
Dye-sensitized solar cells
Encyclopedia

Solar cell
A solar cell is a solid state electrical device that converts the energy of light directly into electricity by the photovoltaic effect....
belonging to the group of thin film solar cell
Thin film solar cell
A thin-film solar cell , also called a thin-film photovoltaic cell , is a solar cell that is made by depositing one or more thin layers of photovoltaic material on a substrate...
s. It is based on a semiconductor
Semiconductor
A semiconductor is a material with electrical conductivity due to electron flow intermediate in magnitude between that of a conductor and an insulator. This means a conductivity roughly in the range of 103 to 10−8 siemens per centimeter...
formed between a photo-sensitized anode and an electrolyte
Electrolyte
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible....
, a photoelectrochemical system. A later version of a dye solar cell, also known as the Grätzel cell, was invented by Michael Grätzel
Michael Grätzel
Michael Grätzel is a professor at the École Polytechnique Fédérale de Lausanne where he directs the Laboratory of Photonics and Interfaces. He pioneered research on energy and electron transfer reactions in mesoscopic-materials and their optoelectronic applications...
and Brian O'Regan at the École Polytechnique Fédérale de Lausanne
École polytechnique fédérale de Lausanne
The École polytechnique fédérale de Lausanne is one of the two Swiss Federal Institutes of Technology and is located in Lausanne, Switzerland.The school was founded by the Swiss Federal Government with the stated mission to:...
in 1991. Michael Grätzel won the 2010 Millennium Technology Prize
Millennium Technology Prize
The Millennium Technology Prize is the largest technology prize in the world. It is awarded once every two years by Technology Academy Finland, an independent fund established by Finnish industry and the Finnish state in partnership. The prize is presented by the President of Finland...
for its invention.
Because it could potentially be made of low-cost materials, if the use of noble materials like Platinum and Ruthenium is limited, and does not require elaborate apparatus to manufacture, this cell is technically attractive. Likewise, manufacture can be significantly less expensive than older solid-state cell designs. It can also be engineered into flexible sheets and is mechanically robust, requiring no protection from minor events like hail or tree strikes. Although its conversion efficiency is less than the best thin-film cells, in theory its price/performance ratio
Price/performance ratio
In economics and engineering, the price/performance ratio refers to a product's ability to deliver performance, of any sort, for its price. Generally speaking, products with a higher price/performance ratio are more desirable, excluding other factors....
(kWh/(m2·annum·dollar)) should be high enough to allow them to compete with fossil fuel electrical generation by achieving grid parity
Grid parity
Grid parity is the point at which alternative means of generating electricity is at least as cheap as grid power.For solar energy, it is achieved first in areas with abundant sun and high costs for electricity such as in California, Hawaii, Spain and Japan. Many solar power advocates predict that...
. Commercial applications, which were held up due to chemical stability problems, are now forecast in the European Union Photovoltaic Roadmap to significantly contribute to renewable electricity generation by 2020.
Current technology: semiconductor solar cells
In a traditional solid-stateSolid state (electronics)
Solid-state electronics are those circuits or devices built entirely from solid materials and in which the electrons, or other charge carriers, are confined entirely within the solid material...
semiconductor
Semiconductor
A semiconductor is a material with electrical conductivity due to electron flow intermediate in magnitude between that of a conductor and an insulator. This means a conductivity roughly in the range of 103 to 10−8 siemens per centimeter...
, a solar cell
Solar cell
A solar cell is a solid state electrical device that converts the energy of light directly into electricity by the photovoltaic effect....
is made from two doped crystals, one doped with n-type impurities (n-type semiconductor
N-type semiconductor
N-type semiconductors are a type of extrinsic semiconductor where the dopant atoms are capable of providing extra conduction electrons to the host material . This creates an excess of negative electron charge carriers....
), which add additional free conduction band electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
s, and the other doped with p-type impurities (p-type semiconductor
P-type semiconductor
A P-type semiconductor is obtained by carrying out a process of doping: that is, adding a certain type of atoms to the semiconductor in order to increase the number of free charge carriers ....
