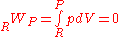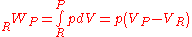Adiabatic flame temperature
Encyclopedia
In the study of combustion
, there are two types of adiabatic flame temperature depending on how the process is completed, constant volume and constant pressure, describing the temperature the combustion products theoretically reach if no energy is lost to the outside environment.
The constant volume adiabatic flame temperature is the temperature
that results from a complete combustion
process that occurs without any work
, heat transfer
or changes in kinetic
or potential energy
. The constant pressure adiabatic
flame temperature is the temperature that results from a complete combustion process that occurs without any heat transfer or changes in kinetic or potential energy. Its temperature is lower than the constant volume process because some of the energy is utilized to change the volume of the system (i.e., generate work).
It is a commonly misunderstood that the adiabatic flame temperature is the maximum temperature that can be achieved for given reactants because any heat transfer from the reacting substances and/or any incomplete combustion would tend to lower the temperature of the products. However, since the assumptions inherent in the adiabatic flame temperature assume chemical equilibrium, states in thermal equilibrium but not chemical equilibrium are not constrained by this limit. In fact, several fuel rich acetylene and methane flames have been found to exceed their adiabatic flame temperatures by hundreds of degrees.
 In daily life, the vast majority of flames one encounters are those of organic compound
In daily life, the vast majority of flames one encounters are those of organic compound
s including wood
, wax
, fat
, common plastic
s, propane
, and gasoline
. The constant-pressure adiabatic flame temperature of such substances in air is in a relatively narrow range around 1950 °C. This is because, in terms of stoichiometry
, the combustion of an organic compound with n carbons involves breaking roughly 2n C–H bonds, n C–C bonds, and 1.5n O2 bonds to form roughly n CO2 molecules and n H2O molecules.
Because most combustion processes that happen naturally occur in the open air, there is nothing that confines the gas to a particular volume like the cylinder in an engine. As a result, these substances will burn at a constant pressure allowing the gas to expand during the process.
 ).
).
Note this is a theoretical flame temperature produced by a flame that loses no heat (i.e. closest will be the hottest part of a flame) where the combustion reaction is quickest. And where complete combustion occurs, so the closest flame temperature to this will be a non-smokey, commonly bluish flame.
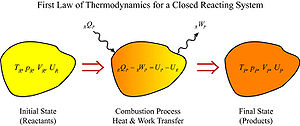 From the first law of thermodynamics
From the first law of thermodynamics
for a closed reacting system we have,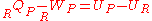
where,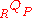 and
and 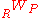 are the heat and work transferred during the process respectively, and
are the heat and work transferred during the process respectively, and 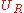 and
and 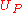 are the internal energy of the reactants and products respectively.
are the internal energy of the reactants and products respectively.
In the constant volume adiabatic flame temperature case, the volume of the system is held constant hence there is no work occurring,
and there is no heat transfer because the process is defined to be adiabatic: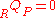 . As a result, the internal energy of the products is equal to the internal energy of the reactants:
. As a result, the internal energy of the products is equal to the internal energy of the reactants:  .
.
Because this is a closed system, the mass of the products and reactants is constant and the first law can be written on a mass basis,
 In the constant pressure adiabatic flame temperature case, the pressure of the system is held constant which results in the following equation for the work,
In the constant pressure adiabatic flame temperature case, the pressure of the system is held constant which results in the following equation for the work,
Again there is no heat transfer occurring because the process is defined to be adiabatic: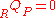 . From the first law, we find that,
. From the first law, we find that,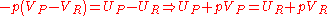
Recalling the definition of enthalpy we recover: . Because this is a closed system, the mass of the products and reactants is constant and the first law can be written on a mass basis,
. Because this is a closed system, the mass of the products and reactants is constant and the first law can be written on a mass basis,
We see that the adiabatic flame temperature of the constant pressure process is lower than that of the constant volume process. This is because some of the energy released during combustion goes into changing the volume of the control system. One analogy that is commonly made between the two processes is through combustion in an internal combustion engine. For the constant volume adiabatic process, combustion is thought to occur instantaneously when the piston reaches the top of its apex (Otto cycle
or constant volume cycle). For the constant pressure adiabatic process, while combustion is occurring the piston is moving in order to keep the pressure constant (Diesel cycle
or constant pressure cycle).
 If we make the assumption that combustion goes to completion (i.e.
