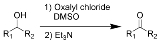
Swern oxidation
Encyclopedia
The Swern oxidation, named after Daniel Swern
, is a chemical reaction
whereby a primary or secondary alcohol
is oxidized
to an aldehyde
or ketone
using oxalyl chloride
, dimethyl sulfoxide (DMSO)
and an organic base, such as triethylamine
. The reaction is known for its mild character and wide tolerance of functional group
s.
 The by-products are dimethyl sulfide
The by-products are dimethyl sulfide
(Me2S), carbon monoxide
(CO), carbon dioxide
(CO2) and — when triethylamine
is used as base — triethylammonium chloride (Et3NHCl). Two of the by-products, dimethyl sulfide
and carbon monoxide
, are very toxic volatile compounds, so the reaction and the work-up needs to be performed in a fume hood. Dimethyl sulfide
is a volatile liquid (B.P. 37 °C) with an unpleasant odour.
Several reviews have been published.
(DMSO), 1a, formally as resonance
contributor 1b, with oxalyl chloride
, 2. The first intermediate, 3, quickly decomposes giving off CO2
and CO
and producing dimethylchlorosulfonium chloride, 4.
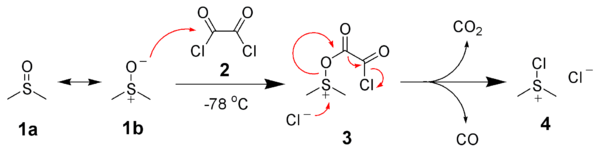 After addition of the alcohol 5, the dimethylchlorosulfonium chloride 4 reacts with the alcohol to give the key alkoxysulfonium ion intermediate, 6. The addition of at least 2 equivalents of base — typically triethylamine
After addition of the alcohol 5, the dimethylchlorosulfonium chloride 4 reacts with the alcohol to give the key alkoxysulfonium ion intermediate, 6. The addition of at least 2 equivalents of base — typically triethylamine
— will deprotonate
the alkoxysulfonium ion to give the sulfur ylide
7. In a five-membered ring transition state
, the sulfur ylide 7 decomposes to give dimethyl sulfide
and the desired ketone (or aldehyde) 8.
agent, the reaction must be kept colder than −60 °C to avoid side reactions. With cyanuric chloride
or trifluoroacetic anhydride
instead of oxalyl chloride, the reaction can be warmed to −30 °C without side reactions. Other methods for the activation of DMSO to initiate the formation of the key intermediate 6 are the use of carbodiimides (Pfitzner–Moffatt oxidation) and pyridine-sulfur trioxide complex (Parikh-Doering oxidation
). The intermediate 4 can also be prepared from dimethyl sulfide and N-chlorosuccinimide
(the Corey-Kim oxidation
).
In some cases, the use of triethylamine
as the base can lead to epimerisation at the carbon alpha to the newly formed carbonyl. Using the bulkier base diisopropylethylamine (iPr2NEt, Hünig's base) can mitigate this side reaction.
, a byproduct of the Swern oxidation, is one of the most foul odors known in organic chemistry. Human olfactory glands can detect this compound in concentrations as low as parts per billion. A simple remedy for this problem is to rinse used glassware with bleach
(usually containing sodium hypochlorite), which will oxidize the dimethyl sulfide, eliminating the smell.
The reaction conditions allow oxidation of acid-sensitive compounds, which might decompose under the acidic conditions of a traditional method such as Jones oxidation
. For example, in Thompson & Heathcock's synthesis of the sesquiterpene
isovelleral, the final step uses the Swern protocol, avoiding rearrangement
of the acid-sensitive cyclopropanemethanol moiety.
Daniel Swern
Daniel Swern was an American chemist who discovered the Swern oxidation.-References:*...
, is a chemical reaction
Chemical reaction
A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Chemical reactions can be either spontaneous, requiring no input of energy, or non-spontaneous, typically following the input of some type of energy, such as heat, light or electricity...
whereby a primary or secondary alcohol
Alcohol
In chemistry, an alcohol is an organic compound in which the hydroxy functional group is bound to a carbon atom. In particular, this carbon center should be saturated, having single bonds to three other atoms....
is oxidized
Redox
Redox reactions describe all chemical reactions in which atoms have their oxidation state changed....
to an aldehyde
Aldehyde
An aldehyde is an organic compound containing a formyl group. This functional group, with the structure R-CHO, consists of a carbonyl center bonded to hydrogen and an R group....
or ketone
Ketone
In organic chemistry, a ketone is an organic compound with the structure RCR', where R and R' can be a variety of atoms and groups of atoms. It features a carbonyl group bonded to two other carbon atoms. Many ketones are known and many are of great importance in industry and in biology...
using oxalyl chloride
Oxalyl chloride
Oxalyl chloride or ethanedioyl dichloride is a chemical compound with the formula 2. This colourless, sharp-smelling liquid, the diacid chloride of oxalic acid, is a useful reagent in organic synthesis...
