
History of the battery
Encyclopedia


Capacitor
A capacitor is a passive two-terminal electrical component used to store energy in an electric field. The forms of practical capacitors vary widely, but all contain at least two electrical conductors separated by a dielectric ; for example, one common construction consists of metal foils separated...
s he used for his experiments with electricity. These capacitors were panels of glass coated with metal on each surface. These capacitors were charged with a static generator and discharged by touching metal to their electrode. Linking them together in a "battery" gave a stronger discharge.
The Baghdad Battery of antiquity
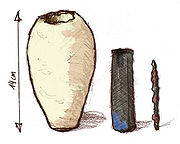
Baghdad Battery
The Baghdad Battery, sometimes referred to as the Parthian Battery, is the common name for a number of artifacts created in Mesopotamia, during the dynasties of Parthian or Sassanid period , and probably discovered in 1936 in the village of Khuyut Rabbou'a, near Baghdad, Iraq...
, sometimes referred to as the Parthian Battery, is the common name for a number of artifacts created in Mesopotamia during the Iranian
Iran
Iran , officially the Islamic Republic of Iran , is a country in Southern and Western Asia. The name "Iran" has been in use natively since the Sassanian era and came into use internationally in 1935, before which the country was known to the Western world as Persia...
dynasties of Parthian
Parthia
Parthia is a region of north-eastern Iran, best known for having been the political and cultural base of the Arsacid dynasty, rulers of the Parthian Empire....
or Sassanid period (the early centuries AD).
Around 1936, archaeologists uncovered in a village near Baghdad
Baghdad
Baghdad is the capital of Iraq, as well as the coterminous Baghdad Governorate. The population of Baghdad in 2011 is approximately 7,216,040...
a set of terracotta jars that each contained a rolled-up sheet of copper that housed an iron rod. Some scientists speculate these to be ancient galvanic cell
Galvanic cell
A Galvanic cell, or Voltaic cell, named after Luigi Galvani, or Alessandro Volta respectively, is an electrochemical cell that derives electrical energy from spontaneous redox reaction taking place within the cell...
s (roughly 2,000 years old, though the find's age is still debated), and dubbed them the "Baghdad Batteries
Baghdad Battery
The Baghdad Battery, sometimes referred to as the Parthian Battery, is the common name for a number of artifacts created in Mesopotamia, during the dynasties of Parthian or Sassanid period , and probably discovered in 1936 in the village of Khuyut Rabbou'a, near Baghdad, Iraq...
". It is believed a common food acid, such as lemon juice or vinegar, served as an electrolyte. Modern replicas have successfully produced currents, lending credence to this hypothesis. If the collection was indeed a battery, it could have been used for electroplating
Electroplating
Electroplating is a plating process in which metal ions in a solution are moved by an electric field to coat an electrode. The process uses electrical current to reduce cations of a desired material from a solution and coat a conductive object with a thin layer of the material, such as a metal...
jewelry, or to produce mild electric shocks as a source of religious experience. The artifacts strongly resemble another type of object with a known purpose — namely, storage vessels for sacred scrolls from nearby Seleucia on the Tigris. Those vessels do not have the outermost clay jar, but are otherwise almost identical. In this way, they could have been simply used to store ancient scrolls.
1800 - The Voltaic Pile

Luigi Galvani
Luigi Aloisio Galvani was an Italian physician and physicist who lived and died in Bologna. In 1791, he discovered that the muscles of dead frogs legs twitched when struck by a spark...
was dissecting a frog affixed to a brass hook. When he touched its leg with his iron scalpel, the leg twitched. Galvani believed the energy that drove this contraction came from the leg itself, and called it "animal electricity".
However, Alessandro Volta
Alessandro Volta
Count Alessandro Giuseppe Antonio Anastasio Gerolamo Umberto Volta was a Lombard physicist known especially for the invention of the battery in 1800.-Early life and works:...
