Nitrate
Encyclopedia
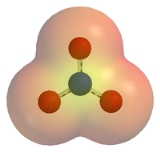
The nitrate ion is a polyatomic ion
with the molecular formula NO and a molecular mass
of 62.0049 g/mol. It is the conjugate base
of nitric acid
, consisting of one central nitrogen
atom
surrounded by three identically-bonded oxygen atoms in a trigonal planar
arrangement. The nitrate ion carries a formal charge
of -1. This results from a combination formal charge in which each of the three oxygens carries a − charge, whereas the nitrogen carries a +1 charge, all these adding up to formal charge of the polyatomic nitrate ion.
This arrangement is commonly used as an example of resonance
. Like the isoelectronic carbonate
ion, the nitrate ion can be represented by resonance structures:
Almost all inorganic nitrate salts are soluble
in water
at standard temperature and pressure. A common example of an inorganic nitrate salt is potassium nitrate
(saltpetre).
In organic chemistry
a nitrate (not to be confused with nitro
) is a functional group
with general chemical formula RONO2 where R stands for any organic residue. They are the esters of nitric acid and alcohols formed by nitroxylation. Examples are methyl nitrate formed by reaction of methanol
and nitric acid
, the nitrate of tartaric acid
, and the inaccurately-named nitroglycerin (which is actually an organic nitrate compound, not a nitro compound).
Like organic nitro compounds (see below) both organic and inorganic nitrates can be used as propellants and explosives. An example of the use of inorganic nitrate was classical gunpowder
. In all these uses the thermal decomposition of the nitrate yields molecular nitrogen
N2 gas plus considerable chemical energy, due to the high strength of the bond in molecular nitrogen. Especially in inorganic nitrate reactions, oxidation from the nitrate oxygens is also an important energy-releasing process.
(NO), the salts of nitrous acid
. Organic compound
s containing the nitro functional group
(which has the same formula and structure as the nitrate ion save that one of the O− atoms is replaced by the group) are known as nitro compound
s. Examples of familiar organic nitro compounds are nitromethane
and trinitrotoluene (TNT).
atoms in hemoglobin
from ferrous
iron (2+) to ferric
iron (3+), rendering it unable to carry oxygen. This process can lead to generalized lack of oxygen
in organ tissue and a dangerous condition called methemoglobinemia
. Although nitrite converts to ammonia
, if there is more nitrite than can be converted, the animal slowly suffers from a lack of oxygen.
being particularly especially vulnerable to methemoglobinemia due to nitrate metabolizing triglyceride
s present at higher concentrations than at other stages of development. Methemoglobinemia in infants is known as blue baby syndrome. Although nitrates in drinking water were once though to be a contributing factor, there are now significant scientific doubts as to whether there is a causal link. Blue baby syndrome is now thought to be the product of a number of factors, which can include any factor which causes gastric upset, such as diarrhoeal infection, protein intolerance, heavy metal toxicity etc., with nitrates playing a minor role. Nitrates, if a factor in a specific case, would most often be ingested by infants in high nitrate drinking water. However, nitrate exposure may also occur if eating, for instance, vegetables containing high levels of nitrate. Lettuce may contain elevated nitrate under growth conditions such as reduced sunlight, undersupply of the essential micronutrients molybdenum (Mo) and iron (Fe), or high concentrations of nitrate due to reduced assimilation of nitrate in the plant. High levels of nitrate fertilization also contribute to elevated levels of nitrate in the harvested plant.
Some adults can be more susceptible to the effects of nitrate than others. The methemoglobin reductase
enzyme
may be under-produced or absent in certain people that have an inherited mutation. Such individuals cannot break down methemoglobin as rapidly as those that do have the enzyme, leading to increased circulating levels of methemoglobin (the implication being that their blood is not as oxygen-rich). Those with insufficient stomach acid (including some vegetarians and vegans) may also be at risk. It is the increased consumption of green, leafy vegetables that typically accompany these types of diets may lead to increased nitrate intake. A wide variety of medical conditions, including food allergies, asthma, hepatitis, and gallstones may be linked with low stomach acid; these individuals may also be highly sensitive to the effects of nitrate.
