
Aziridine
Encyclopedia
Aziridines are organic compound
s containing the aziridine functional group
, a three-membered heterocycle with one amine
group and two methylene
groups. The parent compound is aziridine (or ethylene imine), with molecular formula .
bond angle of 109.5°, which results in angle strain
as in the comparable cyclopropane
and oxirane molecules. A banana bond
model explains bonding in such compounds. Aziridine is less basic
than acyclic
aliphatic amines, with a pKa
of 7.9 for the conjugate acid
, due to increased s character of the nitrogen
free electron pair. Angle strain
in aziridine also increases the barrier to nitrogen inversion
. This barrier height permits the isolation of separate invertomers, for example the cis and trans invertomers of N-chloro-2-methylaziridine.
functional group displaces the adjacent halide
in an intramolecular
nucleophilic substitution
reaction to generate an aziridine. Amino alcohols have the same reactivity, but the hydroxy
group must first be converted into a good leaving group
. The cyclization of an amino alcohol is called a Wenker synthesis
(1935), and that of a haloamine the Gabriel ethylenimine method (1888)
addition to alkene
s is a well-established method for the synthesis of aziridines. Photolysis or thermolysis of azide
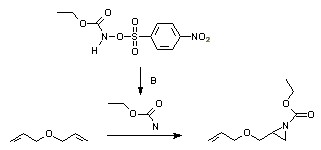
s are good ways to generate nitrenes. Nitrenes can also be prepared in situ from iodosobenzene diacetate and sulfonamide
s, or the ethoxycarbonylnitrene from the N-sulfonyloxy precursor.
, producing an aziridine. Triazolines can be generated by cycloaddition
of alkene
s with an azide
.
with sodium azide
, followed by organic reduction of the azide
with triphenylphosphine
accompanied by expulsion of nitrogen gas:
 The other method involves the ring-opening reaction of an epoxide
The other method involves the ring-opening reaction of an epoxide
with amine
s, followed by ring closing with the Mitsunobu reaction
.
s with Grignard reagents:

s due to their ring strain
. Alcoholysis and aminolysis are basically the reverse reactions of the cyclizations. Carbon nucleophiles such as organolithium reagent
s and organocuprates are also effective.
One application of a ring-opening reaction in asymmetric synthesis is that of trimethylsilylazide with an asymmetric ligand in scheme 2 in an organic synthesis
of oseltamivir
:
thermal or photochemical ring-opening reaction. These ylides can be trapped with a suitable dipolarophile in a 1,3-dipolar cycloaddition
.
When the N-substituent is an electron-withdrawing group such as a tosyl
group, the carbon-nitrogen bond
breaks, forming another zwitterion
This reaction type requires a Lewis acid
catalyst such as boron trifluoride
. In this way 2-phenyl-N-tosylaziridine reacts with alkynes, nitrile
s, ketone
s and alkene
s. Certain 1,4-dipoles form from azetidine
s.
s, aziridines are subject to attack and ring-opening by endogenous nucleophiles such as nitrogenous bases in DNA base pairs, resulting in potential mutagenicity.
Some reports note that the use of gloves has not prevented permeation of aziridine. It is therefore important that users check the breakthrough permeation times for gloves, and pay scrupulous attention to avoiding contamination when degloving.
(IARC) has reviewed aziridine compounds and classified them as possibly carcinogenic to humans (IARC Group 2B). In making the overall evaluation, the IARC Working Group took into consideration that aziridine is a direct-acting alkylating agent which is mutagenic in a wide range of test systems and forms DNA adducts that are promutagenic.
Irritancy
Aziridines are irritants of mucosal surfaces including eyes, nose, respiratory tract and skin.
Sensitization
Aziridine rapidly penetrates skin on contact.
Skin sensitizer — causing allergic contact dermatitis and urticaria
.