, dimethyl sulfoxide (DMSO)
Dimethyl sulfoxide
Dimethyl sulfoxide is an organosulfur compound with the formula 2SO. This colorless liquid is an important polar aprotic solvent that dissolves both polar and nonpolar compounds and is miscible in a wide range of organic solvents as well as water...
and an organic base, such as triethylamine
Triethylamine
Triethylamine is the chemical compound with the formula N3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine, for which TEA is also a common abbreviation....
. The reaction is known for its mild character and wide tolerance of functional group
Functional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
s.

Dimethyl sulfide
Dimethyl sulfide or methylthiomethane is an organosulfur compound with the formula 2S. Dimethyl sulfide is a water-insoluble flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize,...
(Me2S), carbon monoxide
Carbon monoxide
Carbon monoxide , also called carbonous oxide, is a colorless, odorless, and tasteless gas that is slightly lighter than air. It is highly toxic to humans and animals in higher quantities, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal...
(CO), carbon dioxide
Carbon dioxide
Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
(CO2) and — when triethylamine
Triethylamine
Triethylamine is the chemical compound with the formula N3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine, for which TEA is also a common abbreviation....
is used as base — triethylammonium chloride (Et3NHCl). Two of the by-products, dimethyl sulfide
Dimethyl sulfide
Dimethyl sulfide or methylthiomethane is an organosulfur compound with the formula 2S. Dimethyl sulfide is a water-insoluble flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize,...
and carbon monoxide
Carbon monoxide
Carbon monoxide , also called carbonous oxide, is a colorless, odorless, and tasteless gas that is slightly lighter than air. It is highly toxic to humans and animals in higher quantities, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal...
, are very toxic volatile compounds, so the reaction and the work-up needs to be performed in a fume hood. Dimethyl sulfide
Dimethyl sulfide
Dimethyl sulfide or methylthiomethane is an organosulfur compound with the formula 2S. Dimethyl sulfide is a water-insoluble flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize,...
is a volatile liquid (B.P. 37 °C) with an unpleasant odour.
Several reviews have been published.
Mechanism
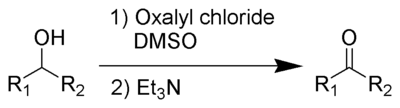
The first step of the Swern oxidation is the low-temperature reaction of dimethyl sulfoxideDimethyl sulfoxide
Dimethyl sulfoxide is an organosulfur compound with the formula 2SO. This colorless liquid is an important polar aprotic solvent that dissolves both polar and nonpolar compounds and is miscible in a wide range of organic solvents as well as water...
(DMSO), 1a, formally as resonance
Resonance (chemistry)
In chemistry, resonance or mesomerism is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by one single Lewis formula...
contributor 1b, with oxalyl chloride
Oxalyl chloride
Oxalyl chloride or ethanedioyl dichloride is a chemical compound with the formula 2. This colourless, sharp-smelling liquid, the diacid chloride of oxalic acid, is a useful reagent in organic synthesis...
, 2. The first intermediate, 3, quickly decomposes giving off CO2
Carbon dioxide
Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
and CO
Carbon monoxide
Carbon monoxide , also called carbonous oxide, is a colorless, odorless, and tasteless gas that is slightly lighter than air. It is highly toxic to humans and animals in higher quantities, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal...
and producing dimethylchlorosulfonium chloride, 4.

Triethylamine
Triethylamine is the chemical compound with the formula N3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine, for which TEA is also a common abbreviation....
— will deprotonate
Deprotonation
Deprotonation is the removal of a proton from a molecule, forming the conjugate base.The relative ability of a molecule to give up a proton is measured by its pKa value. A low pKa value indicates that the compound is acidic and will easily give up its proton to a base...
the alkoxysulfonium ion to give the sulfur ylide
Ylide
An ylide or ylid is a neutral dipolar molecule containing a formally negatively charged atom directly attached to a hetero atom with a formal positive charge , and in which both atoms have full octets of electrons. Ylides are thus 1,2-dipolar compounds...
7. In a five-membered ring transition state
Transition state
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest energy along this reaction coordinate. At this point, assuming a perfectly irreversible reaction, colliding reactant molecules will always...
, the sulfur ylide 7 decomposes to give dimethyl sulfide
Dimethyl sulfide
Dimethyl sulfide or methylthiomethane is an organosulfur compound with the formula 2S. Dimethyl sulfide is a water-insoluble flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize,...
and the desired ketone (or aldehyde) 8.