For half a century, inorganic nitrate has been associated with negative health effects, but more recently, evidence of the contrary has mounted. In the 1990s, a research group at Karolinska Institutet demonstrated how the body can convert nitrate to NO, a molecule involved in many important bodily functions, such as blood pressure regulation, the immune defence and cell metabolism. These compounds can ameliorate mitochondrial dysfunction related diseases, The results, which are published in Cell Metabolism, are of sports-physiological interest, as they show that nitrate reduces oxygen consumption during physical exercise; however, they are also of potential significance to diseases involving mitochondrial dysfunction, such as diabetes and cardiovascular disease.,
Methemoglobinemia
can be treated with methylene blue
, which reduces ferric iron (3+) in affected blood cells back to ferrous iron (2+).
 In freshwater
In freshwater
or estuarine
systems close to land, nitrate can reach high levels that can potentially cause the death of fish. While nitrate is much less toxic than ammonia or nitrite, levels over 30 ppm of nitrate can inhibit growth, impair the immune system and cause stress in some aquatic species. However, in light of inherent problems with past protocols on acute nitrate toxicity experiments, the extent of nitrate toxicity has been the subject of recent debate.
In most cases of excess nitrate concentrations in aquatic systems, the primary source is surface runoff
from agricultural or landscape
d areas that have received excess nitrate fertilizer. This is called eutrophication
and can lead to algae blooms. As well as leading to water anoxia
and dead zones
, these blooms may cause other changes to ecosystem
function, favouring some groups of organisms over others. As a consequence, as nitrate forms a component of total dissolved solids
, they are widely used as an indicator of water quality
.
Nitrate also is a by-product of septic systems
. To be specific, it is a naturally occurring chemical that is left after the breakdown or decomposition of animal or human waste. Water quality may also be affected through ground water resources that have a high number of septic systems in a watershed. Septics leach down into ground water resources or aquifers and supply nearby bodies of water. Lakes that rely on ground water are often affected by nitrification through this process.
Nitrate in drinking water at levels above the national standard poses an immediate threat to young children. Excessive levels can result in a condition known as "blue baby syndrome
". If untreated, the condition can be fatal. Boiling water contaminated with nitrate increases the nitrate concentration and the potential risk.
Symptoms of nitrate poisoning include increased heart rate and respiration; in advanced cases blood and tissue may turn a blue or brown color. Feed can be tested for nitrate; treatment consists of supplementing or substituting existing supplies with lower nitrate material. Safe levels of nitrate for various types of livestock are as follows:
The values above are on a dry (moisture free) basis.
Ion
An ion is an atom or molecule in which the total number of electrons is not equal to the total number of protons, giving it a net positive or negative electrical charge. The name was given by physicist Michael Faraday for the substances that allow a current to pass between electrodes in a...
with the molecular formula NO and a molecular mass
Molecular mass
The molecular mass of a substance is the mass of one molecule of that substance, in unified atomic mass unit u...
of 62.0049 g/mol. It is the conjugate base
Conjugate acid
Within the Brønsted–Lowry acid-base theory , a conjugate acid is the acid member, HX, of a pair of two compounds that transform into each other by gain or loss of a proton. A conjugate acid can also be seen as the chemical substance that releases, or donates, a proton in the forward chemical...
of nitric acid
Nitric acid
Nitric acid , also known as aqua fortis and spirit of nitre, is a highly corrosive and toxic strong acid.Colorless when pure, older samples tend to acquire a yellow cast due to the accumulation of oxides of nitrogen. If the solution contains more than 86% nitric acid, it is referred to as fuming...
, consisting of one central nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
atom
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
surrounded by three identically-bonded oxygen atoms in a trigonal planar
Trigonal planar
In chemistry, trigonal planar is a molecular geometry model with one atom at the center and three atoms at the corners of a triangle, called peripheral atoms, all in one plane. In an ideal trigonal planar species, all three ligands are identical and all bond angles are 120°. Such species belong to...
arrangement. The nitrate ion carries a formal charge
Formal charge
In chemistry, a formal charge is the charge assigned to an atom in a molecule, assuming that electrons in a chemical bond are shared equally between atoms, regardless of relative electronegativity....
of -1. This results from a combination formal charge in which each of the three oxygens carries a − charge, whereas the nitrogen carries a +1 charge, all these adding up to formal charge of the polyatomic nitrate ion.
