Cope rearrangement
Encyclopedia
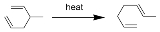
The Cope rearrangement is an extensively studied organic reaction
involving the [3,3]-sigmatropic rearrangement of 1,5-dienes. It was developed by Arthur C. Cope
. For example 3-methyl-1,5-hexadiene heated to 300°C yields 1,5-heptadiene.
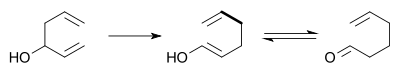
 The Cope rearrangement causes the fluxional states of the molecules in the bullvalene
The Cope rearrangement causes the fluxional states of the molecules in the bullvalene
family.
and pericyclic, it can also be considered to go via a transition state
that is energetically and structurally equivalent to a diradical
. This is an alternative explanation which remains faithful to the uncharged nature of the Cope transition state, while preserving the principles of orbital symmetry. This also explains the high energy requirement to perform a Cope rearrangement. Although illustrated in the chair conformation, the Cope can also occur with cyclohexadienes in the "boat" conformation.
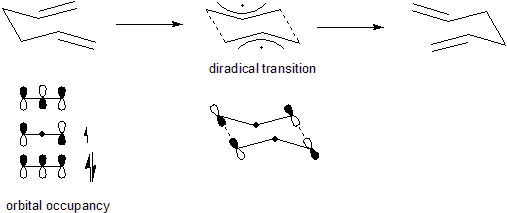
The above description of the transition state is not quite correct. It is currently generally accepted that the Cope rearrangement follows an allowed concerted route through a homoaromatic transition state and not a diradical. That is unless the potential energy surface is perturbed to favor the diradical.
-allowed when it is suprafacial on all components. The transition state of the molecule passes through a boat or chair like transition state. An example of the Cope rearrangement is the expansion of a cyclobutane
ring to a 1,5-cyclooctadiene
ring:
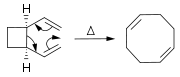
In this case, the reaction must pass through the boat transition state
to produce the two cis
double bond
s. A trans double bond in the ring would be too strained
. The reaction occurs under thermal conditions. The driving force of the reaction is the loss of strain from the cyclobutane ring.
group is added at C3 forming a enal or enone after Keto-enol tautomerism
of the intermediate enol :
for instance in this reaction:
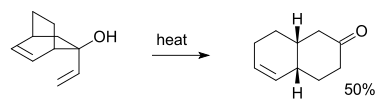
In 1975, Evans
and Golob showed that deprotonation of oxy-Cope substrates to form the corresponding alkali metal alkoxides resulted in rate accelerations of 1010 to 1017 for the oxy-Cope rearrangement. Typically potassium hydride and 18-crown-6 are employed in order to generate a fully dissociated potassium alkoxide :
It is noteworthy that the anion-accelerated oxy-Cope reaction can proceed with high efficiency even in systems that do not permit good orbital overlap
, as seen in this example from Schreiber's synthesis periplanone B :
The authors remark that the corresponding neutral oxy-Cope and siloxy-Cope rearrangements failed, giving only elimination products at 200 °C.
. Also see the divinylcyclopropane-cycloheptadiene rearrangement
.
Organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, photochemical reactions and redox reactions. In organic synthesis,...
involving the [3,3]-sigmatropic rearrangement of 1,5-dienes. It was developed by Arthur C. Cope
Arthur C. Cope
Arthur C. Cope was a highly successful and influential organic chemist and member of the National Academy of Sciences. He is credited with the development of several important chemical reactions which bear his name including the Cope elimination and the Cope rearrangement.Cope was born on June...
. For example 3-methyl-1,5-hexadiene heated to 300°C yields 1,5-heptadiene.

Bullvalene
Bullvalene is a hydrocarbon with the chemical formula C10H10 with the unusual property that the chemical bonds making up the molecule are constantly rearranging as in fluxional molecules...
family.
Mechanism
Although the Cope rearrangement is concertedConcerted reaction
In chemistry, a concerted reaction is a chemical reaction in which all bond breaking and bond making occurs in a single step. Reactive intermediates or other unstable high energy intermediates are not involved. Concerted reaction rates tend not to depend on solvent polarity ruling out large buildup...
and pericyclic, it can also be considered to go via a transition state
Transition state
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest energy along this reaction coordinate. At this point, assuming a perfectly irreversible reaction, colliding reactant molecules will always...
that is energetically and structurally equivalent to a diradical
Radical (chemistry)
Radicals are atoms, molecules, or ions with unpaired electrons on an open shell configuration. Free radicals may have positive, negative, or zero charge...
. This is an alternative explanation which remains faithful to the uncharged nature of the Cope transition state, while preserving the principles of orbital symmetry. This also explains the high energy requirement to perform a Cope rearrangement. Although illustrated in the chair conformation, the Cope can also occur with cyclohexadienes in the "boat" conformation.