, a friend and fellow scientist, disagreed, believing this phenomenon was caused by two different metals joined together by a moist intermediary. He verified this hypothesis through experiment, and published the results in 1791. In 1800, Volta invented the first true battery, which came to be known as the Voltaic Pile
Voltaic pile
A voltaic pile is a set of individual Galvanic cells placed in series. The voltaic pile, invented by Alessandro Volta in 1800, was the first electric battery...
. The Voltaic Pile
Voltaic pile
A voltaic pile is a set of individual Galvanic cells placed in series. The voltaic pile, invented by Alessandro Volta in 1800, was the first electric battery...
consisted of pairs of copper and zinc discs piled on top of each other, separated by a layer of cloth or cardboard soaked in brine (i.e., the electrolyte). Unlike the Leyden jar
Leyden jar
A Leyden jar, or Leiden jar, is a device that "stores" static electricity between two electrodes on the inside and outside of a jar. It was invented independently by German cleric Ewald Georg von Kleist on 11 October 1745 and by Dutch scientist Pieter van Musschenbroek of Leiden in 1745–1746. The...
, the Voltaic Pile produced a continuous and stable current, and lost little charge over time when not in use, though his early models could not produce a voltage strong enough to produce sparks. He experimented with various metals and found that zinc and silver gave the best results.
Thermoelectric effect
The thermoelectric effect is the direct conversion of temperature differences to electric voltage and vice-versa. A thermoelectric device creates a voltage when there is a different temperature on each side. Conversely, when a voltage is applied to it, it creates a temperature difference...
). As a consequence, he regarded the corrosion of the zinc plates as an unrelated flaw that could perhaps be fixed by changing the materials somehow. However, no scientist ever succeeded in preventing this corrosion. In fact, it was observed that the corrosion was faster when a higher current was drawn. This suggested that the corrosion was actually integral to the battery's ability to produce a current. This, in part, led to the rejection of Volta's contact tension theory in favor of electrochemical theory. Volta's illustrations of his Crown of Cups and Voltaic Pile have extra metal disks, now known to be unnecessary, on both the top and bottom. The figure associated with this section, of the zinc-copper voltaic pile, has the modern design, an indication that "contact tension" is not the source of electromotive force
Electromotive force
In physics, electromotive force, emf , or electromotance refers to voltage generated by a battery or by the magnetic force according to Faraday's Law, which states that a time varying magnetic field will induce an electric current.It is important to note that the electromotive "force" is not a...
for the voltaic pile.
William Cruickshank (chemist)
William Cruickshank was a Scottish military surgeon and chemist, and professor of chemistry at the Royal Military Academy, Woolwich.-Career:...
solved this problem by laying the elements in a box instead of piling them in a stack. This was known as the trough battery
Trough battery
The trough battery was a variant of Alessandro Volta's Voltaic Pile and was invented by William Cruickshank circa 1800.-Disadvantage of the pile:...
. Volta himself invented a variant that consisted of a chain of cups filled with a salt solution, linked together by metallic arcs dipped into the liquid. This was known as the Crown of Cups. These arcs were made of two different metals (e.g., zinc and copper) soldered together. This model also proved to be more efficient than his original piles, though it didn't prove as popular.
Another problem with Volta's batteries was short battery life - an hour's worth at best - which was caused by two phenomena. The first was that the current produced electrolysed the electrolyte solution, resulting in a film of hydrogen bubbles forming on the copper, which steadily increased the internal resistance of the battery (This effect, called polarization, is counteracted in modern cells by additional measures). The other was a phenomenon called local action, wherein minute short-circuits would form around impurities in the zinc, causing the zinc to degrade. The latter problem was solved in 1835 by William Sturgeon
William Sturgeon
William Sturgeon was an English physicist and inventor who made the first electromagnets, and invented the first practical English electric motor.-Early Life :...
, who found that mixing some mercury
Mercury (element)
Mercury is a chemical element with the symbol Hg and atomic number 80. It is also known as quicksilver or hydrargyrum...
into the zinc eliminated the local action.