), which add additional electron hole
Electron hole
An electron hole is the conceptual and mathematical opposite of an electron, useful in the study of physics, chemistry, and electrical engineering. The concept describes the lack of an electron at a position where one could exist in an atom or atomic lattice...
s. When placed in contact, some of the electrons in the n-type portion flow into the p-type to "fill in" the missing electrons, also known as electron holes. Eventually enough electrons will flow across the boundary to equalize the Fermi level
Fermi level
The Fermi level is a hypothetical level of potential energy for an electron inside a crystalline solid. Occupying such a level would give an electron a potential energy \epsilon equal to its chemical potential \mu as they both appear in the Fermi-Dirac distribution function,which...
s of the two materials. The result is a region at the interface, the p-n junction
P-n junction
A p–n junction is formed at the boundary between a P-type and N-type semiconductor created in a single crystal of semiconductor by doping, for example by ion implantation, diffusion of dopants, or by epitaxy .If two separate pieces of material were used, this would...
, where charge carriers are depleted and/or accumulated on each side of the interface. In silicon, this transfer of electrons produces a potential barrier of about 0.6 to 0.7 V
Volt
The volt is the SI derived unit for electric potential, electric potential difference, and electromotive force. The volt is named in honor of the Italian physicist Alessandro Volta , who invented the voltaic pile, possibly the first chemical battery.- Definition :A single volt is defined as the...
.
When placed in the sun, photon
Photon
In physics, a photon is an elementary particle, the quantum of the electromagnetic interaction and the basic unit of light and all other forms of electromagnetic radiation. It is also the force carrier for the electromagnetic force...
s of the sunlight can excite electrons on the p-type side of the semiconductor, a process known as photoexcitation. In silicon, sunlight can provide enough energy to push an electron out of the lower-energy valence band
Valence band
In solids, the valence band is the highest range of electron energies in which electrons are normally present at absolute zero temperature....
into the higher-energy conduction band
Conduction band
In the solid-state physics field of semiconductors and insulators, the conduction band is the range of electron energies, higher than that of the valence band, sufficient to free an electron from binding with its individual atom and allow it to move freely within the atomic lattice of the material...
. As the name implies, electrons in the conduction band are free to move about the silicon. When a load is placed across the cell as a whole, these electrons will flow out of the p-type side into the n-type side, lose energy while moving through the external circuit, and then back into the p-type material where they can once again re-combine with the valence-band hole they left behind. In this way, sunlight creates an electrical current.
In any semiconductor, the band gap
Band gap
In solid state physics, a band gap, also called an energy gap or bandgap, is an energy range in a solid where no electron states can exist. In graphs of the electronic band structure of solids, the band gap generally refers to the energy difference between the top of the valence band and the...
means that only photons with that amount of energy, or more, will contribute to producing a current. In the case of silicon, the majority of visible light from red to violet has sufficient energy to make this happen. Unfortunately higher energy photons, those at the blue and violet end of the spectrum, have more than enough energy to cross the band gap; although some of this extra energy is transferred into the electrons, the majority of it is wasted as heat. Another issue is that in order to have a reasonable chance of capturing a photon, the n-type layer has to be fairly thick. This also increases the chance that a freshly ejected electron will meet up with a previously created hole in the material before reaching the p-n junction. These effects produce an upper limit on the efficiency of silicon solar cells, currently around 12 to 15% for common modules and up to 25% for the best laboratory cells (about 30% is the theoretical maximum efficiency for single band gap solar cells, see Shockley-Queisser limit.).
By far the biggest problem with the conventional approach is cost; solar cells require a relatively thick layer of doped silicon in order to have reasonable photon capture rates, and silicon processing is expensive. There have been a number of different approaches to reduce this cost over the last decade, notably the thin-film approaches, but to date they have seen limited application due to a variety of practical problems. Another line of research has been to dramatically improve efficiency through the multi-junction approach, although these cells are very high cost and suitable only for large commercial deployments. In general terms the types of cells suitable for rooftop deployment have not changed significantly in efficiency, although costs have dropped somewhat due to increased supply.