The above description of the transition state is not quite correct. It is currently generally accepted that the Cope rearrangement follows an allowed concerted route through a homoaromatic transition state and not a diradical. That is unless the potential energy surface is perturbed to favor the diradical.
Examples
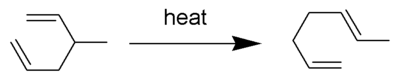
The rearrangement is widely used in organic synthesis. It is symmetrySymmetry
Symmetry generally conveys two primary meanings. The first is an imprecise sense of harmonious or aesthetically pleasing proportionality and balance; such that it reflects beauty or perfection...
-allowed when it is suprafacial on all components. The transition state of the molecule passes through a boat or chair like transition state. An example of the Cope rearrangement is the expansion of a cyclobutane
Cyclobutane
Cyclobutane is an organic compound with the formula 4. Cyclobutane is a colourless gas and commercially available as a liquefied gas. Derivatives of cyclobutane are called cyclobutanes...
ring to a 1,5-cyclooctadiene
1,5-Cyclooctadiene
1,5-Cyclooctadiene is the organic compound with the chemical formula C8H12. Generally abbreviated COD, this diene is a useful precursor to other organic compounds and serves as a ligand in organometallic chemistry.- Synthesis :...
ring:

In this case, the reaction must pass through the boat transition state
Transition state
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest energy along this reaction coordinate. At this point, assuming a perfectly irreversible reaction, colliding reactant molecules will always...
to produce the two cis
Geometric isomerism
In organic chemistry, cis/trans isomerism or geometric isomerism or configuration isomerism or E/Z isomerism is a form of stereoisomerism describing the orientation of functional groups within a molecule...
double bond
Double bond
A double bond in chemistry is a chemical bond between two chemical elements involving four bonding electrons instead of the usual two. The most common double bond, that between two carbon atoms, can be found in alkenes. Many types of double bonds between two different elements exist, for example in...
s. A trans double bond in the ring would be too strained
Ring strain
In organic chemistry, ring strain is the tendency of a cyclic molecule, such as cyclopropane, to destabilize when its atoms are in non-favorable high energy spatial orientations...
. The reaction occurs under thermal conditions. The driving force of the reaction is the loss of strain from the cyclobutane ring.
Oxy-Cope rearrangement
In the Oxy-Cope rearrangement a hydroxylHydroxyl
A hydroxyl is a chemical group containing an oxygen atom covalently bonded with a hydrogen atom. In inorganic chemistry, the hydroxyl group is known as the hydroxide ion, and scientists and reference works generally use these different terms though they refer to the same chemical structure in...
group is added at C3 forming a enal or enone after Keto-enol tautomerism
Keto-enol tautomerism
In organic chemistry, keto-enol tautomerism refers to a chemical equilibrium between a keto form and an enol . The enol and keto forms are said to be tautomers of each other...
of the intermediate enol :
for instance in this reaction:

In 1975, Evans
David A. Evans
David A. Evans is the Abbott and James Lawrence Professor of Chemistry in the Department of Chemistry and Chemical Biology at Harvard University...
and Golob showed that deprotonation of oxy-Cope substrates to form the corresponding alkali metal alkoxides resulted in rate accelerations of 1010 to 1017 for the oxy-Cope rearrangement. Typically potassium hydride and 18-crown-6 are employed in order to generate a fully dissociated potassium alkoxide :
It is noteworthy that the anion-accelerated oxy-Cope reaction can proceed with high efficiency even in systems that do not permit good orbital overlap
Orbital overlap
Orbital overlap was an idea first introduced by Linus Pauling to explain the molecular bond angles observed through experimentation and is the basis for the concept of orbital hybridisation. s orbitals are spherical and have no directionality while p orbitals are oriented 90° to one another...
, as seen in this example from Schreiber's synthesis periplanone B :
The authors remark that the corresponding neutral oxy-Cope and siloxy-Cope rearrangements failed, giving only elimination products at 200 °C.
Variations
Another variation of the Cope rearrangement is the heteroatom Cope reactions such as the Aza-Cope rearrangement. Another widely studied [3, 3] sigmatropic rearrangement is the Claisen rearrangementClaisen rearrangement
The Claisen rearrangement is a powerful carbon-carbon bond-forming chemical reaction discovered by Rainer Ludwig Claisen...
. Also see the divinylcyclopropane-cycloheptadiene rearrangement
Divinylcyclopropane-cycloheptadiene rearrangement
The divinylcyclopropane-cycloheptadiene rearrangement is an organic chemical transformation that involves the isomerization of a 1,2-divinylcyclopropane into a cycloheptadiene or -triene...
.