Respiratory sensitiser — causing occupational asthma
Organic compound
An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. For historical reasons discussed below, a few types of carbon-containing compounds such as carbides, carbonates, simple oxides of carbon, and cyanides, as well as the...
s containing the aziridine functional group
Functional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
, a three-membered heterocycle with one amine
Amine
Amines are organic compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group. Important amines include amino acids, biogenic amines,...
group and two methylene
Methylene
Methylene is a chemical species in which a carbon atom is bonded to two hydrogen atoms. Three different possibilities present themselves:* the -CH2- substituent group: e.g., dichloromethane ....
groups. The parent compound is aziridine (or ethylene imine), with molecular formula .
Structure
The bond angles in aziridine are approximately 60°, considerably less than the normal hydrocarbonHydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons from which one hydrogen atom has been removed are functional groups, called hydrocarbyls....
bond angle of 109.5°, which results in angle strain
Angle strain
Angle strain, also called Baeyer strain in cyclic molecules, is the resistance associated with bond angle compression or bond angle expansion. It occurs when bond angles deviate from the ideal bond angles to achieve maximum bond strength in a specific chemical conformation...
as in the comparable cyclopropane
Cyclopropane
Cyclopropane is a cycloalkane molecule with the molecular formula C3H6, consisting of three carbon atoms linked to each other to form a ring, with each carbon atom bearing two hydrogen atoms...
and oxirane molecules. A banana bond
Banana bond
Bent bond, also known as banana bond, is a term in organic chemistry that refers to a type of covalent chemical bond with a geometry somewhat reminiscent of a banana...
model explains bonding in such compounds. Aziridine is less basic
Base (chemistry)
For the term in genetics, see base A base in chemistry is a substance that can accept hydrogen ions or more generally, donate electron pairs. A soluble base is referred to as an alkali if it contains and releases hydroxide ions quantitatively...
than acyclic
Acyclic
Acyclic can refer to:* In chemistry, a compound which is not cyclic, e.g. alkanes and acyclic aliphatic compounds* In mathematics:** A graph without a cycle, especially*** A directed acyclic graph...
aliphatic amines, with a pKa
PKA
PKA, pKa, or other similar variations may stand for:* pKa, the symbol for the acid dissociation constant at logarithmic scale* Protein kinase A, a class of cAMP-dependent enzymes* Pi Kappa Alpha, the North-American social fraternity...
of 7.9 for the conjugate acid
Conjugate acid
Within the Brønsted–Lowry acid-base theory , a conjugate acid is the acid member, HX, of a pair of two compounds that transform into each other by gain or loss of a proton. A conjugate acid can also be seen as the chemical substance that releases, or donates, a proton in the forward chemical...
, due to increased s character of the nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
free electron pair. Angle strain
Angle strain
Angle strain, also called Baeyer strain in cyclic molecules, is the resistance associated with bond angle compression or bond angle expansion. It occurs when bond angles deviate from the ideal bond angles to achieve maximum bond strength in a specific chemical conformation...
in aziridine also increases the barrier to nitrogen inversion
Nitrogen inversion
In chemistry, a nitrogen compound like ammonia in a trigonal pyramid geometry undergoes rapid nitrogen inversion whereby the molecule turns inside out. This interconversion is a room temperature process because the energy barrier is relatively small. Contrast this to phosphine which does not show...
. This barrier height permits the isolation of separate invertomers, for example the cis and trans invertomers of N-chloro-2-methylaziridine.
Cyclization of haloamines and amino alcohols
An amineAmine
Amines are organic compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group. Important amines include amino acids, biogenic amines,...
functional group displaces the adjacent halide
Halide
A halide is a binary compound, of which one part is a halogen atom and the other part is an element or radical that is less electronegative than the halogen, to make a fluoride, chloride, bromide, iodide, or astatide compound. Many salts are halides...
in an intramolecular
Intramolecular
Intramolecular in chemistry describes a process or characteristic limited within the structure of a single molecule, a property or phenomenon limited to the extent of a single molecule.- Examples :...
nucleophilic substitution
Nucleophilic substitution
In organic and inorganic chemistry, nucleophilic substitution is a fundamental class of reactions in which an electron nucleophile selectively bonds with or attacks the positive or partially positive charge of an atom or a group of atoms called the leaving group; the positive or partially positive...
