.gif)
Conductivity (electrolytic)
Encyclopedia
The conductivity of an electrolyte
solution is a measure of its ability to conduct electricity. The SI
unit of conductivity is siemens
per meter (S/m).
Conductivity measurements are used routinely in many industrial and environmental applications as a fast, inexpensive and reliable way of measuring the ionic content in a solution. For example, the measurement of product conductivity is a typical way to monitor and continuously trend the performance of the water purification
systems.
In many cases, conductivity is linked directly to the total dissolved solids
(T.D.S.). High quality deionized water has a conductivity of about 5.5 μS/m, typical drinking water in the range of 5-50 mS/m, while sea water about 5 S/m (i.e., sea water's conductivity is one million times higher than deionized water).
Conductivity is traditionally determined by measuring the AC
resistance
of the solution between two electrode
s. Dilute solutions follow Kohlrausch's Laws of concentration dependence and additivity of ionic contributions. Onsager
gave a theoretical explanation of Kohlrausch's law by extending Debye–Hückel theory.
unit of conductivity is S
/m and, unless otherwise qualified, it refers to 25 °C (standard temperature). Often encountered in industry is the traditional unit of μS/cm. The numbers in μS/cm are lower than those in μS/m by a factor of 100 (i.e., a solution with a conductivity of 100 μS/m has a conductivity of 1 μS/cm). Occasionally a unit of "EC" (electrical conductivity) is found on scales of instruments: 1 EC = 1 μS/cm. Sometimes encountered is so-called mho (reciprocal of ohm): 1 mho/m = 1 S/m. Historically, mhos antedate Siemens by many decades; good vacuum-tube testers, for instance, gave transconductance readings in micromhos.
The commonly used standard cell has a width of 1 cm, and thus for very pure water in equilibrium with air would have a resistance of about 106 ohm, known as a megohm, occasionally misspelled as "megaohm". Ultra-pure water
could achieve 18 megohms or more. Thus in the past megohm-cm was used, sometimes abbreviated to "megohm". Sometimes, a conductivity is given just in "microSiemens" (omitting the distance term in the unit). While this is an error, it can often be assumed to be equal to the traditional μS/cm. The typical conversion of conductivity to the total dissolved solids is done assuming that the solid is sodium chloride: 1 μS/cm is then an equivalent of about 0.6 mg of NaCl per kg of water.
Molar conductivity has the SI unit S m2 mol−1. Older publications use the unit Ω−1 cm2 mol−1.
 The electrical conductivity of a solution of an electrolyte
The electrical conductivity of a solution of an electrolyte
is measured by determining the resistance
of the solution between two flat or cylindrical electrode
s separated by a fixed distance. An alternating voltage is used in order to avoid electrolysis
. The resistance is measured by a conductivity meter. Typical frequencies used are in the range 1–3 kHz
. The dependence on the frequency is usually small, but may become appreciable at very high frequencies, an effect known as the Debye–Falkenhagen effect.
A wide variety of instrumentation is commercially available. There are two types of cell, the classical type with flat or cylindrical electrodes and a second type based on induction. Many commercial systems offer automatic temperature correction.
),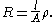
In practice the conductivity cell is calibrated by using solutions of known specific resistance, ρ*, so the quantities l and A need not be known precisely. If the resistance of the calibration solution is R*, a cell-constant, C, is derived.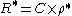
The specific conductance, κ (kappa) is the reciprocal of the specific resistance.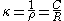
Conductivity is also temperature-dependent.
, is denoted by Λm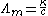
s are believed to dissociate completely in solution. The conductivity of a solution of a strong electrolyte at low concentration follows Kohlrausch's Law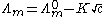
where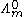 is known as the limiting molar conductivity, K is an empirical constant and c is the electrolyte concentration. (Limiting here means "at the limit of the infinite dilution".)
is known as the limiting molar conductivity, K is an empirical constant and c is the electrolyte concentration. (Limiting here means "at the limit of the infinite dilution".)
Moreover, Kohlrausch also found that the limiting conductivity of anions and cations are additive: the conductivity of a solution of a salt is equal to the sum of conductivity contributions from the cation and anion.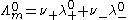
where:
The following table gives values for limiting molar conductivities for selected ions.