If we make the assumption that combustion goes to completion (i.e.  and
and 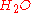 ), we can calculate the adiabatic flame temperature by hand either at stoichiometric conditions or lean of stoichiometry (excess air). This is because there are enough variables and molar equations to balance the left and right hand sides,
), we can calculate the adiabatic flame temperature by hand either at stoichiometric conditions or lean of stoichiometry (excess air). This is because there are enough variables and molar equations to balance the left and right hand sides,
Rich of stoichiometry there are not enough variables because combustion cannot go to completion with at least and
and  needed for the molar balance (these are the most common incomplete products of combustion),
needed for the molar balance (these are the most common incomplete products of combustion),

However, if we include the Water gas shift reaction
,
and use the equilibrium constant for this reaction, we will have enough variables to complete the calculation.
Different fuels with different levels of energy and molar constituents will have different adiabatic flame temperatures.
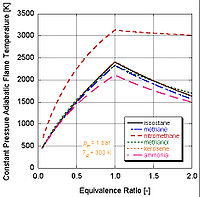
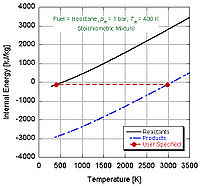
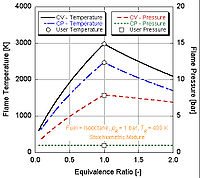
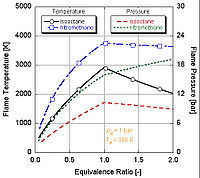 We can see by the following figure why nitromethane
We can see by the following figure why nitromethane
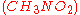 is often used as a power boost for cars. Since it contains two moles of oxygen in its molecular makeup, it can burn much hotter because it provides its own oxidant along with fuel. This in turn allows it to build-up more pressure during a constant volume process. The higher the pressure, the more force upon the piston creating more work and more power in the engine. It is interesting to note that it stays relatively hot rich of stoichiometry because it contains its own oxidant. However, continual running of an engine on nitromethane will eventually melt the piston and/or cylinder because of this higher temperature.
is often used as a power boost for cars. Since it contains two moles of oxygen in its molecular makeup, it can burn much hotter because it provides its own oxidant along with fuel. This in turn allows it to build-up more pressure during a constant volume process. The higher the pressure, the more force upon the piston creating more work and more power in the engine. It is interesting to note that it stays relatively hot rich of stoichiometry because it contains its own oxidant. However, continual running of an engine on nitromethane will eventually melt the piston and/or cylinder because of this higher temperature.
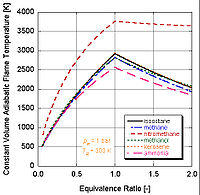
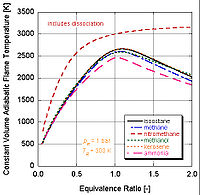 In real world applications, complete combustion does not typically occur. Chemistry dictates that dissociation
In real world applications, complete combustion does not typically occur. Chemistry dictates that dissociation
and kinetics
will change the relative constituents of the products. There are a number of programs available that can calculate the adiabatic flame temperature taking into account dissociation through equilibrium constants (Stanjan, NASA CEA, AFTP). The following figure illustrates that the effects of dissociation tend to lower the adiabatic flame temperature. This result can be explained through Le Chatelier's principle
.
Combustion
Combustion or burning is the sequence of exothermic chemical reactions between a fuel and an oxidant accompanied by the production of heat and conversion of chemical species. The release of heat can result in the production of light in the form of either glowing or a flame...
, there are two types of adiabatic flame temperature depending on how the process is completed, constant volume and constant pressure, describing the temperature the combustion products theoretically reach if no energy is lost to the outside environment.
The constant volume adiabatic flame temperature is the temperature
Temperature
Temperature is a physical property of matter that quantitatively expresses the common notions of hot and cold. Objects of low temperature are cold, while various degrees of higher temperatures are referred to as warm or hot...
that results from a complete combustion
Combustion
Combustion or burning is the sequence of exothermic chemical reactions between a fuel and an oxidant accompanied by the production of heat and conversion of chemical species. The release of heat can result in the production of light in the form of either glowing or a flame...
process that occurs without any work
Work (thermodynamics)
In thermodynamics, work performed by a system is the energy transferred to another system that is measured by the external generalized mechanical constraints on the system. As such, thermodynamic work is a generalization of the concept of mechanical work in mechanics. Thermodynamic work encompasses...