reaction to generate an aziridine. Amino alcohols have the same reactivity, but the hydroxy
Hydroxyl
A hydroxyl is a chemical group containing an oxygen atom covalently bonded with a hydrogen atom. In inorganic chemistry, the hydroxyl group is known as the hydroxide ion, and scientists and reference works generally use these different terms though they refer to the same chemical structure in...
group must first be converted into a good leaving group
Leaving group
In chemistry, a leaving group is a molecular fragment that departs with a pair of electrons in heterolytic bond cleavage. Leaving groups can be anions or neutral molecules. Common anionic leaving groups are halides such as Cl−, Br−, and I−, and sulfonate esters, such as para-toluenesulfonate...
. The cyclization of an amino alcohol is called a Wenker synthesis
Wenker synthesis
The Wenker synthesis is an organic reaction converting a beta amino alcohol to an aziridine with the aid of sulfuric acid.The original Wenker synthesis of aziridine itself takes place in two steps...
(1935), and that of a haloamine the Gabriel ethylenimine method (1888)
Nitrene addition
NitreneNitrene
In chemistry, a nitrene is the nitrogen analogue of a carbene. The nitrogen atom has only 6 valence electrons and is therefore considered an electrophile...
addition to alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
s is a well-established method for the synthesis of aziridines. Photolysis or thermolysis of azide
Azide
Azide is the anion with the formula N3−. It is the conjugate base of hydrazoic acid. N3− is a linear anion that is isoelectronic with CO2 and N2O. Per valence bond theory, azide can be described by several resonance structures, an important one being N−=N+=N−...
s are good ways to generate nitrenes. Nitrenes can also be prepared in situ from iodosobenzene diacetate and sulfonamide
Sulfonamide (chemistry)
In chemistry, the sulfonamide functional group is -S2-NH2, a sulfonyl group connected to an amine group.A sulfonamide is a compound that contains this group. The general formula is RSO2NH2, where R is some organic group. For example, "methanesulfonamide" is CH3SO2NH2...
s, or the ethoxycarbonylnitrene from the N-sulfonyloxy precursor.

Triazoline decomposition
Thermal treatment or photolysis of triazolines expels nitrogenNitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
, producing an aziridine. Triazolines can be generated by cycloaddition
Cycloaddition
A cycloaddition is a pericyclic chemical reaction, in which "two or more unsaturated molecules combine with the formation of a cyclic adduct in which there is a net reduction of the bond multiplicity." The resulting reaction is a cyclization reaction.Cycloadditions are usually described by the...
of alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
s with an azide
Azide
Azide is the anion with the formula N3−. It is the conjugate base of hydrazoic acid. N3− is a linear anion that is isoelectronic with CO2 and N2O. Per valence bond theory, azide can be described by several resonance structures, an important one being N−=N+=N−...
.
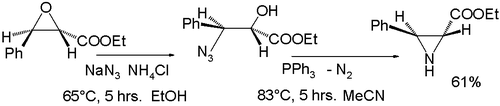
From epoxides
One method involves the ring-opening reaction of an epoxideEpoxide
An epoxide is a cyclic ether with three ring atoms. This ring approximately defines an equilateral triangle, which makes it highly strained. The strained ring makes epoxides more reactive than other ethers. Simple epoxides are named from the parent compound ethylene oxide or oxirane, such as in...
with sodium azide
Sodium azide
Sodium azide is the inorganic compound with the formula NaN3. This colourless azide salt is the gas-forming component in many car airbag systems. It is used for the preparation of other azide compounds. It is an ionic substance and is highly soluble in water. It is extremely...