A theoretical interpretation of these results was provided by the Debye–Hückel.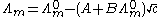
where A and B are constants that depend only on known quantities such as temperature, the charges on the ions and the dielectric constant
and viscosity
of the solvent. As the name suggests, this is an extension of the Debye–Hückel theory, due to Onsager
. It is very successful for solutions at low concentration.
s and weak base
s. The concentration of ions in a solution of a weak electrolyte is less than the concentration of the electrolyte itself. For acids and bases the concentrations can be calculated when the value(s) of the acid dissociation constant
(s) is(are) known.
For a monoprotic acid, HA, with a dissociation constant Ka, an explicit expression for the conductivity as a function of concentration, c, known as Ostwald's dilution law, can be obtained.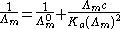
is a moot point. However, It has often been assumed that cation and anion interact to form an ion-pair
. Thus the electrolyte is treated as if it were like a weak acid and a constant, K, can be derived for the equilibrium
Davies describes the results of such calculations in great detail, but states that K should not necessarily be thought of as a true equilibrium constant, rather, the inclusion of an "ion-association" term is useful in extending the range of good agreement between theory and experimental conductivity data. Various attempts have been made to extend Onsager's treament to more concentrated solutions.
The existence of a so-called conductance minimum has proved to be a controversial subject as regards interpretation. Fuoss and Kraus suggested that it is caused by the formation of ion-triplets, and this suggestion has received some support recently.
(T.D.S.) if the composition of the solution and its conductivity behavior are known.
Sometimes, conductivity measurements are linked with other methods to increase the sensitivity of detection of specific types of ions. For example, in the boiler water technology, the boiler blowdown is continuously monitored for "cation conductivity", which is the conductivity of the water after it has been passed through a cation exchange resin. This is a sensitive method of monitoring anion impurities in the boiler water in the presence of excess cations (those of the alkalizing agent usually used for water treatment). The sensitivity of this method relies on the high mobility of H+ in comparison with the mobility of other cations or anions.
Conductivity detectors are commonly used with ion chromatography.
Electrolyte
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible....
solution is a measure of its ability to conduct electricity. The SI
Si
Si, si, or SI may refer to :- Measurement, mathematics and science :* International System of Units , the modern international standard version of the metric system...
unit of conductivity is siemens
Siemens (unit)
The siemens is the SI derived unit of electric conductance and electric admittance. Conductance and admittance are the reciprocals of resistance and impedance respectively, hence one siemens is equal to the reciprocal of one ohm, and is sometimes referred to as the mho. In English, the term...
per meter (S/m).
Conductivity measurements are used routinely in many industrial and environmental applications as a fast, inexpensive and reliable way of measuring the ionic content in a solution. For example, the measurement of product conductivity is a typical way to monitor and continuously trend the performance of the water purification
Water purification
Water purification is the process of removing undesirable chemicals, materials, and biological contaminants from contaminated water. The goal is to produce water fit for a specific purpose...
systems.
In many cases, conductivity is linked directly to the total dissolved solids
Total dissolved solids
Total Dissolved Solids is a measure of the combined content of all inorganic and organic substances contained in a liquid in: molecular, ionized or micro-granular suspended form. Generally the operational definition is that the solids must be small enough to survive filtration through a sieve...
(T.D.S.). High quality deionized water has a conductivity of about 5.5 μS/m, typical drinking water in the range of 5-50 mS/m, while sea water about 5 S/m (i.e., sea water's conductivity is one million times higher than deionized water).
Conductivity is traditionally determined by measuring the AC
Alternating current
In alternating current the movement of electric charge periodically reverses direction. In direct current , the flow of electric charge is only in one direction....
resistance
Electrical resistance
The electrical resistance of an electrical element is the opposition to the passage of an electric current through that element; the inverse quantity is electrical conductance, the ease at which an electric current passes. Electrical resistance shares some conceptual parallels with the mechanical...
of the solution between two electrode
Electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit...
s. Dilute solutions follow Kohlrausch's Laws of concentration dependence and additivity of ionic contributions. Onsager
Onsager
Onsager may refer to:* Lars Onsager, a Norwegian–American physical chemist and theoretical physicist* Onsager reciprocal relations, certain relations between flows and forces in thermodynamic systems...
gave a theoretical explanation of Kohlrausch's law by extending Debye–Hückel theory.
