
Molecular tweezer
Encyclopedia
Hydrogen bond
A hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, such as nitrogen, oxygen or fluorine, that comes from another molecule or chemical group. The hydrogen must be covalently bonded to another electronegative atom to create the bond...
ing, metal coordination, hydrophobic forces
Hydrophobic effect
The hydrophobic effect is the observed tendency of nonpolar substances to aggregate in aqueous solution and exclude water molecules. The name, literally meaning "water-fearing," describes the segregation and apparent repulsion between water and nonpolar substances...
, van der Waals force
Van der Waals force
In physical chemistry, the van der Waals force , named after Dutch scientist Johannes Diderik van der Waals, is the sum of the attractive or repulsive forces between molecules other than those due to covalent bonds or to the electrostatic interaction of ions with one another or with neutral...
s, π-π interactions, and/or electrostatic effects. These complexes are a subset of macrocyclic
Macrocycle
A macrocycle is, as defined by IUPAC, "a cyclic macromolecule or a macromolecular cyclic portion of a molecule." In the chemical literature, organic chemists may consider any molecule containing a ring of nine or more atoms to be a macrocycle...
molecular receptors
Host-guest chemistry
In supramolecular chemistry, host-guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host-guest chemistry encompasses the idea of molecular recognition...
and their structure is that the two "arms" that bind the guest molecule between them are only connected at one end.
Examples
One example of molecular tweezers has been reported by LehnJean-Marie Lehn
Jean-Marie Lehn is a French chemist. He received the Nobel Prize together with Donald Cram and Charles Pedersen in 1987 for his work in Chemistry, particularly his synthesis of the cryptands...
and coworkers. This molecule is capable of binding aromatic guests.
The molecular tweezers are composed of two anthracene
Anthracene
Anthracene is a solid polycyclic aromatic hydrocarbon consisting of three fused benzene rings. It is a component of coal-tar. Anthracene is used in the production of the red dye alizarin and other dyes...
arms held at a distance that allows aromatic guests to gain π-π interactions from both.
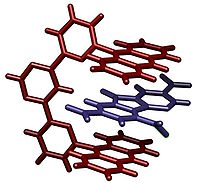
Another class of molecular tweezers is composed of two substituted porphyrin macrocycles tethered by an amide linker with a variable length. This example of a molecular tweezer shows the potential mobility of this class of molecules, as the orientation of the porphyrin planes which comprise the tweezer can be altered by the guest which is bound
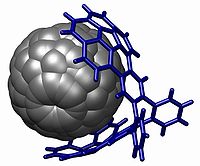
Yet another structure for molecular tweezers which specifically bind fullerenes is called a buckycatcher and has been reported. This molecular tweezer is composed of two concaved corannulene
Corannulene
Corannulene is a polycyclic aromatic hydrocarbon with chemical formula C20H10. The molecule consists of a cyclopentane ring fused with 5 benzene rings, so another name for it is [5]circulene. It is of scientific interest because it is a geodesic polyarene and can be considered a fragment of...
pincers that complement the surface of the convex fullerene
Fullerene
A fullerene is any molecule composed entirely of carbon, in the form of a hollow sphere, ellipsoid, or tube. Spherical fullerenes are also called buckyballs, and they resemble the balls used in association football. Cylindrical ones are called carbon nanotubes or buckytubes...
guest. An association constant (Ka) of 8600 M−1 between the host buckycatcher and a C60 fullerene was calculated using 1H NMR spectroscopy
NMR spectroscopy
Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy, is a research technique that exploits the magnetic properties of certain atomic nuclei to determine physical and chemical properties of atoms or the molecules in which they are contained...
.
The above examples show the potential reactivity and specificity of these molecules. The binding site between the planes of the tweezer can evolve to bind to an appropriate guest with resulting high association constants and consequent stability, depending on the configuration of the tweezer. That makes this overall class of macromolecule truly a synthetic molecular receptor.
External links
- Journal of Chemical EducationJournal of Chemical EducationThe Journal of Chemical Education is a monthly peer-reviewed academic journal available in both print and electronic versions. It is published by the Division of Chemical Education of the American Chemical Society and was established in 1924...
Featured Molecules December 2004: Nanoscale Molecular Tweezers and article - Crystalmaker molecular tweezers