This arrangement is commonly used as an example of resonance
Resonance (chemistry)
In chemistry, resonance or mesomerism is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by one single Lewis formula...
. Like the isoelectronic carbonate
Carbonate
In chemistry, a carbonate is a salt of carbonic acid, characterized by the presence of the carbonate ion, . The name may also mean an ester of carbonic acid, an organic compound containing the carbonate group C2....
ion, the nitrate ion can be represented by resonance structures:
Almost all inorganic nitrate salts are soluble
Solubility
Solubility is the property of a solid, liquid, or gaseous chemical substance called solute to dissolve in a solid, liquid, or gaseous solvent to form a homogeneous solution of the solute in the solvent. The solubility of a substance fundamentally depends on the used solvent as well as on...
in water
Water
Water is a chemical substance with the chemical formula H2O. A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at ambient conditions, but it often co-exists on Earth with its solid state, ice, and gaseous state . Water also exists in a...
at standard temperature and pressure. A common example of an inorganic nitrate salt is potassium nitrate
Potassium nitrate
Potassium nitrate is a chemical compound with the formula KNO3. It is an ionic salt of potassium ions K+ and nitrate ions NO3−.It occurs as a mineral niter and is a natural solid source of nitrogen. Its common names include saltpetre , from medieval Latin sal petræ: "stone salt" or possibly "Salt...
(saltpetre).
In organic chemistry
Organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, composition, reactions, and preparation of carbon-based compounds, hydrocarbons, and their derivatives...
a nitrate (not to be confused with nitro
Nitro compound
Nitro compounds are organic compounds that contain one or more nitro functional groups . They are often highly explosive, especially when the compound contains more than one nitro group and is impure. The nitro group is one of the most common explosophores used globally...
) is a functional group
Functional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
with general chemical formula RONO2 where R stands for any organic residue. They are the esters of nitric acid and alcohols formed by nitroxylation. Examples are methyl nitrate formed by reaction of methanol
Methanol
Methanol, also known as methyl alcohol, wood alcohol, wood naphtha or wood spirits, is a chemical with the formula CH3OH . It is the simplest alcohol, and is a light, volatile, colorless, flammable liquid with a distinctive odor very similar to, but slightly sweeter than, ethanol...
and nitric acid
Nitric acid
Nitric acid , also known as aqua fortis and spirit of nitre, is a highly corrosive and toxic strong acid.Colorless when pure, older samples tend to acquire a yellow cast due to the accumulation of oxides of nitrogen. If the solution contains more than 86% nitric acid, it is referred to as fuming...
, the nitrate of tartaric acid
Tartaric acid
Tartaric acid is a white crystalline diprotic organic acid. It occurs naturally in many plants, particularly grapes, bananas, and tamarinds; is commonly combined with baking soda to function as a leavening agent in recipes, and is one of the main acids found in wine. It is added to other foods to...
, and the inaccurately-named nitroglycerin (which is actually an organic nitrate compound, not a nitro compound).
Like organic nitro compounds (see below) both organic and inorganic nitrates can be used as propellants and explosives. An example of the use of inorganic nitrate was classical gunpowder
Gunpowder
Gunpowder, also known since in the late 19th century as black powder, was the first chemical explosive and the only one known until the mid 1800s. It is a mixture of sulfur, charcoal, and potassium nitrate - with the sulfur and charcoal acting as fuels, while the saltpeter works as an oxidizer...
. In all these uses the thermal decomposition of the nitrate yields molecular nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
N2 gas plus considerable chemical energy, due to the high strength of the bond in molecular nitrogen. Especially in inorganic nitrate reactions, oxidation from the nitrate oxygens is also an important energy-releasing process.
Related materials
Nitrate should not be confused with nitriteNitrite
The nitrite ion has the chemical formula NO2−. The anion is symmetric with equal N-O bond lengths and a O-N-O bond angle of ca. 120°. On protonation the unstable weak acid nitrous acid is produced. Nitrite can be oxidised or reduced, with product somewhat dependent on the oxidizing/reducing agent...