Despite its flaws, Volta's batteries provided a steadier current than Leyden jars, and made possible many new experiments and discoveries, such as the first electrolysis of water by Anthony Carlisle and William Nicholson.
1836 - The Daniell cell
John Frederic Daniell
John Frederic Daniell was an English chemist and physicist.Daniell was born in London, and in 1831 became the first professor of chemistry at the newly founded King's College London. His name is best known for his invention of the Daniell cell , an electric battery much better than voltaic cells...
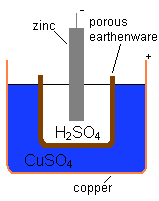
searched for a way to eliminate the hydrogen bubble problem found in the Voltaic Pile, and his solution was to use a second electrolyte to consume the hydrogen produced by the first. In 1836, he invented the Daniell cell
Daniell cell
The Daniell cell was invented in 1836 by John Frederic Daniell, a British chemist and meteorologist, and consisted of a copper pot filled with a copper sulfate solution, in which was immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode...
, which consisted of a copper pot filled with a copper sulphate solution, in which was immersed an unglazed earthenware
Earthenware
Earthenware is a common ceramic material, which is used extensively for pottery tableware and decorative objects.-Types of earthenware:Although body formulations vary between countries and even between individual makers, a generic composition is 25% ball clay, 28% kaolin, 32% quartz, and 15%...
container filled with sulphuric acid and a zinc electrode. The earthenware
Earthenware
Earthenware is a common ceramic material, which is used extensively for pottery tableware and decorative objects.-Types of earthenware:Although body formulations vary between countries and even between individual makers, a generic composition is 25% ball clay, 28% kaolin, 32% quartz, and 15%...
barrier was porous, which allowed ions to pass through but kept the solutions from mixing. Without this barrier, when no current was drawn the copper ions would drift to the zinc anode and undergo reduction without producing a current, which would destroy the battery's life.
Over time, copper buildup would block the pores in the earthenware barrier and cut short the battery's life. Nevertheless, the Daniell cell provided a longer and more reliable current than the Voltaic cell because the electrolyte deposited copper (a conductor) rather than hydrogen (an insulator) on the cathode. It was also safer and less corrosive. It had an operating voltage of roughly 1.1 volts. It saw widespread use in telegraph networks until it was supplanted by the Leclanché cell in the late 1860s.
1844 - The Grove cell
The Grove cellGrove cell
The Grove cell was an early electric primary cell named after its inventor, British chemist William Robert Grove, and consisted of a zinc anode in dilute sulfuric acid and a platinum cathode in concentrated nitric acid, the two separated by a porous ceramic pot.-Cell details:The Grove cell voltage...
was invented by William Robert Grove
William Robert Grove
Sir William Robert Grove PC QC FRS was a judge and physical scientist. He anticipated the general theory of the conservation of energy, and was a pioneer of fuel cell technology.-Early life:...
in 1844. It consisted of a zinc
Zinc
Zinc , or spelter , is a metallic chemical element; it has the symbol Zn and atomic number 30. It is the first element in group 12 of the periodic table. Zinc is, in some respects, chemically similar to magnesium, because its ion is of similar size and its only common oxidation state is +2...
anode dipped in sulfuric acid
Sulfuric acid
Sulfuric acid is a strong mineral acid with the molecular formula . Its historical name is oil of vitriol. Pure sulfuric acid is a highly corrosive, colorless, viscous liquid. The salts of sulfuric acid are called sulfates...
and a platinum
Platinum
Platinum is a chemical element with the chemical symbol Pt and an atomic number of 78. Its name is derived from the Spanish term platina del Pinto, which is literally translated into "little silver of the Pinto River." It is a dense, malleable, ductile, precious, gray-white transition metal...
cathode
Cathode
A cathode is an electrode through which electric current flows out of a polarized electrical device. Mnemonic: CCD .Cathode polarity is not always negative...
dipped in nitric acid
Nitric acid
Nitric acid , also known as aqua fortis and spirit of nitre, is a highly corrosive and toxic strong acid.Colorless when pure, older samples tend to acquire a yellow cast due to the accumulation of oxides of nitrogen. If the solution contains more than 86% nitric acid, it is referred to as fuming...