, followed by organic reduction of the azide
Azide
Azide is the anion with the formula N3−. It is the conjugate base of hydrazoic acid. N3− is a linear anion that is isoelectronic with CO2 and N2O. Per valence bond theory, azide can be described by several resonance structures, an important one being N−=N+=N−...
with triphenylphosphine
Triphenylphosphine
Triphenylphosphine is a common organophosphorus compound with the formula P3 - often abbreviated to PPh3 or Ph3P. It is widely used in the synthesis of organic and organometallic compounds. PPh3 exists as relatively air stable, colorless crystals at room temperature...
accompanied by expulsion of nitrogen gas:

Epoxide
An epoxide is a cyclic ether with three ring atoms. This ring approximately defines an equilateral triangle, which makes it highly strained. The strained ring makes epoxides more reactive than other ethers. Simple epoxides are named from the parent compound ethylene oxide or oxirane, such as in...
with amine
Amine
Amines are organic compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group. Important amines include amino acids, biogenic amines,...
s, followed by ring closing with the Mitsunobu reaction
Mitsunobu reaction
The Mitsunobu reaction is an organic reaction that converts an alcohol into a variety of functional groups, such as an ester, using triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate or diisopropyl azodicarboxylate . The alcohol undergoes an inversion of stereochemistry...
.
From oximes
The Hoch-Campbell ethylenimine synthesis is the reaction of certain oximeOxime
An oxime is a chemical compound belonging to the imines, with the general formula R1R2C=NOH, where R1 is an organic side chain and R2 may be hydrogen, forming an aldoxime, or another organic group, forming a ketoxime. O-substituted oximes form a closely related family of compounds...
s with Grignard reagents:

Nucleophilic ring opening
Aziridines are reactive substrates in ring-opening reactions with many nucleophileNucleophile
A nucleophile is a species that donates an electron-pair to an electrophile to form a chemical bond in a reaction. All molecules or ions with a free pair of electrons can act as nucleophiles. Because nucleophiles donate electrons, they are by definition Lewis bases.Nucleophilic describes the...
s due to their ring strain
Ring strain
In organic chemistry, ring strain is the tendency of a cyclic molecule, such as cyclopropane, to destabilize when its atoms are in non-favorable high energy spatial orientations...
. Alcoholysis and aminolysis are basically the reverse reactions of the cyclizations. Carbon nucleophiles such as organolithium reagent
Organolithium reagent
An organolithium reagent is an organometallic compound with a direct bond between a carbon and a lithium atom. As the electropositive nature of lithium puts most of the charge density of the bond on the carbon atom, effectively creating a carbanion, organolithium compounds are extremely powerful...
s and organocuprates are also effective.
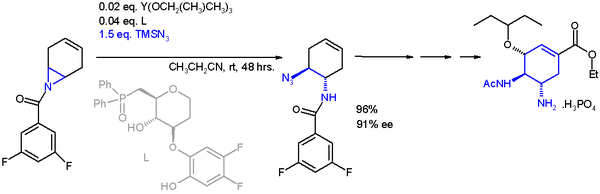
One application of a ring-opening reaction in asymmetric synthesis is that of trimethylsilylazide with an asymmetric ligand in scheme 2 in an organic synthesis
Organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the construction of organic compounds via organic reactions. Organic molecules can often contain a higher level of complexity compared to purely inorganic compounds, so the synthesis of organic compounds has...
of oseltamivir
Oseltamivir total synthesis
Oseltamivir total synthesis concerns the total synthesis of the antiinfluenza drug oseltamivir marketed by Hoffmann-La Roche under the trade name Tamiflu. Its commercial production starts from the biomolecule shikimic acid harvested from Chinese star anise with a limited worldwide supply...
:

1,3-dipole formation
Certain N-substituted azirines with electron withdrawing groups on both carbons form azomethine ylides in an electrocyclicElectrocyclic reaction
In organic chemistry, an electrocyclic reaction is a type of pericyclic rearrangement reaction where the net result is one pi bond being converted into one sigma bond or vice-versa...
thermal or photochemical ring-opening reaction. These ylides can be trapped with a suitable dipolarophile in a 1,3-dipolar cycloaddition
1,3-dipolar cycloaddition
The 1,3-dipolar cycloaddition, also known as the Huisgen cycloaddition or Huisgen reaction, is an organic chemical reaction belonging to the larger class of concerted, pericyclic cycloadditions. It is the reaction between a 1,3-dipole and a dipolarophile, most of which are substituted alkenes, to...
