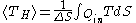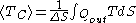Carnot heat engine
Encyclopedia
A Carnot heat engine is a hypothetical engine that operates on the reversible Carnot cycle
. The basic model for this engine was developed by Nicolas Léonard Sadi Carnot
in 1824. The Carnot engine model was graphically expanded upon by Benoît Paul Émile Clapeyron
in 1834 and mathematically elaborated upon by Rudolf Clausius
in the 1850s and 60s from which the concept of entropy
emerged.
Every thermodynamic system exists in a particular state
. A thermodynamic cycle
occurs when a system
is taken through a series of different states, and finally returned to its initial state. In the process of going through this cycle, the system may perform work on its surroundings, thereby acting as a heat engine
.
A heat engine acts by transferring energy from a warm region to a cool region of space and, in the process, converting some of that energy to mechanical work
. The cycle may also be reversed. The system may be worked upon by an external force, and in the process, it can transfer thermal energy from a cooler system to a warmer one, thereby acting as a refrigerator
or heat pump
rather than a heat engine.
In the adjacent diagram, from Carnot's 1824 work, Reflections on the Motive Power of Fire
, there are "two bodies A and B, kept each at a constant temperature, that of A being higher than that of B. These two bodies, to which we can give or from which we can remove the heat without causing their temperatures to vary, exercise the functions of two unlimited reservoirs of caloric
. We will call the first the furnace
and the second the refrigerator.” Carnot then explains how we can obtain motive power
, i.e. “work”, by carrying a certain quantity of heat from body A to body B.
 The previous image shows the original piston-and-cylinder diagram used by Carnot in discussing his ideal engine. The figure at right shows a block diagram of a generic heat engine, such as the Carnot engine. In the diagram, the “working body” (system), a term introduced by Clausius in 1850, can be any fluid or vapor body through which heat
The previous image shows the original piston-and-cylinder diagram used by Carnot in discussing his ideal engine. The figure at right shows a block diagram of a generic heat engine, such as the Carnot engine. In the diagram, the “working body” (system), a term introduced by Clausius in 1850, can be any fluid or vapor body through which heat
Q can be introduced or transmitted to produce work. Carnot had postulated that the fluid body could be any substance capable of expansion, such as vapor of water, vapor of alcohol, vapor of mercury, a permanent gas, or air, etc. Although, in these early years, engines came in a number of configurations, typically QH was supplied by a boiler, wherein water was boiled over a furnace; QC was typically a stream of cold flowing water in the form of a condenser
located on a separate part of the engine. The output work W here is the movement of the piston as it is used to turn a crank-arm, which was then typically used to turn a pulley so to lift water out of flooded salt mines. Carnot defined work as “weight lifted through a height”.
 Carnot's theorem is a formal statement of this fact: No engine operating between two heat reservoirs can be more efficient than a Carnot engine operating between the same reservoirs.
Carnot's theorem is a formal statement of this fact: No engine operating between two heat reservoirs can be more efficient than a Carnot engine operating between the same reservoirs.
This maximum efficiency is defined to be:
is defined to be: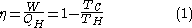
where
A corollary to Carnot's theorem states that: All reversible engines operating between the same heat reservoirs are equally efficient.
In other words, maximum efficiency is achieved if and only if no new entropy
is created in the cycle. Otherwise, since entropy
is a state function
, the required dumping of heat into the environment to dispose of excess entropy
leads to a reduction in efficiency. So Equation (1) gives the efficiency of any reversible
heat engine
.
The Coefficient of Performance (COP) of the heat engine is the reciprocal of its efficiency.
Although Carnot's cycle is an idealisation, the expression
of Carnot efficiency is still useful. Consider the average
temperatures,
at which heat is input and output, respectively. Replace TH and TC in Equation (1) by <TH> and <TC> respectively.
For the Carnot cycle, or an equivalent, <TH> is the highest temperature available and <TC> the lowest. For other less efficient cycles, <TH> will be lower than TH, and <TC> will be higher than TC. This can help illustrate, for example, why a reheater
or a regenerator can improve thermal efficiency.
Carnot cycle
The Carnot cycle is a theoretical thermodynamic cycle proposed by Nicolas Léonard Sadi Carnot in 1824 and expanded by Benoit Paul Émile Clapeyron in the 1830s and 40s. It can be shown that it is the most efficient cycle for converting a given amount of thermal energy into work, or conversely,...
. The basic model for this engine was developed by Nicolas Léonard Sadi Carnot
Nicolas Léonard Sadi Carnot
Nicolas Léonard Sadi Carnot was a French military engineer who, in his 1824 Reflections on the Motive Power of Fire, gave the first successful theoretical account of heat engines, now known as the Carnot cycle, thereby laying the foundations of the second law of thermodynamics...
in 1824. The Carnot engine model was graphically expanded upon by Benoît Paul Émile Clapeyron
Benoit Paul Émile Clapeyron
Benoît Paul Émile Clapeyron was a French engineer and physicist, one of the founders of thermodynamics.-Life:...
in 1834 and mathematically elaborated upon by Rudolf Clausius
Rudolf Clausius
Rudolf Julius Emanuel Clausius , was a German physicist and mathematician and is considered one of the central founders of the science of thermodynamics. By his restatement of Sadi Carnot's principle known as the Carnot cycle, he put the theory of heat on a truer and sounder basis...
in the 1850s and 60s from which the concept of entropy
Entropy
Entropy is a thermodynamic property that can be used to determine the energy available for useful work in a thermodynamic process, such as in energy conversion devices, engines, or machines. Such devices can only be driven by convertible energy, and have a theoretical maximum efficiency when...
emerged.