(NO), the salts of nitrous acid
Nitrous acid
Nitrous acid is a weak and monobasic acid known only in solution and in the form of nitrite salts.Nitrous acid is used to make diazides from amines; this occurs by nucleophilic attack of the amine onto the nitrite, reprotonation by the surrounding solvent, and double-elimination of water...
. Organic compound
Organic compound
An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. For historical reasons discussed below, a few types of carbon-containing compounds such as carbides, carbonates, simple oxides of carbon, and cyanides, as well as the...
s containing the nitro functional group
Functional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
(which has the same formula and structure as the nitrate ion save that one of the O− atoms is replaced by the group) are known as nitro compound
Nitro compound
Nitro compounds are organic compounds that contain one or more nitro functional groups . They are often highly explosive, especially when the compound contains more than one nitro group and is impure. The nitro group is one of the most common explosophores used globally...
s. Examples of familiar organic nitro compounds are nitromethane
Nitromethane
Nitromethane is an organic compound with the chemical formula . It is the simplest organic nitro compound. It is a slightly viscous, highly polar liquid commonly used as a solvent in a variety of industrial applications such as in extractions, as a reaction medium, and as a cleaning solvent...
and trinitrotoluene (TNT).
Toxicity
Nitrate of itself is not poisonous to animals. However nitrate toxicosis can occur through enterohepatic metabolism of nitrate to nitrite being an intermediate. Nitrites oxidize the ironIron
Iron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust...
atoms in hemoglobin
Hemoglobin
Hemoglobin is the iron-containing oxygen-transport metalloprotein in the red blood cells of all vertebrates, with the exception of the fish family Channichthyidae, as well as the tissues of some invertebrates...
from ferrous
Ferrous
Ferrous , in chemistry, indicates a divalent iron compound , as opposed to ferric, which indicates a trivalent iron compound ....
iron (2+) to ferric
Ferric
Ferric refers to iron-containing materials or compounds. In chemistry the term is reserved for iron with an oxidation number of +3, also denoted iron or Fe3+. On the other hand, ferrous refers to iron with oxidation number of +2, denoted iron or Fe2+...
iron (3+), rendering it unable to carry oxygen. This process can lead to generalized lack of oxygen
Hypoxia (medical)
Hypoxia, or hypoxiation, is a pathological condition in which the body as a whole or a region of the body is deprived of adequate oxygen supply. Variations in arterial oxygen concentrations can be part of the normal physiology, for example, during strenuous physical exercise...
in organ tissue and a dangerous condition called methemoglobinemia
Methemoglobinemia
Methemoglobinemia is a disorder characterized by the presence of a higher than normal level of methemoglobin in the blood. Methemoglobin is an oxidized form of hemoglobin that has an increased affinity for oxygen, resulting in a reduced ability to release oxygen to tissues. The oxygen–hemoglobin...
. Although nitrite converts to ammonia
Ammonia
Ammonia is a compound of nitrogen and hydrogen with the formula . It is a colourless gas with a characteristic pungent odour. Ammonia contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to food and fertilizers. Ammonia, either directly or...
, if there is more nitrite than can be converted, the animal slowly suffers from a lack of oxygen.
Human health effects
Humans are subject to nitrate toxicity, with infantsInfant
A newborn or baby is the very young offspring of a human or other mammal. A newborn is an infant who is within hours, days, or up to a few weeks from birth. In medical contexts, newborn or neonate refers to an infant in the first 28 days after birth...
being particularly especially vulnerable to methemoglobinemia due to nitrate metabolizing triglyceride
Triglyceride
A triglyceride is an ester derived from glycerol and three fatty acids. There are many triglycerides, depending on the oil source, some are highly unsaturated, some less so....
s present at higher concentrations than at other stages of development. Methemoglobinemia in infants is known as blue baby syndrome. Although nitrates in drinking water were once though to be a contributing factor, there are now significant scientific doubts as to whether there is a causal link. Blue baby syndrome is now thought to be the product of a number of factors, which can include any factor which causes gastric upset, such as diarrhoeal infection, protein intolerance, heavy metal toxicity etc., with nitrates playing a minor role. Nitrates, if a factor in a specific case, would most often be ingested by infants in high nitrate drinking water. However, nitrate exposure may also occur if eating, for instance, vegetables containing high levels of nitrate. Lettuce may contain elevated nitrate under growth conditions such as reduced sunlight, undersupply of the essential micronutrients molybdenum (Mo) and iron (Fe), or high concentrations of nitrate due to reduced assimilation of nitrate in the plant. High levels of nitrate fertilization also contribute to elevated levels of nitrate in the harvested plant.
