
Associative substitution
Encyclopedia
Associative substitution describes a pathway by which compounds interchange ligand
s. The terminology is typically applied to coordination and organometallic
complexes, but resembles the Sn2 mechanism
in organic chemistry
. The opposite pathway is dissociative substitution
, being analogous to Sn1 pathway
. Intermediate pathways exist between the pure associative and pure dissociative pathways, these are called interchange mechanisms.
Associative pathways are characterized by binding of the attacking (nucleophile
) to give a discrete, detectable intermediate followed by loss of another ligand. Complexes that undergo associative substitution are either coordinatively unsaturated or contain a ligand that can change its bonding to the metal, e.g. change in hapticity
or bending of NO. In homogeneous catalysis
, the associative pathway is desirable because the binding event, and hence the selectivity of the reaction, depends not only on the nature of the metal catalyst but also on the substrate.
Examples of associative mechanisms are commonly found in the chemistry of 16e square planar metal complexes, e.g. Vaska's complex
and tetrachloroplatinate
. These compounds (MX4) bind the incoming (substituting) ligand Y to form pentacoordinate intermediates MX4Y that in a subsequent step dissociates one of their ligands. Dissociation of Y results in no reaction, but dissociation of X results in net substitution, giving the 16e complex MX3Y. The first step is typically rate determining. Thus, the entropy of activation
is negative, which indicates an increase in order in the system. These reactions follow second order kinetics: the rate of the appearance of product depends on the concentration of MX4 and Y.
in which an indenyl
ligand reversibly "slips' from pentahapto (η5) coordination to trihapto (η3). Other pi-ligands behave in this way, e.g. allyl (η3 to η1) and naphthalene
(η6 to η4). Nitric oxide typically binds to metals to make a linear MNO arrangement, wherein the nitrogen oxide is said to donate 3e- to the metal. In the course of substitution reactions, the MNO unit can bend, converting the 3e- linear NO ligand to a 1e- bent NO ligand.
of cobalt(III) ammine halide complexes are deceptive, appearing to be associative but proceeding by an alternative pathway. The hydrolysis of [Co(NH3)5Cl]2+ follows second order kinetics: the rate increases linearly with concentration of hydroxide as well as the starting complex. Based on this information, the reactions would appear to proceed via nucleophilic attack of hydroxide at cobalt. Studies show, however, that in the hydroxide deprotonates one NH3 ligand to give the conjugate base of the starting complex, i.e., [Co(NH3)4(NH2)Cl]+. In this monocation, the chloride spontaneously dissociates. This pathway is called the Sn1CB mechanism
.
Ligand
In coordination chemistry, a ligand is an ion or molecule that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from...
s. The terminology is typically applied to coordination and organometallic
Organometallic chemistry
Organometallic chemistry is the study of chemical compounds containing bonds between carbon and a metal. Since many compounds without such bonds are chemically similar, an alternative may be compounds containing metal-element bonds of a largely covalent character...
complexes, but resembles the Sn2 mechanism
SN2 reaction
The SN2 reaction is a type of nucleophilic substitution, where a lone pair from a nucleophile attacks an electron deficient electrophilic center and bonds to it, expelling another group called a leaving group. Thus the incoming group replaces the leaving group in one step...
in organic chemistry
Organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, composition, reactions, and preparation of carbon-based compounds, hydrocarbons, and their derivatives...
. The opposite pathway is dissociative substitution
Dissociative substitution
Dissociative substitution describes a pathway by which compounds interchange ligands. The term is typically applied to coordination and organometallic complexes, but resembles the Sn1 mechanism in organic chemistry. The opposite pathway is associative substitution, being analogous to Sn2 pathway...
, being analogous to Sn1 pathway
SN1 reaction
The SN1 reaction is a substitution reaction in organic chemistry. "SN" stands for nucleophilic substitution and the "1" represents the fact that the rate-determining step is unimolecular...
. Intermediate pathways exist between the pure associative and pure dissociative pathways, these are called interchange mechanisms.
Associative pathways are characterized by binding of the attacking (nucleophile
Nucleophile
A nucleophile is a species that donates an electron-pair to an electrophile to form a chemical bond in a reaction. All molecules or ions with a free pair of electrons can act as nucleophiles. Because nucleophiles donate electrons, they are by definition Lewis bases.Nucleophilic describes the...
) to give a discrete, detectable intermediate followed by loss of another ligand. Complexes that undergo associative substitution are either coordinatively unsaturated or contain a ligand that can change its bonding to the metal, e.g. change in hapticity
Hapticity
The term hapticity is used to describe how a group of contiguous atoms of a ligand are coordinated to a central atom. Hapticity of a ligand is indicated by the Greek character 'eta', η. A superscripted number following the η denotes the number of contiguous atoms of the ligand that are bound to...
or bending of NO. In homogeneous catalysis
Homogeneous catalysis
In chemistry, homogeneous catalysis is a sequence of reactions that involve a catalyst in the same phase as the reactants. Most commonly, a homogeneous catalyst is codissolved in a solvent with the reactants.-Acid catalysis:...
, the associative pathway is desirable because the binding event, and hence the selectivity of the reaction, depends not only on the nature of the metal catalyst but also on the substrate.