Units
The SISi
Si, si, or SI may refer to :- Measurement, mathematics and science :* International System of Units , the modern international standard version of the metric system...
unit of conductivity is S
Siemens (unit)
The siemens is the SI derived unit of electric conductance and electric admittance. Conductance and admittance are the reciprocals of resistance and impedance respectively, hence one siemens is equal to the reciprocal of one ohm, and is sometimes referred to as the mho. In English, the term...
/m and, unless otherwise qualified, it refers to 25 °C (standard temperature). Often encountered in industry is the traditional unit of μS/cm. The numbers in μS/cm are lower than those in μS/m by a factor of 100 (i.e., a solution with a conductivity of 100 μS/m has a conductivity of 1 μS/cm). Occasionally a unit of "EC" (electrical conductivity) is found on scales of instruments: 1 EC = 1 μS/cm. Sometimes encountered is so-called mho (reciprocal of ohm): 1 mho/m = 1 S/m. Historically, mhos antedate Siemens by many decades; good vacuum-tube testers, for instance, gave transconductance readings in micromhos.
The commonly used standard cell has a width of 1 cm, and thus for very pure water in equilibrium with air would have a resistance of about 106 ohm, known as a megohm, occasionally misspelled as "megaohm". Ultra-pure water
Ultrapure Water
The term ultrapure water offers a challenge in being described with a single definition. Until the advent of SEMI F63, ultrapure water used for semiconductor manufacturing was loosely defined. The SEMI F63 template has been followed recently with a UPW version for photovoltaic cell manufacturing...
could achieve 18 megohms or more. Thus in the past megohm-cm was used, sometimes abbreviated to "megohm". Sometimes, a conductivity is given just in "microSiemens" (omitting the distance term in the unit). While this is an error, it can often be assumed to be equal to the traditional μS/cm. The typical conversion of conductivity to the total dissolved solids is done assuming that the solid is sodium chloride: 1 μS/cm is then an equivalent of about 0.6 mg of NaCl per kg of water.
Molar conductivity has the SI unit S m2 mol−1. Older publications use the unit Ω−1 cm2 mol−1.
Measurement

Electrolyte
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible....
is measured by determining the resistance
Electrical resistance
The electrical resistance of an electrical element is the opposition to the passage of an electric current through that element; the inverse quantity is electrical conductance, the ease at which an electric current passes. Electrical resistance shares some conceptual parallels with the mechanical...
of the solution between two flat or cylindrical electrode
Electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit...
s separated by a fixed distance. An alternating voltage is used in order to avoid electrolysis
Electrolysis
In chemistry and manufacturing, electrolysis is a method of using a direct electric current to drive an otherwise non-spontaneous chemical reaction...
. The resistance is measured by a conductivity meter. Typical frequencies used are in the range 1–3 kHz
Hertz
The hertz is the SI unit of frequency defined as the number of cycles per second of a periodic phenomenon. One of its most common uses is the description of the sine wave, particularly those used in radio and audio applications....
. The dependence on the frequency is usually small, but may become appreciable at very high frequencies, an effect known as the Debye–Falkenhagen effect.
A wide variety of instrumentation is commercially available. There are two types of cell, the classical type with flat or cylindrical electrodes and a second type based on induction. Many commercial systems offer automatic temperature correction.
Definitions
Resistance, R, is proportional to the distance, l, between the electrodes and is inversely proportional to the cross-sectional area of the sample, A. Writing ρ (rho) for the specific resistance (or resistivityResistivity
Electrical resistivity is a measure of how strongly a material opposes the flow of electric current. A low resistivity indicates a material that readily allows the movement of electric charge. The SI unit of electrical resistivity is the ohm metre...
),
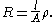
In practice the conductivity cell is calibrated by using solutions of known specific resistance, ρ*, so the quantities l and A need not be known precisely. If the resistance of the calibration solution is R*, a cell-constant, C, is derived.
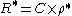
The specific conductance, κ (kappa) is the reciprocal of the specific resistance.