, heat transfer
Heat transfer
Heat transfer is a discipline of thermal engineering that concerns the exchange of thermal energy from one physical system to another. Heat transfer is classified into various mechanisms, such as heat conduction, convection, thermal radiation, and phase-change transfer...
or changes in kinetic
Kinetic energy
The kinetic energy of an object is the energy which it possesses due to its motion.It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acceleration, the body maintains this kinetic energy unless its speed changes...
or potential energy
Potential energy
In physics, potential energy is the energy stored in a body or in a system due to its position in a force field or due to its configuration. The SI unit of measure for energy and work is the Joule...
. The constant pressure adiabatic
Adiabatic process
In thermodynamics, an adiabatic process or an isocaloric process is a thermodynamic process in which the net heat transfer to or from the working fluid is zero. Such a process can occur if the container of the system has thermally-insulated walls or the process happens in an extremely short time,...
flame temperature is the temperature that results from a complete combustion process that occurs without any heat transfer or changes in kinetic or potential energy. Its temperature is lower than the constant volume process because some of the energy is utilized to change the volume of the system (i.e., generate work).
It is a commonly misunderstood that the adiabatic flame temperature is the maximum temperature that can be achieved for given reactants because any heat transfer from the reacting substances and/or any incomplete combustion would tend to lower the temperature of the products. However, since the assumptions inherent in the adiabatic flame temperature assume chemical equilibrium, states in thermal equilibrium but not chemical equilibrium are not constrained by this limit. In fact, several fuel rich acetylene and methane flames have been found to exceed their adiabatic flame temperatures by hundreds of degrees.
Common flames

Organic compound
An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. For historical reasons discussed below, a few types of carbon-containing compounds such as carbides, carbonates, simple oxides of carbon, and cyanides, as well as the...
s including wood
Wood
Wood is a hard, fibrous tissue found in many trees. It has been used for hundreds of thousands of years for both fuel and as a construction material. It is an organic material, a natural composite of cellulose fibers embedded in a matrix of lignin which resists compression...
, wax
Wax
thumb|right|[[Cetyl palmitate]], a typical wax ester.Wax refers to a class of chemical compounds that are plastic near ambient temperatures. Characteristically, they melt above 45 °C to give a low viscosity liquid. Waxes are insoluble in water but soluble in organic, nonpolar solvents...
, fat
Fat
Fats consist of a wide group of compounds that are generally soluble in organic solvents and generally insoluble in water. Chemically, fats are triglycerides, triesters of glycerol and any of several fatty acids. Fats may be either solid or liquid at room temperature, depending on their structure...
, common plastic
Plastic
A plastic material is any of a wide range of synthetic or semi-synthetic organic solids used in the manufacture of industrial products. Plastics are typically polymers of high molecular mass, and may contain other substances to improve performance and/or reduce production costs...
s, propane
Propane
Propane is a three-carbon alkane with the molecular formula , normally a gas, but compressible to a transportable liquid. A by-product of natural gas processing and petroleum refining, it is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves, and residential central...
, and gasoline
Gasoline
Gasoline , or petrol , is a toxic, translucent, petroleum-derived liquid that is primarily used as a fuel in internal combustion engines. It consists mostly of organic compounds obtained by the fractional distillation of petroleum, enhanced with a variety of additives. Some gasolines also contain...
. The constant-pressure adiabatic flame temperature of such substances in air is in a relatively narrow range around 1950 °C. This is because, in terms of stoichiometry
Stoichiometry
Stoichiometry is a branch of chemistry that deals with the relative quantities of reactants and products in chemical reactions. In a balanced chemical reaction, the relations among quantities of reactants and products typically form a ratio of whole numbers...
, the combustion of an organic compound with n carbons involves breaking roughly 2n C–H bonds, n C–C bonds, and 1.5n O2 bonds to form roughly n CO2 molecules and n H2O molecules.
Because most combustion processes that happen naturally occur in the open air, there is nothing that confines the gas to a particular volume like the cylinder in an engine. As a result, these substances will burn at a constant pressure allowing the gas to expand during the process.