Dye-sensitized solar cells
In the late 1960s it was discovered that illuminated organic dyes can generate electricity at oxide electrodes in electrochemical cells . In an effort to understand and simulate the primary processes in photosynthesis the phenomenon was studied at the University of Berkeley with chlorophyll extracted from spinach (bio-mimetic or bionic approach) . On the basis of such experiments electric power generation via the dye sensitization solar cell (DSSC) principle was demonstrated and discussed in 1972 . The instability of the dye solar cell was identified as a main challenge. Its efficiency could, during the following two decades, be improved by optimizing the porosity of the electrode prepared from fine oxide powder, but the instability remained a problem . A modern DSSC, the Graetzel cell, is composed of a porous layer of titanium dioxideTitanium dioxide
Titanium dioxide, also known as titanium oxide or titania, is the naturally occurring oxide of titanium, chemical formula . When used as a pigment, it is called titanium white, Pigment White 6, or CI 77891. Generally it comes in two different forms, rutile and anatase. It has a wide range of...
nanoparticle
Nanoparticle
In nanotechnology, a particle is defined as a small object that behaves as a whole unit in terms of its transport and properties. Particles are further classified according to size : in terms of diameter, coarse particles cover a range between 10,000 and 2,500 nanometers. Fine particles are sized...
s, covered with a molecular dye that absorbs sunlight, like the chlorophyll
Chlorophyll
Chlorophyll is a green pigment found in almost all plants, algae, and cyanobacteria. Its name is derived from the Greek words χλωρος, chloros and φύλλον, phyllon . Chlorophyll is an extremely important biomolecule, critical in photosynthesis, which allows plants to obtain energy from light...
in green leaves. The titanium dioxide is immersed under an electrolyte
Electrolyte
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible....
solution, above which is a platinum
Platinum
Platinum is a chemical element with the chemical symbol Pt and an atomic number of 78. Its name is derived from the Spanish term platina del Pinto, which is literally translated into "little silver of the Pinto River." It is a dense, malleable, ductile, precious, gray-white transition metal...
-based catalyst. As in a conventional alkaline battery
Alkaline battery
Alkaline batteries are a type of primary batteries dependent upon the reaction between zinc and manganese dioxide . A rechargeable alkaline battery allows reuse of specially designed cells....
, an anode
Anode
An anode is an electrode through which electric current flows into a polarized electrical device. Mnemonic: ACID ....
(the titanium dioxide) and a cathode
Cathode
A cathode is an electrode through which electric current flows out of a polarized electrical device. Mnemonic: CCD .Cathode polarity is not always negative...
(the platinum) are placed on either side of a liquid conductor (the electrolyte).
Sunlight passes through the transparent electrode into the dye layer where it can excite electrons that then flow into the titanium dioxide. The electrons flow toward the transparent electrode where they are collected for powering a load. After flowing through the external circuit, they are re-introduced into the cell on a metal electrode on the back, flowing into the electrolyte. The electrolyte then transports the electrons back to the dye molecules.
Dye-sensitized solar cells separate the two functions provided by silicon in a traditional cell design. Normally the silicon acts as both the source of photoelectrons, as well as providing the electric field to separate the charges and create a current. In the dye-sensitized solar cell, the bulk of the semiconductor is used solely for charge transport, the photoelectrons are provided from a separate photosensitive dye
Dye
A dye is a colored substance that has an affinity to the substrate to which it is being applied. The dye is generally applied in an aqueous solution, and requires a mordant to improve the fastness of the dye on the fiber....
. Charge separation occurs at the surfaces between the dye, semiconductor and electrolyte.
The dye molecules are quite small (nanometer sized), so in order to capture a reasonable amount of the incoming light the layer of dye molecules needs to be made fairly thick, much thicker than the molecules themselves. To address this problem, a nanomaterial is used as a scaffold to hold large numbers of the dye molecules in a 3-D matrix, increasing the number of molecules for any given surface area of cell. In existing designs, this scaffolding is provided by the semiconductor material, which serves double-duty.
Construction
In the case of the original Grätzel and O'Regan design, the cell has 3 primary parts. On top is a transparent anodeAnode
An anode is an electrode through which electric current flows into a polarized electrical device. Mnemonic: ACID ....
made of fluoride-doped tin dioxide
Tin dioxide
Tin dioxide is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin. With many other names , this oxide of tin is the most important raw material in tin chemistry...