Examples of associative mechanisms are commonly found in the chemistry of 16e square planar metal complexes, e.g. Vaska's complex
Vaska's complex
Vaska's complex is the trivial name for the chemical compound trans-chlorocarbonylbisiridium, which has the formula IrCl[P3]2. This square planar diamagnetic organometallic complex consists of a central iridium atom bound to two mutually trans triphenylphosphine ligands, carbon monoxide, and a...
and tetrachloroplatinate
Potassium tetrachloroplatinate
Potassium tetrachloroplatinate is the chemical compound with the formula K2PtCl4. This reddish orange salt is an important reagent for the preparation of other coordination complexes of platinum. It consists of potassium cations and the square planar dianion PtCl42-...
. These compounds (MX4) bind the incoming (substituting) ligand Y to form pentacoordinate intermediates MX4Y that in a subsequent step dissociates one of their ligands. Dissociation of Y results in no reaction, but dissociation of X results in net substitution, giving the 16e complex MX3Y. The first step is typically rate determining. Thus, the entropy of activation
Entropy of activation
The entropy of activation is one of the two parameters typically obtained from the temperature dependence of a reaction rate, when these data are analysed using the Eyring equation. Symbolized ΔS‡, the entropy of activation provides clues about the molecularity of the rate determining step in a...
is negative, which indicates an increase in order in the system. These reactions follow second order kinetics: the rate of the appearance of product depends on the concentration of MX4 and Y.
Associative interchange pathway
In many substitution reactions, well-defined intermediates are not observed, when the rate of such processes are influenced by the nature of the entering ligand, the pathway is called associative interchange, abbreviated Ia. Representative is the interchange of bulk and coordinated water in [V(H2O)6]2+. In contrast, the slightly more compact ion [Ni(H2O)6]2+ exchanges water via the Id.Effects of ion pairing
Polycationic complexes tend to form ion pairs with anions and these ion pairs often undergo reactions via the Ia pathway. The electrostatically held nucleophile can exchange positions with a ligand in the first coordination sphere, resulting in net substitution. An illustrative process comes from the "anation" (reaction with an anion) of chromium(III) hexaaquo complex:-
- [Cr(H2O)6]3+ + SCN-ThiocyanateThiocyanate is the anion [SCN]−. It is the conjugate base of thiocyanic acid. Common derivatives include the colourless salts potassium thiocyanate and sodium thiocyanate. Organic compounds containing the functional group SCN are also called thiocyanates...
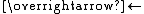 {[Cr(H2O)6], NCS}2+
{[Cr(H2O)6], NCS}2+ - {[Cr(H2O)6], NCS}2+
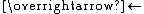 [Cr(H2O)5NCS]2+ + H2O
[Cr(H2O)5NCS]2+ + H2O
- [Cr(H2O)6]3+ + SCN-
Special ligand effects
In special situations, some ligands participate in substitution reactions leading to associative pathways. These ligands can adopt multiple motifs for binding to the metal, each of which involves a different number of electrons "donated." A classic case is the indenyl effectIndenyl effect
In organometallic chemistry, the indenyl effect refers to the enhanced rates of substitution displayed by η5-indenyl complexes vs the related η5-cyclopentadienyl complexes.-Mechanism:...
in which an indenyl
Indene
Indene is a flammable polycyclic hydrocarbon with chemical formula C9H8. It is composed of a benzene ring fused with a cyclopentene ring. This aromatic liquid is colorless although samples often are pale yellow...
ligand reversibly "slips' from pentahapto (η5) coordination to trihapto (η3). Other pi-ligands behave in this way, e.g. allyl (η3 to η1) and naphthalene
Naphthalene
Naphthalene is an organic compound with formula . It is a white crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 ppm by mass. As an aromatic hydrocarbon, naphthalene's structure consists of a fused pair of benzene rings...
(η6 to η4). Nitric oxide typically binds to metals to make a linear MNO arrangement, wherein the nitrogen oxide is said to donate 3e- to the metal. In the course of substitution reactions, the MNO unit can bend, converting the 3e- linear NO ligand to a 1e- bent NO ligand.
Sn1CB mechanism
The rate for the hydrolysisHydrolysis
Hydrolysis is a chemical reaction during which molecules of water are split into hydrogen cations and hydroxide anions in the process of a chemical mechanism. It is the type of reaction that is used to break down certain polymers, especially those made by condensation polymerization...
of cobalt(III) ammine halide complexes are deceptive, appearing to be associative but proceeding by an alternative pathway. The hydrolysis of [Co(NH3)5Cl]2+ follows second order kinetics: the rate increases linearly with concentration of hydroxide as well as the starting complex. Based on this information, the reactions would appear to proceed via nucleophilic attack of hydroxide at cobalt. Studies show, however, that in the hydroxide deprotonates one NH3 ligand to give the conjugate base of the starting complex, i.e., [Co(NH3)4(NH2)Cl]+. In this monocation, the chloride spontaneously dissociates. This pathway is called the Sn1CB mechanism
Sn1CB mechanism
The SN1CB mechanism describes the pathway by which many metal amine complexes undergo substitution, that is ligand exchange. Typically, the reaction entails reaction of a polyamino metal halide with aqueous base to give the corresponding polyamine metal hydroxide:The rate law for the reaction...
.