, separated by porous earthenware
Earthenware
Earthenware is a common ceramic material, which is used extensively for pottery tableware and decorative objects.-Types of earthenware:Although body formulations vary between countries and even between individual makers, a generic composition is 25% ball clay, 28% kaolin, 32% quartz, and 15%...
. The Grove cell provided a high current and nearly twice the voltage of the Daniell cell, which made it the favoured cell of the American telegraph networks for a time. However, it gave off poisonous nitric oxide fumes when operated. The voltage also dropped sharply as the charge diminished, which became a liability as telegraph networks grew more complex. Platinum
Platinum
Platinum is a chemical element with the chemical symbol Pt and an atomic number of 78. Its name is derived from the Spanish term platina del Pinto, which is literally translated into "little silver of the Pinto River." It is a dense, malleable, ductile, precious, gray-white transition metal...
was also very expensive. The Grove cell was replaced by the cheaper, safer and better performing gravity cell in the 1860s.
1859 - The lead-acid cell: the first rechargeable battery
Gaston Planté
Gaston Planté was the French physicist who invented the lead-acid battery in 1859. The lead-acid battery eventually became the first rechargeable electric battery marketed for commercial use.Planté was born on April 22, 1834, in Orthez, France...
invented the lead-acid battery
Lead-acid battery
Lead–acid batteries, invented in 1859 by French physicist Gaston Planté, are the oldest type of rechargeable battery. Despite having a very low energy-to-weight ratio and a low energy-to-volume ratio, their ability to supply high surge currents means that the cells maintain a relatively large...
, the first-ever battery that could be recharged by passing a reverse current through it. A lead acid cell consists of a lead anode and a lead dioxide cathode immersed in sulphuric acid. Both electrodes react with the acid to produce lead sulfate, but the reaction at the lead anode releases electrons whilst the reaction at the lead dioxide consumes them, thus producing a current. These chemical reactions can be reversed by passing a reverse current through the battery, thereby recharging it.
Planté's first model consisted of two lead sheets separated by rubber strips and rolled into a spiral. His batteries were first used to power the lights in train carriages while stopped at a station. In 1881, Camille Alphonse Faure invented an improved version that consisted of a lead grid lattice into which a lead oxide paste was pressed, forming a plate. Multiple plates could be stacked for greater performance. This design was easier to mass-produce.
Compared to other batteries, Planté's was rather heavy and bulky for the amount of energy it could hold. However, it could produce remarkably large currents in surges. It also had very low internal resistance, meaning that a single battery could be used to power multiple circuits.
The lead-acid battery is still used today in automobiles and other applications where weight is not a big factor. The basic principle has not changed since 1859, though in the 1970s a variant was developed that used a gel electrolyte instead of a liquid (commonly known as a "gel cell"), allowing the battery to be used in different positions without failure or leakage.
Today cells are classified as "primary" if they produce a current only until their chemical reactants are exhausted, and "secondary" if the chemical reactions can be reversed by recharging the cell. The lead-acid cell was the first "secondary" cell.