Every thermodynamic system exists in a particular state
Thermodynamic state
A thermodynamic state is a set of values of properties of a thermodynamic system that must be specified to reproduce the system. The individual parameters are known as state variables, state parameters or thermodynamic variables. Once a sufficient set of thermodynamic variables have been...
. A thermodynamic cycle
Thermodynamic cycle
A thermodynamic cycle consists of a series of thermodynamic processes transferring heat and work, while varying pressure, temperature, and other state variables, eventually returning a system to its initial state...
occurs when a system
Thermodynamic system
A thermodynamic system is a precisely defined macroscopic region of the universe, often called a physical system, that is studied using the principles of thermodynamics....
is taken through a series of different states, and finally returned to its initial state. In the process of going through this cycle, the system may perform work on its surroundings, thereby acting as a heat engine
Heat engine
In thermodynamics, a heat engine is a system that performs the conversion of heat or thermal energy to mechanical work. It does this by bringing a working substance from a high temperature state to a lower temperature state. A heat "source" generates thermal energy that brings the working substance...
.
A heat engine acts by transferring energy from a warm region to a cool region of space and, in the process, converting some of that energy to mechanical work
Mechanical work
In physics, work is a scalar quantity that can be described as the product of a force times the distance through which it acts, and it is called the work of the force. Only the component of a force in the direction of the movement of its point of application does work...
. The cycle may also be reversed. The system may be worked upon by an external force, and in the process, it can transfer thermal energy from a cooler system to a warmer one, thereby acting as a refrigerator
Refrigerator
A refrigerator is a common household appliance that consists of a thermally insulated compartment and a heat pump that transfers heat from the inside of the fridge to its external environment so that the inside of the fridge is cooled to a temperature below the ambient temperature of the room...
or heat pump
Heat pump
A heat pump is a machine or device that effectively "moves" thermal energy from one location called the "source," which is at a lower temperature, to another location called the "sink" or "heat sink", which is at a higher temperature. An air conditioner is a particular type of heat pump, but the...
rather than a heat engine.
In the adjacent diagram, from Carnot's 1824 work, Reflections on the Motive Power of Fire
Reflections on the Motive Power of Fire
In 1824, French physicist Sadi Carnot published the book Reflections on the Motive Power of Fire and on Machines Fitted to Develop that Power....
, there are "two bodies A and B, kept each at a constant temperature, that of A being higher than that of B. These two bodies, to which we can give or from which we can remove the heat without causing their temperatures to vary, exercise the functions of two unlimited reservoirs of caloric
Caloric theory
The caloric theory is an obsolete scientific theory that heat consists of a self-repellent fluid called caloric that flows from hotter bodies to colder bodies. Caloric was also thought of as a weightless gas that could pass in and out of pores in solids and liquids...
. We will call the first the furnace
Furnace
A furnace is a device used for heating. The name derives from Latin fornax, oven.In American English and Canadian English, the term furnace on its own is generally used to describe household heating systems based on a central furnace , and sometimes as a synonym for kiln, a device used in the...
and the second the refrigerator.” Carnot then explains how we can obtain motive power
Motive power
In thermodynamics, motive power is an agency, as water or steam, used to impart motion. Generally, motive power is defined as a natural agent, as water, steam, wind, electricity, etc., used to impart motion to machinery; a motor; a mover. The term may also define something, as a locomotive or a...
, i.e. “work”, by carrying a certain quantity of heat from body A to body B.
Modern diagram

Heat
In physics and thermodynamics, heat is energy transferred from one body, region, or thermodynamic system to another due to thermal contact or thermal radiation when the systems are at different temperatures. It is often described as one of the fundamental processes of energy transfer between...
Q can be introduced or transmitted to produce work. Carnot had postulated that the fluid body could be any substance capable of expansion, such as vapor of water, vapor of alcohol, vapor of mercury, a permanent gas, or air, etc. Although, in these early years, engines came in a number of configurations, typically QH was supplied by a boiler, wherein water was boiled over a furnace; QC was typically a stream of cold flowing water in the form of a condenser
Condenser (heat transfer)
In systems involving heat transfer, a condenser is a device or unit used to condense a substance from its gaseous to its liquid state, typically by cooling it. In so doing, the latent heat is given up by the substance, and will transfer to the condenser coolant...
located on a separate part of the engine. The output work W here is the movement of the piston as it is used to turn a crank-arm, which was then typically used to turn a pulley so to lift water out of flooded salt mines. Carnot defined work as “weight lifted through a height”.