.
When the N-substituent is an electron-withdrawing group such as a tosyl
Tosyl
A tosyl group is CH3C6H4SO2. This group is usually derived from the compound 4-toluenesulfonyl chloride, CH3C6H4SO2Cl, which forms esters and amides of toluenesulfonic or tosylic acid...
group, the carbon-nitrogen bond
Carbon-nitrogen bond
A carbon–nitrogen bond is a covalent bond between carbon and nitrogen and is one of the most abundant bonds in organic chemistry and biochemistry....
breaks, forming another zwitterion
Zwitterion
In chemistry, a zwitterion is a neutral molecule with a positive and a negative electrical charge at different locations within that molecule. Zwitterions are sometimes also called inner salts.-Examples:...
This reaction type requires a Lewis acid
Lewis acid
]The term Lewis acid refers to a definition of acid published by Gilbert N. Lewis in 1923, specifically: An acid substance is one which can employ a lone pair from another molecule in completing the stable group of one of its own atoms. Thus, H+ is a Lewis acid, since it can accept a lone pair,...
catalyst such as boron trifluoride
Boron trifluoride
Boron trifluoride is the chemical compound with the formula BF3. This pungent colourless toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.-Structure and bonding:...
. In this way 2-phenyl-N-tosylaziridine reacts with alkynes, nitrile
Nitrile
A nitrile is any organic compound that has a -C≡N functional group. The prefix cyano- is used interchangeably with the term nitrile in industrial literature. Nitriles are found in many useful compounds, one example being super glue .Inorganic compounds containing the -C≡N group are not called...
s, ketone
Ketone
In organic chemistry, a ketone is an organic compound with the structure RCR', where R and R' can be a variety of atoms and groups of atoms. It features a carbonyl group bonded to two other carbon atoms. Many ketones are known and many are of great importance in industry and in biology...
s and alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
s. Certain 1,4-dipoles form from azetidine
Azetidine
Azetidine is a heterocyclic organic compound. It belongs to the class of four membered rings and it contains a nitrogen atom.-External links:* *...
s.
Other
N-unsubstituted aziridines can be opened with olefins in the presence of strong Lewis acid B(.Human toxicology
The toxicology of a particular aziridine compound depends on its structure and activity, although sharing the general characteristics of aziridines. As electrophileElectrophile
In general electrophiles are positively charged species that are attracted to an electron rich centre. In chemistry, an electrophile is a reagent attracted to electrons that participates in a chemical reaction by accepting an electron pair in order to bond to a nucleophile...
s, aziridines are subject to attack and ring-opening by endogenous nucleophiles such as nitrogenous bases in DNA base pairs, resulting in potential mutagenicity.
Exposure
Inhalation and direct contact.Some reports note that the use of gloves has not prevented permeation of aziridine. It is therefore important that users check the breakthrough permeation times for gloves, and pay scrupulous attention to avoiding contamination when degloving.
Carcinogenicity
The International Agency for Research on CancerInternational Agency for Research on Cancer
The International Agency for Research on Cancer is an intergovernmental agency forming part of the World Health Organisation of the United Nations....
(IARC) has reviewed aziridine compounds and classified them as possibly carcinogenic to humans (IARC Group 2B). In making the overall evaluation, the IARC Working Group took into consideration that aziridine is a direct-acting alkylating agent which is mutagenic in a wide range of test systems and forms DNA adducts that are promutagenic.
Irritancy
Aziridines are irritants of mucosal surfaces including eyes, nose, respiratory tract and skin.
Sensitization
Aziridine rapidly penetrates skin on contact.
Skin sensitizer — causing allergic contact dermatitis and urticaria
Urticaria
Urticaria is a kind of skin rash notable for pale red, raised, itchy bumps. Hives is frequently caused by allergic reactions; however, there are many non-allergic causes...
.
Respiratory sensitiser — causing occupational asthma