Common flame temperatures
Assuming initial atmospheric conditions (1 bar and 20 °C), the following table list the adiabatic flame temperature for various gases under constant pressure conditions. The temperatures mentioned here are for a stoichiometric fuel-oxidizer mixture (i.e. equivalence ratio ).
).Note this is a theoretical flame temperature produced by a flame that loses no heat (i.e. closest will be the hottest part of a flame) where the combustion reaction is quickest. And where complete combustion occurs, so the closest flame temperature to this will be a non-smokey, commonly bluish flame.
| Fuel | Oxidizer | 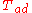 (°C) (°C) | 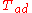 (°F) (°F) |
|---|---|---|---|
| Acetylene Acetylene Acetylene is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in pure form and thus is usually handled as a solution.As an alkyne, acetylene is unsaturated because... |
air | 2500 | 4532 |
| Acetylene Acetylene Acetylene is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in pure form and thus is usually handled as a solution.As an alkyne, acetylene is unsaturated because... |
Oxygen Oxygen Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition... |
3480 | 6296 |
| Butane Butane Butane is a gas with the formula C4H10 that is an alkane with four carbon atoms. The term may refer to any of two structural isomers, or to a mixture of them: in the IUPAC nomenclature, however, butane refers only to the unbranched n-butane isomer; the other one being called "methylpropane" or... |
air | 1970 | 3578 |
| Cyanogen Cyanogen Cyanogen is the chemical compound with the formula 2. It is a colorless, toxic gas with a pungent odor.The molecule is a pseudohalogen. Cyanogen molecules consist of two CN groups — analogous to diatomic halogen molecules, such as Cl2, but far less oxidizing... |
Oxygen | 4525 | 8177 |
| Dicyanoacetylene | Oxygen | 4990 | 9010 |
| Ethane Ethane Ethane is a chemical compound with chemical formula C2H6. It is the only two-carbon alkane that is an aliphatic hydrocarbon. At standard temperature and pressure, ethane is a colorless, odorless gas.... |
air | 1955 | 3551 |
| Hydrogen Hydrogen Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly... |
air | 2210 | 4010 |
| Hydrogen Hydrogen Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly... |
Oxygen | 3200 | 5792 |
| Methane Methane Methane is a chemical compound with the chemical formula . It is the simplest alkane, the principal component of natural gas, and probably the most abundant organic compound on earth. The relative abundance of methane makes it an attractive fuel... |
air | 1950 | 3542 |
| Natural gas Natural gas Natural gas is a naturally occurring gas mixture consisting primarily of methane, typically with 0–20% higher hydrocarbons . It is found associated with other hydrocarbon fuel, in coal beds, as methane clathrates, and is an important fuel source and a major feedstock for fertilizers.Most natural... |
air | 1960 | 3562 |
| Propane Propane Propane is a three-carbon alkane with the molecular formula , normally a gas, but compressible to a transportable liquid. A by-product of natural gas processing and petroleum refining, it is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves, and residential central... |
air | 1980 | 3596 |
| Propane Propane Propane is a three-carbon alkane with the molecular formula , normally a gas, but compressible to a transportable liquid. A by-product of natural gas processing and petroleum refining, it is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves, and residential central... |
Oxygen | 2526 | 4579 |
| MAPP gas MAPP gas MAPP gas is a trademarked name belonging to Linde Group, previously to Dow, for a fuel gas based on a stabilized mixture of methylacetylene and propadiene. The name comes from the original chemical composition: methylacetylene-propadiene propane... Methylacetylene Methylacetylene Methylacetylene is an alkyne with the chemical formula H3C≡CH. It is a component of MAPP gas along with its isomer 1,2-propadiene , which is commonly used in gas welding... |
air | 2010 | 3650 |
| MAPP gas MAPP gas MAPP gas is a trademarked name belonging to Linde Group, previously to Dow, for a fuel gas based on a stabilized mixture of methylacetylene and propadiene. The name comes from the original chemical composition: methylacetylene-propadiene propane... Methylacetylene Methylacetylene Methylacetylene is an alkyne with the chemical formula H3C≡CH. It is a component of MAPP gas along with its isomer 1,2-propadiene , which is commonly used in gas welding... |
Oxygen | 2927 | 5301 |
| Wood Wood Wood is a hard, fibrous tissue found in many trees. It has been used for hundreds of thousands of years for both fuel and as a construction material. It is an organic material, a natural composite of cellulose fibers embedded in a matrix of lignin which resists compression... |
air | 1980 | 3596 |
| Kerosene Kerosene Kerosene, sometimes spelled kerosine in scientific and industrial usage, also known as paraffin or paraffin oil in the United Kingdom, Hong Kong, Ireland and South Africa, is a combustible hydrocarbon liquid. The name is derived from Greek keros... |
air | 2093 | 3801 |
| Light fuel oil | air | 2104 | 3820 |