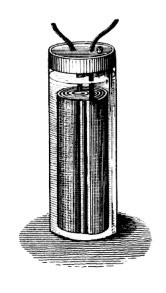
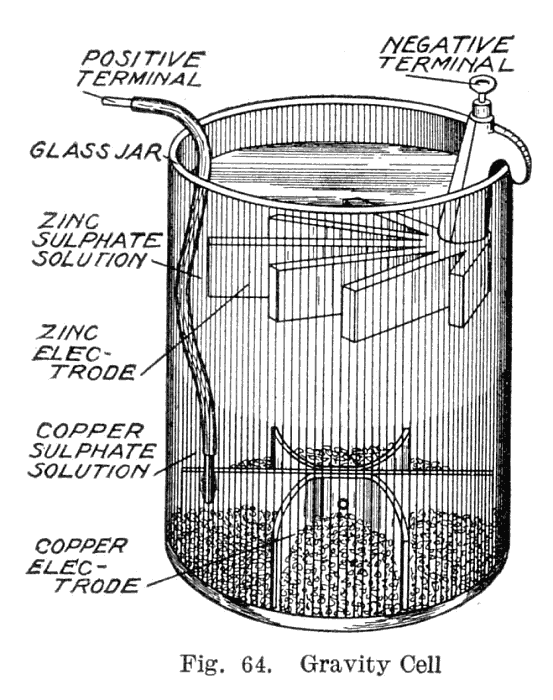
1860s - The gravity cell
Daniell cell
The Daniell cell was invented in 1836 by John Frederic Daniell, a British chemist and meteorologist, and consisted of a copper pot filled with a copper sulfate solution, in which was immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode...
called the gravity cell. This simpler version dispensed with the porous barrier. This reduced the internal resistance of the system and, thus, the battery yielded a stronger current. It quickly became the battery of choice for the American and British telegraph networks, and was used right up until the 1950s. In the telegraph industry, this battery was often assembled on site by the telegraph workers themselves, and when it ran down it could be renewed by replacing the consumed components.
The gravity cell consisted of a glass jar, in which a copper cathode sat on the bottom and a zinc anode was suspended beneath the rim. Copper sulfate crystals would be scattered around the cathode and then the jar would be filled with distilled water. As the current was drawn, a layer of zinc sulfate solution would form at the top around the anode. This top layer was kept separate from the bottom copper sulfate layer by its lower density and by the polarity of the cell.
The zinc sulfate layer was clear in contrast to the deep blue copper sulfate layer, which allowed a technician to measure the battery life with a glance. On the other hand, this setup meant the battery could be used only in a stationary appliance, else the solutions would mix or spill. Another disadvantage was that a current had to be continually drawn to keep the two solutions from mixing by diffusion, so it was unsuitable for intermittent use.
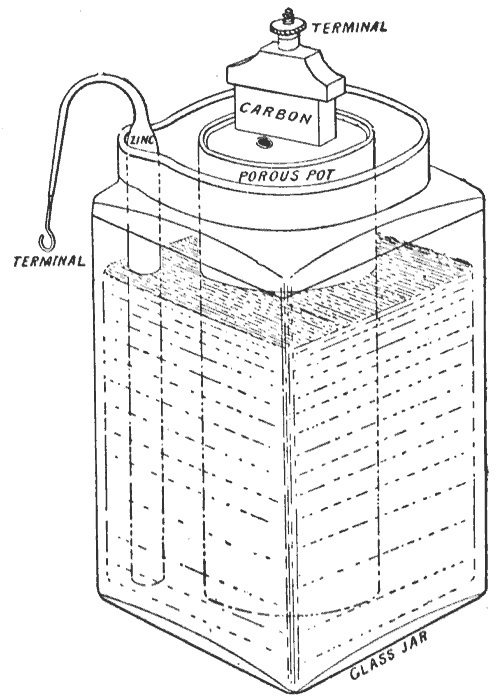
1866 - The Leclanché cell
In 1866, Georges LeclanchéGeorges Leclanché
Georges Leclanché was a French electrical engineer chiefly remembered for his invention of the Leclanché cell, one of the first modern electrical batteries and the forerunner of the modern dry cell battery.-Biography:...
invented a battery
Leclanché cell
Georges Leclanché invented and patented his battery, the Leclanché cell, in 1866. The battery contained a conducting solution of ammonium chloride, a cathode of carbon, a depolarizer of manganese dioxide, and an anode of zinc...
that consisted of a zinc anode and a manganese dioxide cathode wrapped in a porous material, dipped in a jar of ammonium chloride
Ammonium chloride
Ammonium chloride NH4Cl is an inorganic compound with the formula NH4Cl. It is a white crystalline salt that is highly soluble in water. Solutions of ammonium chloride are mildly acidic. Sal ammoniac is a name of natural, mineralogical form of ammonium chloride...