Some adults can be more susceptible to the effects of nitrate than others. The methemoglobin reductase
Cytochrome b5 reductase
Cytochrome-b5 reductase is a NADH-dependent enzyme that converts methemoglobin to hemoglobin...
enzyme
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
may be under-produced or absent in certain people that have an inherited mutation. Such individuals cannot break down methemoglobin as rapidly as those that do have the enzyme, leading to increased circulating levels of methemoglobin (the implication being that their blood is not as oxygen-rich). Those with insufficient stomach acid (including some vegetarians and vegans) may also be at risk. It is the increased consumption of green, leafy vegetables that typically accompany these types of diets may lead to increased nitrate intake. A wide variety of medical conditions, including food allergies, asthma, hepatitis, and gallstones may be linked with low stomach acid; these individuals may also be highly sensitive to the effects of nitrate.
For half a century, inorganic nitrate has been associated with negative health effects, but more recently, evidence of the contrary has mounted. In the 1990s, a research group at Karolinska Institutet demonstrated how the body can convert nitrate to NO, a molecule involved in many important bodily functions, such as blood pressure regulation, the immune defence and cell metabolism. These compounds can ameliorate mitochondrial dysfunction related diseases, The results, which are published in Cell Metabolism, are of sports-physiological interest, as they show that nitrate reduces oxygen consumption during physical exercise; however, they are also of potential significance to diseases involving mitochondrial dysfunction, such as diabetes and cardiovascular disease.,
Methemoglobinemia
Methemoglobinemia
Methemoglobinemia is a disorder characterized by the presence of a higher than normal level of methemoglobin in the blood. Methemoglobin is an oxidized form of hemoglobin that has an increased affinity for oxygen, resulting in a reduced ability to release oxygen to tissues. The oxygen–hemoglobin...
can be treated with methylene blue
Methylene blue
Methylene blue is a heterocyclic aromatic chemical compound with the molecular formula C16H18N3SCl. It has many uses in a range of different fields, such as biology and chemistry. At room temperature it appears as a solid, odorless, dark green powder, that yields a blue solution when dissolved in...
, which reduces ferric iron (3+) in affected blood cells back to ferrous iron (2+).
Marine toxicity

Freshwater
Fresh water is naturally occurring water on the Earth's surface in ice sheets, ice caps, glaciers, bogs, ponds, lakes, rivers and streams, and underground as groundwater in aquifers and underground streams. Fresh water is generally characterized by having low concentrations of dissolved salts and...
or estuarine
Estuary
An estuary is a partly enclosed coastal body of water with one or more rivers or streams flowing into it, and with a free connection to the open sea....
systems close to land, nitrate can reach high levels that can potentially cause the death of fish. While nitrate is much less toxic than ammonia or nitrite, levels over 30 ppm of nitrate can inhibit growth, impair the immune system and cause stress in some aquatic species. However, in light of inherent problems with past protocols on acute nitrate toxicity experiments, the extent of nitrate toxicity has been the subject of recent debate.
In most cases of excess nitrate concentrations in aquatic systems, the primary source is surface runoff
Surface runoff
Surface runoff is the water flow that occurs when soil is infiltrated to full capacity and excess water from rain, meltwater, or other sources flows over the land. This is a major component of the water cycle. Runoff that occurs on surfaces before reaching a channel is also called a nonpoint source...
from agricultural or landscape
Landscape
Landscape comprises the visible features of an area of land, including the physical elements of landforms such as mountains, hills, water bodies such as rivers, lakes, ponds and the sea, living elements of land cover including indigenous vegetation, human elements including different forms of...