| Medium fuel oil | air | 2101 | 3815 |
| Heavy fuel oil | air | 2102 | 3817 |
| Bituminous Coal Bituminous coal Bituminous coal or black coal is a relatively soft coal containing a tarlike substance called bitumen. It is of higher quality than lignite coal but of poorer quality than Anthracite... |
air | 2172 | 3943 |
| Anthracite | air | 2180 | 3957 |
| Anthracite | Oxygen | ≈2900 The temperature equal to ≈3200 K Kelvin The kelvin is a unit of measurement for temperature. It is one of the seven base units in the International System of Units and is assigned the unit symbol K. The Kelvin scale is an absolute, thermodynamic temperature scale using as its null point absolute zero, the temperature at which all... corresponds to 50 % of chemical dissociation for CO2 Carbon dioxide Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom... at pressure 1 atm Atmosphere (unit) The standard atmosphere is an international reference pressure defined as 101325 Pa and formerly used as unit of pressure. For practical purposes it has been replaced by the bar which is 105 Pa... . The latter one stays invariant Invariant (physics) In mathematics and theoretical physics, an invariant is a property of a system which remains unchanged under some transformation.-Examples:In the current era, the immobility of polaris under the diurnal motion of the celestial sphere is a classical illustration of physical invariance.Another... for adiabatic flame and the carbon dioxide constitutes 97 % of total gas output in the case of anthracite burning in oxygen Oxygen Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition... . Higher temperatures will occure for reaction output while it going under higher pressure (up to 3800 K Kelvin The kelvin is a unit of measurement for temperature. It is one of the seven base units in the International System of Units and is assigned the unit symbol K. The Kelvin scale is an absolute, thermodynamic temperature scale using as its null point absolute zero, the temperature at which all... and above, see e.g. Jongsup Hong et al, p.8). |
≈5255 |
Thermodynamics

First law of thermodynamics
The first law of thermodynamics is an expression of the principle of conservation of work.The law states that energy can be transformed, i.e. changed from one form to another, but cannot be created nor destroyed...
for a closed reacting system we have,
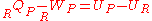
where,
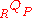 and
and 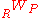 are the heat and work transferred during the process respectively, and
are the heat and work transferred during the process respectively, and 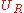 and
and 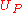 are the internal energy of the reactants and products respectively.
are the internal energy of the reactants and products respectively.In the constant volume adiabatic flame temperature case, the volume of the system is held constant hence there is no work occurring,
and there is no heat transfer because the process is defined to be adiabatic:
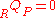 . As a result, the internal energy of the products is equal to the internal energy of the reactants:
. As a result, the internal energy of the products is equal to the internal energy of the reactants:  .
.Because this is a closed system, the mass of the products and reactants is constant and the first law can be written on a mass basis,
-
 .
.


Again there is no heat transfer occurring because the process is defined to be adiabatic:
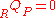 . From the first law, we find that,
. From the first law, we find that,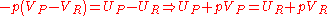
Recalling the definition of enthalpy we recover:
 . Because this is a closed system, the mass of the products and reactants is constant and the first law can be written on a mass basis,
. Because this is a closed system, the mass of the products and reactants is constant and the first law can be written on a mass basis,
-
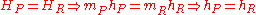 .
.
We see that the adiabatic flame temperature of the constant pressure process is lower than that of the constant volume process. This is because some of the energy released during combustion goes into changing the volume of the control system. One analogy that is commonly made between the two processes is through combustion in an internal combustion engine. For the constant volume adiabatic process, combustion is thought to occur instantaneously when the piston reaches the top of its apex (Otto cycle
Otto cycle
An Otto cycle is an idealized thermodynamic cycle which describes the functioning of a typical reciprocating piston engine, the thermodynamic cycle most commonly found in automobile engines....
or constant volume cycle). For the constant pressure adiabatic process, while combustion is occurring the piston is moving in order to keep the pressure constant (Diesel cycle
Diesel cycle
The Diesel cycle is the thermodynamic cycle which approximates the pressure and volume of the combustion chamber of the Diesel engine, invented by Rudolph Diesel in 1897. It is assumed to have constant pressure during the first part of the "combustion" phase...
or constant pressure cycle).