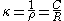
Conductivity is also temperature-dependent.
Theory
The conductivity of a solution containing one electrolyte depends on the concentration of the electrolyte. Therefore it is convenient to divide the conductivity by concentration. This quotient is termed molar conductivityMolar conductivity
Molar conductivity is defined as the conductivity of an electrolyte solution divided by the molar concentration of the electrolyte, and so measures the efficiency with which a given electrolyte conducts electricity in solution. Its units are siemens per meter per molarity, or siemens meter-squared...
, is denoted by Λm
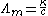
Strong electrolytes
Strong electrolyteStrong electrolyte
A strong electrolyte is a solute that completely, or almost completely, ionizes or dissociates in a solution. These ions are good conductors of electric current in the solution....
s are believed to dissociate completely in solution. The conductivity of a solution of a strong electrolyte at low concentration follows Kohlrausch's Law
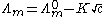
where
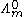 is known as the limiting molar conductivity, K is an empirical constant and c is the electrolyte concentration. (Limiting here means "at the limit of the infinite dilution".)
is known as the limiting molar conductivity, K is an empirical constant and c is the electrolyte concentration. (Limiting here means "at the limit of the infinite dilution".)Moreover, Kohlrausch also found that the limiting conductivity of anions and cations are additive: the conductivity of a solution of a salt is equal to the sum of conductivity contributions from the cation and anion.
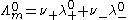
where:
-
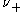 and
and 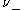 are the number of moles of cations and anions, respectively, which are created from the dissociation of 1 mole of the dissolved electrolyte;
are the number of moles of cations and anions, respectively, which are created from the dissociation of 1 mole of the dissolved electrolyte; -
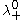 and
and 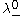 are the limiting molar conductivities of the individual ions.
are the limiting molar conductivities of the individual ions.
The following table gives values for limiting molar conductivities for selected ions.
Limiting ion conductivity in water at 298 K Cations λ+0 /mS m2mol-1 anions λ-0 /mS m2mol-1 H+ 34.96 OH- 19.91 Li+ 3.869 Cl- 7.634 Na+ 5.011 Br- 7.84 K+ 7.350 I- 7.68 Mg2+ 10.612 SO42- 15.96 Ca2+ 11.900 NO3- 7.14 Ba2+ 12.728 CH3CO2- AcetateAn acetate is a derivative of acetic acid. This term includes salts and esters, as well as the anion found in solution. Most of the approximately 5 billion kilograms of acetic acid produced annually in industry are used in the production of acetates, which usually take the form of polymers. In...4.09
A theoretical interpretation of these results was provided by the Debye–Hückel.
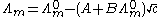
where A and B are constants that depend only on known quantities such as temperature, the charges on the ions and the dielectric constant
Dielectric constant
The relative permittivity of a material under given conditions reflects the extent to which it concentrates electrostatic lines of flux. In technical terms, it is the ratio of the amount of electrical energy stored in a material by an applied voltage, relative to that stored in a vacuum...
and viscosity
Viscosity
Viscosity is a measure of the resistance of a fluid which is being deformed by either shear or tensile stress. In everyday terms , viscosity is "thickness" or "internal friction". Thus, water is "thin", having a lower viscosity, while honey is "thick", having a higher viscosity...
of the solvent. As the name suggests, this is an extension of the Debye–Hückel theory, due to Onsager
Lars Onsager
Lars Onsager was a Norwegian-born American physical chemist and theoretical physicist, winner of the 1968 Nobel Prize in Chemistry.He held the Gibbs Professorship of Theoretical Chemistry at Yale University....
. It is very successful for solutions at low concentration.
Weak electrolytes
A weak electrolyte is one that is not fully dissociated . Typical weak electrolytes are weak acidWeak acid
A weak acid is an acid that dissociates incompletely. It does not release all of its hydrogens in a solution, donating only a partial amount of its protons to the solution...
s and weak base
Weak base
In chemistry, a weak base is a chemical base that does not ionize fully in an aqueous solution. As Brønsted–Lowry bases are proton acceptors, a weak base may also be defined as a chemical base in which protonation is incomplete. This results in a relatively low pH compared to strong bases...
s. The concentration of ions in a solution of a weak electrolyte is less than the concentration of the electrolyte itself. For acids and bases the concentrations can be calculated when the value(s) of the acid dissociation constant
Acid dissociation constant
An acid dissociation constant, Ka, is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction known as dissociation in the context of acid-base reactions...