solution. The manganese dioxide cathode had a little carbon mixed into it as well, which improved electrolyte conductivity and absorption. It provided a voltage of 1.4 to 1.6 volts. This cell achieved very quick success in telegraphy, signalling and electric bell work. It was used to power early telephones—usually from an adjacent wooden box affixed to the wall—before telephones could draw power from the line itself. It could not provide a sustained current for very long. In lengthy conversations, the battery would run down, rendering the conversation inaudible. This was because certain chemical reactions in the cell increased the internal resistance and, thus, lowered the voltage. These reactions reversed themselves when the battery was left idle, so it was good only for intermittent use.
1887 - The zinc-carbon cell: the first dry cell
Many experimenters tried to immobilize the electrolyte of an electrochemical cell to make it more convenient to use. The Zamboni pileZamboni pile
The Zamboni pile is an early electric battery, invented by Giuseppe Zamboni in 1812.A Zamboni pile is an "electrostatic battery" and is constructed from discs of silver foil, zinc foil, and paper...
of 1812 was a high-voltage dry battery but capable of delivering only minute currents. Various experiments were made with cellulose, sawdust, spun glass, asbestos fibers, and gelatine.
In 1886, Carl Gassner obtained a German patent (No. 37,758) on a variant of the Leclanché cell, which came to be known as the dry cell
Zinc-carbon battery
A zinc–carbon dry cell or battery is packaged in a zinc can that serves as both a container and negative terminal. It was developed from the wet Leclanché cell . The positive terminal is a carbon rod surrounded by a mixture of manganese dioxide and carbon powder. The electrolyte used is a paste of...
because it did not have a free liquid electrolyte. Instead, the ammonium chloride was mixed with Plaster of Paris to create a paste, with a small amount of zinc chloride
Zinc chloride
Zinc chloride is the name of chemical compound with the formula ZnCl2 and its hydrates. Zinc chlorides, of which nine crystalline forms are known, are colorless or white, and are highly soluble in water. ZnCl2 itself is hygroscopic and even deliquescent. Samples should therefore be protected from...
added in to extend the shelf life. The manganese dioxide cathode was dipped in this paste, and both were sealed in a zinc shell, which also acted as the anode. In November 1887, he obtained a US patent for the same device.
Unlike previous wet cells, Gassner's dry cell was more solid, did not require maintenance, did not spill, and could be used in any orientation. It provided a potential of 1.5 volts. The first mass-produced model was the Columbia dry cell, first marketed by the National Carbon Company
National Carbon Company
The National Carbon Company was founded in 1886 by the former Brush Electric Company executive W. H. Lawrence, in association with Myron T. Herrick, James Parmelee, and Webb Hayes, son of U.S. President Rutherford B. Hayes, in Cleveland, Ohio. In 1890, National Carbon merged with Thomson-Houston,...
in 1896. The NCC improved Gassner's model by replacing the plaster of Paris with coiled cardboard, an innovation that left more space for the cathode and made the battery easier to assemble. It was the first convenient battery for the masses and made portable electrical devices practical. The flashlight
Flashlight
A flashlight is a hand-held electric-powered light source. Usually the light source is a small incandescent lightbulb or light-emitting diode...
was invented that same year.
The zinc-carbon battery
Zinc-carbon battery
A zinc–carbon dry cell or battery is packaged in a zinc can that serves as both a container and negative terminal. It was developed from the wet Leclanché cell . The positive terminal is a carbon rod surrounded by a mixture of manganese dioxide and carbon powder. The electrolyte used is a paste of...
(as it came to be known) is still manufactured today.