(SnO2:F) deposited on the back of a (typically glass) plate. On the back of this conductive plate is a thin layer of titanium dioxide
Titanium dioxide
Titanium dioxide, also known as titanium oxide or titania, is the naturally occurring oxide of titanium, chemical formula . When used as a pigment, it is called titanium white, Pigment White 6, or CI 77891. Generally it comes in two different forms, rutile and anatase. It has a wide range of...
(TiO2), which forms into a highly porous structure with an extremely high surface area
Surface area
Surface area is the measure of how much exposed area a solid object has, expressed in square units. Mathematical description of the surface area is considerably more involved than the definition of arc length of a curve. For polyhedra the surface area is the sum of the areas of its faces...
. TiO2 only absorbs a small fraction of the solar photons (those in the UV). The plate is then immersed in a mixture of a photosensitive ruthenium
Ruthenium
Ruthenium is a chemical element with symbol Ru and atomic number 44. It is a rare transition metal belonging to the platinum group of the periodic table. Like the other metals of the platinum group, ruthenium is inert to most chemicals. The Russian scientist Karl Ernst Claus discovered the element...
-polypyridine
Pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula C5H5N. It is structurally related to benzene, with one C-H group replaced by a nitrogen atom...
dye
Dye
A dye is a colored substance that has an affinity to the substrate to which it is being applied. The dye is generally applied in an aqueous solution, and requires a mordant to improve the fastness of the dye on the fiber....
(also called molecular sensitizers) and a solvent
Solvent
A solvent is a liquid, solid, or gas that dissolves another solid, liquid, or gaseous solute, resulting in a solution that is soluble in a certain volume of solvent at a specified temperature...
. After soaking the film in the dye solution, a thin layer of the dye is left covalently bonded to the surface of the TiO2.
A separate plate is then made with a thin layer of the iodide
Iodide
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. This page is for the iodide ion and its salts. For information on organoiodides, see organohalides. In everyday life, iodide is most commonly encountered as a component of iodized salt,...
electrolyte spread over a conductive sheet, typically platinum
Platinum
Platinum is a chemical element with the chemical symbol Pt and an atomic number of 78. Its name is derived from the Spanish term platina del Pinto, which is literally translated into "little silver of the Pinto River." It is a dense, malleable, ductile, precious, gray-white transition metal...
metal. The two plates are then joined and sealed together to prevent the electrolyte from leaking. The construction is simple enough that there are hobby kits available to hand-construct them. Although they use a number of "advanced" materials, these are inexpensive compared to the silicon needed for normal cells because they require no expensive manufacturing steps. TiO2, for instance, is already widely used as a paint base.
Operation
Sunlight enters the cell through the transparent SnO2:F top contact, striking the dye on the surface of the TiO2. Photons striking the dye with enough energy to be absorbed create an excited state of the dye, from which an electron can be "injected" directly into the conduction band of the TiO2. From there it moves by diffusionDiffusion
Molecular diffusion, often called simply diffusion, is the thermal motion of all particles at temperatures above absolute zero. The rate of this movement is a function of temperature, viscosity of the fluid and the size of the particles...
(as a result of an electron concentration gradient
Gradient
In vector calculus, the gradient of a scalar field is a vector field that points in the direction of the greatest rate of increase of the scalar field, and whose magnitude is the greatest rate of change....
) to the clear anode
Anode
An anode is an electrode through which electric current flows into a polarized electrical device. Mnemonic: ACID ....
on top.
Meanwhile, the dye molecule has lost an electron and the molecule will decompose if another electron is not provided. The dye strips one from iodide
Iodide
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. This page is for the iodide ion and its salts. For information on organoiodides, see organohalides. In everyday life, iodide is most commonly encountered as a component of iodized salt,...
in electrolyte below the TiO2, oxidizing it into triiodide
Triiodide
In chemistry, triiodide can have several meanings. Triiodide primarily refers to the triiodide ion, I3−, a polyatomic anion composed of three iodine atoms. For some chemical compounds, triiodide indicates a salt of the named cation with the triiodide anion. Examples include sodium triiodide, ...
. This reaction occurs quite quickly compared to the time that it takes for the injected electron to recombine with the oxidized dye molecule, preventing this recombination reaction that would effectively short-circuit the solar cell.