(s) is(are) known.
For a monoprotic acid, HA, with a dissociation constant Ka, an explicit expression for the conductivity as a function of concentration, c, known as Ostwald's dilution law, can be obtained.
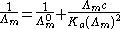
Higher concentrations
Both Kolrausch's law and the Debye-Hückel-Onsager equation break down as the concentration of the electrolyte increases above a certain value. The reason for this is that as concentration increases the average distance between cation and anion decreases, so that there is more inter-ionic interaction. Whether this constitutes ion-associationIon-association
Ion-association is a chemical reaction whereby ions of opposite electrical charge come together in solution to form a distinct chemical entity. Ion-associates are classified according to the number of ions that associate with each other, and the nature of the interaction. The most important factor...
is a moot point. However, It has often been assumed that cation and anion interact to form an ion-pair
Ion-association
Ion-association is a chemical reaction whereby ions of opposite electrical charge come together in solution to form a distinct chemical entity. Ion-associates are classified according to the number of ions that associate with each other, and the nature of the interaction. The most important factor...
. Thus the electrolyte is treated as if it were like a weak acid and a constant, K, can be derived for the equilibrium
- A+ + B- A+B-; K=[A+][B-]/[A+B-]
Davies describes the results of such calculations in great detail, but states that K should not necessarily be thought of as a true equilibrium constant, rather, the inclusion of an "ion-association" term is useful in extending the range of good agreement between theory and experimental conductivity data. Various attempts have been made to extend Onsager's treament to more concentrated solutions.
The existence of a so-called conductance minimum has proved to be a controversial subject as regards interpretation. Fuoss and Kraus suggested that it is caused by the formation of ion-triplets, and this suggestion has received some support recently.
Applications
Notwithstanding the difficulty of theoretical interpretation, conductivity measurements are used extensively in many industries. For example, conductivity measurements are used to monitor quality in public water supplies, in hospitals, in boiler water and industries which depend on water quality such as brewing. This type of measurement is not ion-specific; it can sometimes be used to determine the amount of total dissolved solidsTotal dissolved solids
Total Dissolved Solids is a measure of the combined content of all inorganic and organic substances contained in a liquid in: molecular, ionized or micro-granular suspended form. Generally the operational definition is that the solids must be small enough to survive filtration through a sieve...
(T.D.S.) if the composition of the solution and its conductivity behavior are known.
Sometimes, conductivity measurements are linked with other methods to increase the sensitivity of detection of specific types of ions. For example, in the boiler water technology, the boiler blowdown is continuously monitored for "cation conductivity", which is the conductivity of the water after it has been passed through a cation exchange resin. This is a sensitive method of monitoring anion impurities in the boiler water in the presence of excess cations (those of the alkalizing agent usually used for water treatment). The sensitivity of this method relies on the high mobility of H+ in comparison with the mobility of other cations or anions.
Conductivity detectors are commonly used with ion chromatography.
See also
- Debye–Falkenhagen effect
- Svante ArrheniusSvante ArrheniusSvante August Arrhenius was a Swedish scientist, originally a physicist, but often referred to as a chemist, and one of the founders of the science of physical chemistry...
- Alfred WernerAlfred WernerAlfred Werner was a Swiss chemist who was a student at ETH Zurich and a professor at the University of Zurich. He won the Nobel Prize in Chemistry in 1913 for proposing the octahedral configuration of transition metal complexes. Werner developed the basis for modern coordination chemistry...
- coordination chemistry - Conductimetric titrationEquivalence pointThe equivalence point, or stoichiometric point, of a chemical reaction when a titrant is added and is stoichiometrically equal to the amount of moles of substance present in the sample: the smallest amount of titrant that is sufficient to fully neutralize or react with the analyte...
- methods to determine the equivalence point
External links
- Cole-Parmer Technical Library Conductivity
- Conductivity measurement PowerPoint presentation