In parallel, in 1887 Frederik Louis Wilhelm Hellesen
Frederik Louis Wilhelm Hellesen
Frederik Louis Wilhelm Hellesen was a Danish inventor and industrialist. In 1887 he designed what is thought to be the first dry cell battery based on the Leclanché cell design. The same year he founded the company W. Hellesen In 1889 he sold his first batteries to the Danish Telephone Compagny...
developed his own dry cell design. It has been claimed that Hellesen's design preceded that of Gassner.
In 1889, a dry-battery was developed by Sakizou Yai (屋井先蔵) of Japan, then patented in 1891. Sakizou Yai coined the word "dry-battery". The word "dry-battery" originates from the Japanese word "Kandenchi" (乾 kan = dry, 電池 denchi = battery). In 1893, Sakizou Yai's dry-battery was exhibited in World's Columbian Exposition
World's Columbian Exposition
The World's Columbian Exposition was a World's Fair held in Chicago in 1893 to celebrate the 400th anniversary of Christopher Columbus's arrival in the New World in 1492. Chicago bested New York City; Washington, D.C.; and St...
and commanded considerable international attention.
1899 - The nickel-cadmium battery: the first alkaline battery
In 1899, a Swedish scientist named Waldemar JungnerWaldemar Jungner
Ernst Waldemar Jungner, June 19, 1869- August 30, 1924, was a Swedish inventor and engineer. In 1899 he invented the nickel-iron electric storage battery and invented the nickel-cadmium battery.-Literature:...
invented the nickel-cadmium battery
Nickel-cadmium battery
The nickel–cadmium battery ' is a type of rechargeable battery using nickel oxide hydroxide and metallic cadmium as electrodes....
, a rechargeable battery that had nickel and cadmium electrodes in a potassium hydroxide
Potassium hydroxide
Potassium hydroxide is an inorganic compound with the formula KOH, commonly called caustic potash.Along with sodium hydroxide , this colorless solid is a prototypical strong base. It has many industrial and niche applications. Most applications exploit its reactivity toward acids and its corrosive...
solution; the first battery to use an alkaline electrolyte. It was commercialised in Sweden in 1910 and reached the United States in 1946. The first models were robust and had significantly better energy density than lead-acid batteries, but were much more expensive.
1903 - The nickel-iron battery
Jungner also invented a nickel-iron batteryNickel-iron battery
The nickel–iron battery is a storage battery having a nickel oxide-hydroxide cathode and an iron anode, with an electrolyte of potassium hydroxide. The active materials are held in nickel-plated steel tubes or perforated pockets...
the same year as his Ni-Cad battery, but found it to be inferior to its cadmium counterpart and, as a consequence, never bothered patenting it. It produced a lot more hydrogen gas when being charged, meaning it could not be sealed, and the charging process was less efficient (it was, however, cheaper). However, Thomas Edison
Thomas Edison
Thomas Alva Edison was an American inventor and businessman. He developed many devices that greatly influenced life around the world, including the phonograph, the motion picture camera, and a long-lasting, practical electric light bulb. In addition, he created the world’s first industrial...
picked up Jugner's nickel-iron battery design, patented it himself and sold it in 1903. Edison wanted to commercialise a more lightweight and durable substitute for the lead-acid battery that powered some early automobiles, and hoped that by doing so electric cars would become the standard, with his firm as its main battery vendor. However, customers found his first model to be prone to leakage and short battery life, and it did not outperform the lead-acid cell by much either. Although Edison was able to produce a more reliable and powerful model seven years later, by this time the inexpensive and reliable Model T Ford had made gasoline engine cars the standard. Nevertheless, Edison's battery achieved great success in other applications.
