species in order to maintain electric neutrality. In table salt the sodium cation is the counterion for the chlorine anion and vice versa.In a charged transition metal complex, a simple A counterion is the ion that accompanies an ionic species in...
.
Side reactions
The Williamson reaction often competes with the base-catalyzed elimination of the alkylating agent, and the nature of the leaving group as well as the reaction conditions (particularly the temperature and solvent) can have a strong effect on which is favored. In particular, some structures of alkylating agent can be particularly prone to elimination.When the nucleophile is an aroxide ion, the Williamson reaction can also compete with alkylation on the ring since the aroxide is an ambident nucleophile.