Carnot's theorem

This maximum efficiency
 is defined to be:
is defined to be: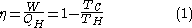
where
-
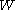 is the work done by the system (energy exiting the system as work),
is the work done by the system (energy exiting the system as work), -
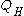 is the heat put into the system (heat energy entering the system),
is the heat put into the system (heat energy entering the system), -
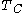 is the absolute temperature of the cold reservoir, and
is the absolute temperature of the cold reservoir, and -
 is the absolute temperature of the hot reservoir.
is the absolute temperature of the hot reservoir.
A corollary to Carnot's theorem states that: All reversible engines operating between the same heat reservoirs are equally efficient.
In other words, maximum efficiency is achieved if and only if no new entropy
Entropy
Entropy is a thermodynamic property that can be used to determine the energy available for useful work in a thermodynamic process, such as in energy conversion devices, engines, or machines. Such devices can only be driven by convertible energy, and have a theoretical maximum efficiency when...
is created in the cycle. Otherwise, since entropy
Entropy
Entropy is a thermodynamic property that can be used to determine the energy available for useful work in a thermodynamic process, such as in energy conversion devices, engines, or machines. Such devices can only be driven by convertible energy, and have a theoretical maximum efficiency when...
is a state function
State function
In thermodynamics, a state function, function of state, state quantity, or state variable is a property of a system that depends only on the current state of the system, not on the way in which the system acquired that state . A state function describes the equilibrium state of a system...
, the required dumping of heat into the environment to dispose of excess entropy
Entropy
Entropy is a thermodynamic property that can be used to determine the energy available for useful work in a thermodynamic process, such as in energy conversion devices, engines, or machines. Such devices can only be driven by convertible energy, and have a theoretical maximum efficiency when...
leads to a reduction in efficiency. So Equation (1) gives the efficiency of any reversible
Reversible process (thermodynamics)
In thermodynamics, a reversible process, or reversible cycle if the process is cyclic, is a process that can be "reversed" by means of infinitesimal changes in some property of the system without loss or dissipation of energy. Due to these infinitesimal changes, the system is in thermodynamic...
heat engine
Heat engine
In thermodynamics, a heat engine is a system that performs the conversion of heat or thermal energy to mechanical work. It does this by bringing a working substance from a high temperature state to a lower temperature state. A heat "source" generates thermal energy that brings the working substance...
.
The Coefficient of Performance (COP) of the heat engine is the reciprocal of its efficiency.
Efficiency of real heat engines
Carnot realized that in reality it is not possible to build a thermodynamically reversible engine, so real heat engines are less efficient than indicated by Equation (1). Nevertheless, Equation (1) is extremely useful for determining the maximum efficiency that could ever be expected for a given set of thermal reservoirs.Although Carnot's cycle is an idealisation, the expression
of Carnot efficiency is still useful. Consider the average
Average
In mathematics, an average, or central tendency of a data set is a measure of the "middle" value of the data set. Average is one form of central tendency. Not all central tendencies should be considered definitions of average....
temperatures,
at which heat is input and output, respectively. Replace TH and TC in Equation (1) by <TH> and <TC> respectively.
For the Carnot cycle, or an equivalent, <TH> is the highest temperature available and <TC> the lowest. For other less efficient cycles, <TH> will be lower than TH, and <TC> will be higher than TC. This can help illustrate, for example, why a reheater
Economizer
Economizers , or economisers , are mechanical devices intended to reduce energy consumption, or to perform another useful function such as preheating a fluid. The term economizer is used for other purposes as well. Boiler, powerplant, and heating, ventilating, and air-conditioning uses are...
or a regenerator can improve thermal efficiency.
- See also: Heat engineHeat engineIn thermodynamics, a heat engine is a system that performs the conversion of heat or thermal energy to mechanical work. It does this by bringing a working substance from a high temperature state to a lower temperature state. A heat "source" generates thermal energy that brings the working substance...
(efficiency and other performance criteria)
See also
- Carnot cycleCarnot cycleThe Carnot cycle is a theoretical thermodynamic cycle proposed by Nicolas Léonard Sadi Carnot in 1824 and expanded by Benoit Paul Émile Clapeyron in the 1830s and 40s. It can be shown that it is the most efficient cycle for converting a given amount of thermal energy into work, or conversely,...
- Heat engineHeat engineIn thermodynamics, a heat engine is a system that performs the conversion of heat or thermal energy to mechanical work. It does this by bringing a working substance from a high temperature state to a lower temperature state. A heat "source" generates thermal energy that brings the working substance...
- Thermal efficiencyThermal efficiencyIn thermodynamics, the thermal efficiency is a dimensionless performance measure of a device that uses thermal energy, such as an internal combustion engine, a boiler, a furnace, or a refrigerator for example.-Overview:...
- History of the internal combustion engineHistory of the internal combustion engineAlthough various forms of internal combustion engines were developed before the 19th century, their use was hindered until the commercial drilling and production of petroleum began in the mid-1850s...
- Adiabatic processAdiabatic processIn thermodynamics, an adiabatic process or an isocaloric process is a thermodynamic process in which the net heat transfer to or from the working fluid is zero. Such a process can occur if the container of the system has thermally-insulated walls or the process happens in an extremely short time,...