The triiodide then recovers its missing electron by mechanically diffusing to the bottom of the cell, where the counter electrode re-introduces the electrons after flowing through the external circuit.
Efficiency
Several important measures are used to characterize solar cells. The most obvious is the total amount of electrical power produced for a given amount of solar power shining on the cell. Expressed as a percentage, this is known as the solar conversion efficiency. Electrical power is the product of current and voltage, so the maximum values for these measurements are important as well, Jsc and Voc respectively. Finally, in order to understand the underlying physics, the "quantum efficiency" is used to compare the chance that one photon (of a particular energy) will create one electron.In quantum efficiency
Quantum efficiency
Quantum efficiency is a quantity defined for a photosensitive device such as photographic film or a charge-coupled device as the percentage of photons hitting the photoreactive surface that will produce an electron–hole pair. It is an accurate measurement of the device's electrical sensitivity to...
terms, DSSCs are extremely efficient. Due to their "depth" in the nanostructure there is a very high chance that a photon will be absorbed, and the dyes are very effective at converting them to electrons. Most of the small losses that do exist in DSSC's are due to conduction losses in the TiO2 and the clear electrode, or optical losses in the front electrode. The overall quantum efficiency for green light is about 90%, with the "lost" 10% being largely accounted for by the optical losses in top electrode. The quantum efficiency of traditional designs vary, depending on their thickness, but are about the same as the DSSC.
In theory, the maximum voltage generated by such a cell is simply the difference between the (quasi-)Fermi level
Fermi energy
The Fermi energy is a concept in quantum mechanics usually referring to the energy of the highest occupied quantum state in a system of fermions at absolute zero temperature....
of the TiO2 and the redox potential of the electrolyte, about 0.7 V under solar illumination conditions (Voc). That is, if an illuminated DSSC is connected to a voltmeter in an "open circuit", it would read about 0.7 V. In terms of voltage, DSSCs offer slightly higher Voc than silicon, about 0.7 V compared to 0.6 V. This is a fairly small difference, so real-world differences are dominated by current production, Jsc.
Although the dye is highly efficient at converting absorbed photons into free electrons in the TiO2, only photons absorbed by the dye ultimately produce current. The rate of photon absorption depends upon the absorption spectrum of the sensitized TiO2 layer and upon the solar flux spectrum. The overlap between these two spectra determines the maximum possible photocurrent. Typically used dye molecules generally have poorer absorption in the red part of the spectrum compared to silicon, which means that fewer of the photons in sunlight are usable for current generation. These factors limit the current generated by a DSSC, for comparison, a traditional silicon-based solar cell offers about 35 mA
Ampere
The ampere , often shortened to amp, is the SI unit of electric current and is one of the seven SI base units. It is named after André-Marie Ampère , French mathematician and physicist, considered the father of electrodynamics...
/cm2, whereas current DSSCs offer about 20 mA/cm2.
Overall peak power production efficiency for current DSSCs is about 11%.
Degradation
DSSCs degrade when exposed to ultravioletUltraviolet
Ultraviolet light is electromagnetic radiation with a wavelength shorter than that of visible light, but longer than X-rays, in the range 10 nm to 400 nm, and energies from 3 eV to 124 eV...
radiation. The barrier layer may include UV stabilizers and/or UV absorbing luminescent
Luminescence
Luminescence is emission of light by a substance not resulting from heat; it is thus a form of cold body radiation. It can be caused by chemical reactions, electrical energy, subatomic motions, or stress on a crystal. This distinguishes luminescence from incandescence, which is light emitted by a...
chromophore
Chromophore
A chromophore is the part of a molecule responsible for its color. The color arises when a molecule absorbs certain wavelengths of visible light and transmits or reflects others. The chromophore is a region in the molecule where the energy difference between two different molecular orbitals falls...
s (which emit at longer wavelengths) and antioxidant
Antioxidant
An antioxidant is a molecule capable of inhibiting the oxidation of other molecules. Oxidation is a chemical reaction that transfers electrons or hydrogen from a substance to an oxidizing agent. Oxidation reactions can produce free radicals. In turn, these radicals can start chain reactions. When...
s to protect and improve the efficiency of the cell.