d areas that have received excess nitrate fertilizer. This is called eutrophication
Eutrophication
Eutrophication or more precisely hypertrophication, is the movement of a body of water′s trophic status in the direction of increasing plant biomass, by the addition of artificial or natural substances, such as nitrates and phosphates, through fertilizers or sewage, to an aquatic system...
and can lead to algae blooms. As well as leading to water anoxia
Hypoxia (environmental)
Hypoxia, or oxygen depletion, is a phenomenon that occurs in aquatic environments as dissolved oxygen becomes reduced in concentration to a point where it becomes detrimental to aquatic organisms living in the system...
and dead zones
Dead zone (ecology)
Dead zones are hypoxic areas in the world's oceans, the observed incidences of which have been increasing since oceanographers began noting them in the 1970s. These occur near inhabited coastlines, where aquatic life is most concentrated...
, these blooms may cause other changes to ecosystem
Ecosystem
An ecosystem is a biological environment consisting of all the organisms living in a particular area, as well as all the nonliving , physical components of the environment with which the organisms interact, such as air, soil, water and sunlight....
function, favouring some groups of organisms over others. As a consequence, as nitrate forms a component of total dissolved solids
Total dissolved solids
Total Dissolved Solids is a measure of the combined content of all inorganic and organic substances contained in a liquid in: molecular, ionized or micro-granular suspended form. Generally the operational definition is that the solids must be small enough to survive filtration through a sieve...
, they are widely used as an indicator of water quality
Water quality
Water quality is the physical, chemical and biological characteristics of water. It is a measure of the condition of water relative to the requirements of one or more biotic species and or to any human need or purpose. It is most frequently used by reference to a set of standards against which...
.
Nitrate also is a by-product of septic systems
Septic tank
A septic tank is a key component of the septic system, a small-scale sewage treatment system common in areas with no connection to main sewage pipes provided by local governments or private corporations...
. To be specific, it is a naturally occurring chemical that is left after the breakdown or decomposition of animal or human waste. Water quality may also be affected through ground water resources that have a high number of septic systems in a watershed. Septics leach down into ground water resources or aquifers and supply nearby bodies of water. Lakes that rely on ground water are often affected by nitrification through this process.
Nitrate in drinking water at levels above the national standard poses an immediate threat to young children. Excessive levels can result in a condition known as "blue baby syndrome
Blue baby syndrome
Blue baby syndrome is a layman's term used to describe newborns with cyanotic heart lesions, such as* Persistent Truncus Arteriosus* Transposition of the great vessels* Tricuspid atresia* Tetralogy of Fallot...
". If untreated, the condition can be fatal. Boiling water contaminated with nitrate increases the nitrate concentration and the potential risk.
Nitrate toxicity in livestock
Nitrate poisoning is a concern for cattle producers primarily, but also for other ruminant growers. Nitrate is naturally consumed by growing plants; however when growth is interrupted, the roots of plants will continue to provide nitrate from soil, and unless new plant growth occurs, the nitrate will accumulate. Thus, harvesting after nitrate buildup results in a crop that is high in nitrates. Factors that can cause nitrate buildup in plants are frost, hail damage, drought, hot dry winds, pesticide damage, herbicide damage and any other plant stunting environmental causes.Symptoms of nitrate poisoning include increased heart rate and respiration; in advanced cases blood and tissue may turn a blue or brown color. Feed can be tested for nitrate; treatment consists of supplementing or substituting existing supplies with lower nitrate material. Safe levels of nitrate for various types of livestock are as follows:
| Category | %NO3 | %NO3-N | %KNO3 | Effects |
|---|---|---|---|---|
| 1 | <0.5 | <0.12 | <0.81 | Generally safe for beef cattle and sheep |
| 2 | 0.5-1.0 | 0.12-0.23 | 0.81-1.63 | Caution - some subclinical symptoms may appear in pregnant horses, sheep and beef cattle |
| 3 | 1.0 | 0.23 | 1.63 | High nitrate problems - death losses and abortions can occur in beef cattle and sheep |
| 4 | <1.23 | <0.28 | <2.00 | Maximum safe level for horses. Do not feed high nitrate forages to pregnant mares |
The values above are on a dry (moisture free) basis.