 and
and 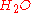 ), we can calculate the adiabatic flame temperature by hand either at stoichiometric conditions or lean of stoichiometry (excess air). This is because there are enough variables and molar equations to balance the left and right hand sides,
), we can calculate the adiabatic flame temperature by hand either at stoichiometric conditions or lean of stoichiometry (excess air). This is because there are enough variables and molar equations to balance the left and right hand sides,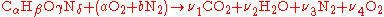
Rich of stoichiometry there are not enough variables because combustion cannot go to completion with at least
 and
and 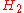 needed for the molar balance (these are the most common incomplete products of combustion),
needed for the molar balance (these are the most common incomplete products of combustion),
However, if we include the Water gas shift reaction
Water gas shift reaction
The water-gas shift reaction is a chemical reaction in which carbon monoxide reacts with water vapor to form carbon dioxide and hydrogen:The water-gas shift reaction is an important industrial reaction. It is often used in conjunction with steam reforming of methane or other hydrocarbons, which is...
,
and use the equilibrium constant for this reaction, we will have enough variables to complete the calculation.
Different fuels with different levels of energy and molar constituents will have different adiabatic flame temperatures.


Nitromethane
Nitromethane is an organic compound with the chemical formula . It is the simplest organic nitro compound. It is a slightly viscous, highly polar liquid commonly used as a solvent in a variety of industrial applications such as in extractions, as a reaction medium, and as a cleaning solvent...
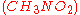 is often used as a power boost for cars. Since it contains two moles of oxygen in its molecular makeup, it can burn much hotter because it provides its own oxidant along with fuel. This in turn allows it to build-up more pressure during a constant volume process. The higher the pressure, the more force upon the piston creating more work and more power in the engine. It is interesting to note that it stays relatively hot rich of stoichiometry because it contains its own oxidant. However, continual running of an engine on nitromethane will eventually melt the piston and/or cylinder because of this higher temperature.
is often used as a power boost for cars. Since it contains two moles of oxygen in its molecular makeup, it can burn much hotter because it provides its own oxidant along with fuel. This in turn allows it to build-up more pressure during a constant volume process. The higher the pressure, the more force upon the piston creating more work and more power in the engine. It is interesting to note that it stays relatively hot rich of stoichiometry because it contains its own oxidant. However, continual running of an engine on nitromethane will eventually melt the piston and/or cylinder because of this higher temperature.
Dissociation (chemistry)
Dissociation in chemistry and biochemistry is a general process in which ionic compounds separate or split into smaller particles, ions, or radicals, usually in a reversible manner...
and kinetics
Chemical kinetics
Chemical kinetics, also known as reaction kinetics, is the study of rates of chemical processes. Chemical kinetics includes investigations of how different experimental conditions can influence the speed of a chemical reaction and yield information about the reaction's mechanism and transition...
will change the relative constituents of the products. There are a number of programs available that can calculate the adiabatic flame temperature taking into account dissociation through equilibrium constants (Stanjan, NASA CEA, AFTP). The following figure illustrates that the effects of dissociation tend to lower the adiabatic flame temperature. This result can be explained through Le Chatelier's principle
Le Châtelier's principle
In chemistry, Le Chatelier's principle, also called the Chatelier's principle, can be used to predict the effect of a change in conditions on a chemical equilibrium. The principle is named after Henry Louis Le Chatelier and sometimes Karl Ferdinand Braun who discovered it independently...
.
General information
Tables
adiabatic flame temperature of hydrogen, methane, propane and octane with oxygen or air as oxidizersCalculators
- Online adiabatic flame temperature calculator using CanteraCantera (software)Cantera is an open-source chemical kinetics software used for solving chemically reacting laminar flows. It has been used as a third party library in external reacting flow simulation codes, such as FUEGO and CADS, using Fortran, C++, etc. to evaluate properties and chemical source terms that...
- Adiabatic flame temperature program
- Gaseq, program for performing chemical equilibrium calculations.
- Flame Temperature Calculator - Constant pressure bipropellant adiabatic combustion
- Adiabatic Flame Temperature calculator