1955 - The common alkaline battery
Up until the late 1950s the zinc-carbon batteryZinc-carbon battery
A zinc–carbon dry cell or battery is packaged in a zinc can that serves as both a container and negative terminal. It was developed from the wet Leclanché cell . The positive terminal is a carbon rod surrounded by a mixture of manganese dioxide and carbon powder. The electrolyte used is a paste of...
continued to be a popular primary cell battery, but its relatively low battery life hampered sales. In 1955, an engineer working for Union Carbide at the National Carbon Company Parma Research Laboratory named Lewis Urry
Lewis Urry
Lewis Frederick Urry, , was a Canadian chemical engineer and inventor. He invented both the alkaline battery and lithium battery while working for the Eveready Battery company....
was tasked with finding a way to extend the life of zinc-carbon batteries, but Urry decided instead that alkaline batteries held more promise. Up until now, longer-lasting alkaline batteries were unfeasibly expensive. Urry's battery consisted of a manganese dioxide cathode and a powdered zinc anode with an alkaline electrolyte. Using powdered zinc gave the anode a greater surface area. These batteries hit the market in 1959.
Early 1970s - The nickel hydrogen battery
The nickel hydrogen batteryNickel hydrogen battery
A nickel–hydrogen battery is a rechargeable electrochemical power source based on nickel and hydrogen. It differs from a nickel–metal hydride battery by the use of hydrogen in a pressurized cell at up to 1200 psi pressure.The cathode is made up of a dry sintered porous nickel plaque, which...
entered the market as an energy-storage subsystem for commercial communication satellites.
Late 1980s - The nickel metal-hydride battery
The first consumer grade NiMH batteries for smaller applications appeared on the market in 1989 as a variation of the 1970s nickel hydrogen batteryNickel hydrogen battery
A nickel–hydrogen battery is a rechargeable electrochemical power source based on nickel and hydrogen. It differs from a nickel–metal hydride battery by the use of hydrogen in a pressurized cell at up to 1200 psi pressure.The cathode is made up of a dry sintered porous nickel plaque, which...
. Nickel–metal hydride (NIMH) battery tend to have longer lifespans than NiCd batteries (and their lifespans continue to increase as manufacturers experiment with new alloys) and, since cadmium
Cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, bluish-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Similar to zinc, it prefers oxidation state +2 in most of its compounds and similar to mercury it shows a low...
is toxic, NiMH batteries are less damaging to the environment.
1970s and 1990s - The lithium and lithium-ion batteries
Lithium is the metal with lowest density and with the greatest electrochemical potential and energy-to-weight ratio, so in theory it would be an ideal material with which to make batteries. Experimentation with lithium batteriesLithium battery
Lithium batteries are disposable batteries that have lithium metal or lithium compounds as an anode. Depending on the design and chemical compounds used, lithium cells can produce voltages from 1.5 V to about 3.7 V, over twice the voltage of an ordinary zinc–carbon battery or alkaline battery...
began in 1912 under G.N. Lewis, and in the 1970s the first lithium batteries were sold.
In the 1980s, an American chemist John B. Goodenough
John B. Goodenough
John Bannister Goodenough is an American professor and prominent solid-state physicist. He is currently a professor of mechanical engineering and materials science at the University of Texas at Austin...
led a research team at Sony that would produce the lithium ion battery
Lithium ion battery
A lithium-ion battery is a family of rechargeable battery types in which lithium ions move from the negative electrode to the positive electrode during discharge, and back when charging. Chemistry, performance, cost, and safety characteristics vary across LIB types...
, a rechargeable and more stable version of the lithium battery; the first ones were sold in 1991.
In 1996, the lithium ion polymer battery
Lithium ion polymer battery
Lithium-ion polymer batteries, polymer lithium ion, or more commonly lithium polymer batteries are rechargeable batteries...
was released. These batteries hold their electrolyte in a solid polymer composite instead of a liquid solvent, and the electrodes and separators are laminated to each other. The latter difference allows the battery to be encased in a flexible wrapping instead of a rigid metal casing, which means such batteries can be specifically shaped to fit a particular device. They also have a higher energy density than normal lithium ion batteries. These advantages have made it a choice battery for portable electronics such as mobile phones and PDAs
Personal digital assistant
A personal digital assistant , also known as a palmtop computer, or personal data assistant, is a mobile device that functions as a personal information manager. Current PDAs often have the ability to connect to the Internet...
, as they allow for more flexible and compact design.