Variations
When using oxalyl chloride as the dehydrationDehydration
In physiology and medicine, dehydration is defined as the excessive loss of body fluid. It is literally the removal of water from an object; however, in physiological terms, it entails a deficiency of fluid within an organism...
agent, the reaction must be kept colder than −60 °C to avoid side reactions. With cyanuric chloride
Cyanuric chloride
Cyanuric chloride is the inorganic compound with the formula 3. This colorless solid is the chlorinated derivative of 1,3,5-triazine. It is the trimer of cyanogen chloride...
or trifluoroacetic anhydride
Trifluoroacetic anhydride
Trifluoroacetic anhydride is the acid anhydride of trifluoroacetic acid. In particular, trifluoroacetic anhydride is the perfluorinated derivative of acetic anhydride. Like many acid anhydrides, it may be used to introduce the corresponding trifluoroacetyl group. The corresponding trifluoroacetyl...
instead of oxalyl chloride, the reaction can be warmed to −30 °C without side reactions. Other methods for the activation of DMSO to initiate the formation of the key intermediate 6 are the use of carbodiimides (Pfitzner–Moffatt oxidation) and pyridine-sulfur trioxide complex (Parikh-Doering oxidation
Parikh-Doering oxidation
The Parikh-Doering oxidation is an oxidation reaction that transforms primary and secondary alcohols into aldehydes and ketones, respectively. The procedure uses DMSO as the oxidant, activated by sulfur trioxide-pyridine complex in the presence of triethylamine base:The procedure can be run at...
). The intermediate 4 can also be prepared from dimethyl sulfide and N-chlorosuccinimide
N-Chlorosuccinimide
N-Chlorosuccinimide is used for chlorinations and as a mild oxidant.N-Iodosuccinimide , the iodine analog of N-chlorosuccinimide, and N-bromosuccinimide , the bromine analog, are used for similar applications.-External links:...
(the Corey-Kim oxidation
Corey-Kim oxidation
The Corey–Kim oxidation is an oxidation reaction used to synthesise aldehydes and ketones from primary and secondary alcohols. It is named for American chemist and Nobelist Elias James Corey and Korean-American chemist Choung Un Kim....
).
In some cases, the use of triethylamine
Triethylamine
Triethylamine is the chemical compound with the formula N3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine, for which TEA is also a common abbreviation....
as the base can lead to epimerisation at the carbon alpha to the newly formed carbonyl. Using the bulkier base diisopropylethylamine (iPr2NEt, Hünig's base) can mitigate this side reaction.
Considerations
Dimethyl sulfideDimethyl sulfide
Dimethyl sulfide or methylthiomethane is an organosulfur compound with the formula 2S. Dimethyl sulfide is a water-insoluble flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize,...
, a byproduct of the Swern oxidation, is one of the most foul odors known in organic chemistry. Human olfactory glands can detect this compound in concentrations as low as parts per billion. A simple remedy for this problem is to rinse used glassware with bleach
Bleach
Bleach refers to a number of chemicals that remove color, whiten, or disinfect, often via oxidation. Common chemical bleaches include household chlorine bleach , lye, oxygen bleach , and bleaching powder...
(usually containing sodium hypochlorite), which will oxidize the dimethyl sulfide, eliminating the smell.
The reaction conditions allow oxidation of acid-sensitive compounds, which might decompose under the acidic conditions of a traditional method such as Jones oxidation
Jones oxidation
The Jones oxidation, is an organic reaction for the oxidation of primary and secondary alcohols to carboxylic acids and ketones, respectively. It is named after its discoverer, Sir Ewart Jones....
. For example, in Thompson & Heathcock's synthesis of the sesquiterpene
Sesquiterpene
Sesquiterpenes are a class of terpenes that consist of three isoprene units and have the molecular formula C15H24. Like monoterpenes, sesquiterpenes may be acyclic or contain rings, including many unique combinations...
isovelleral, the final step uses the Swern protocol, avoiding rearrangement
Rearrangement reaction
A rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a molecule is rearranged to give a structural isomer of the original molecule. Often a substituent moves from one atom to another atom in the same molecule...
of the acid-sensitive cyclopropanemethanol moiety.
See also
- Parikh-Doering oxidationParikh-Doering oxidationThe Parikh-Doering oxidation is an oxidation reaction that transforms primary and secondary alcohols into aldehydes and ketones, respectively. The procedure uses DMSO as the oxidant, activated by sulfur trioxide-pyridine complex in the presence of triethylamine base:The procedure can be run at...
- Dess–Martin periodinane
- Corey–Kim oxidation
- Pfitzner–Moffatt oxidation