Advantages
DSSCs are currently the most efficient third-generation (2005 Basic Research Solar Energy Utilization 16) solar technology available. Other thin-film technologies are typically between 5% and 13%, and traditional low-cost commercial silicon panels operate between 12% and 15%. This makes DSSCs attractive as a replacement for existing technologies in "low density" applications like rooftop solar collectors, where the mechanical robustness and light weight of the glass-less collector is a major advantage. They may not be as attractive for large-scale deployments where higher-cost higher-efficiency cells are more viable, but even small increases in the DSSC conversion efficiency might make them suitable for some of these roles as well.There is another area where DSSCs are particularly attractive. The process of injecting an electron directly into the TiO2 is qualitatively different to that occurring in a traditional cell, where the electron is "promoted" within the original crystal. In theory, given low rates of production, the high-energy electron in the silicon could re-combine with its own hole, giving off a photon (or other form of energy) and resulting in no current being generated. Although this particular case may not be common, it is fairly easy for an electron generated in another molecule to hit a hole left behind in a previous photoexcitation.
In comparison, the injection process used in the DSSC does not introduce a hole in the TiO2, only an extra electron. Although it is energetically possible for the electron to recombine back into the dye, the rate at which this occurs is quite slow compared to the rate that the dye regains an electron from the surrounding electrolyte. Recombination directly from the TiO2 to species in the electrolyte is also possible although, again, for optimized devices this reaction is rather slow. On the contrary, electron transfer from the platinum coated electrode to species in the electrolyte is necessarily very fast.
As a result of these favorable "differential kinetics", DSSCs work even in low-light conditions. DSSCs are therefore able to work under cloudy skies and non-direct sunlight, whereas traditional designs would suffer a "cutout" at some lower limit of illumination, when charge carrier mobility is low and recombination becomes a major issue. The cutoff is so low they are even being proposed for indoor use, collecting energy for small devices from the lights in the house.
A practical advantage, one DSSCs share with most thin-film technologies, is that the cell's mechanical robustness indirectly leads to higher efficiencies in higher temperatures. In any semiconductor, increasing temperature will promote some electrons into the conduction band "mechanically". The fragility of traditional silicon cells requires them to be protected from the elements, typically by encasing them in a glass box similar to a greenhouse
Greenhouse
A greenhouse is a building in which plants are grown. These structures range in size from small sheds to very large buildings...
, with a metal backing for strength. Such systems suffer noticeable decreases in efficiency as the cells heat up internally. DSSCs are normally built with only a thin layer of conductive plastic on the front layer, allowing them to radiate away heat much easier, and therefore operate at lower internal temperatures.
Disadavantages
The major disadvantage to the DSSC design is the use of the liquid electrolyte, which has temperature stability problems. At low temperatures the electrolyte can freeze, ending power production and potentially leading to physical damage. Higher temperatures cause the liquid to expand, making sealing the panels a serious problem. The fact, that costly Ruthenium (dye), Platinum (catalyst) and conducting glass or plastic (contact) is needed in the Graetzel cell, has to be considered in cost projections. Another major drawback is the electrolyte solution, which contains volatile organic solvents and must be carefully sealed. This, along with the fact that the solvents permeate plastics, has precluded large-scale outdoor application and integration into flexible structure.Replacing the liquid electrolyte with a solid has been a major ongoing field of research. Recent experiments using solidified melted salts have shown some promise, but currently suffer from higher degradation during continued operation, and are not flexible.
Photocathodes and tandem cells
Grätzel’s cell operates as a photoanode (n-DSC), where photocurrent result from electron injection by the sensitized dye. Photocathodes (p-DSCs) operate in an inverse mode compared to the conventional n-DSC, where dye-excitation is followed by rapid electron transfer from a p-type semiconductor to the dye (dye-sensitized hole injection, instead of electron injection). Such p-DSCs and n-DSCs can be combined to construct tandem solar cells (pn-DSCs) and the theoretical efficiency of tandem DSCs is well beyond that of single-junction DSCs.A standard tandem cell consists of one n-DSC and one p-DSC in a simple sandwich configuration with an intermediate electrolyte layer. n-DSC and p-DSC are connected in series, which implies that the resulting photocurrent will be controlled by the weakest photoelectrode, whereas photovoltages are additive. Thus, photocurrent matching is very important for the construction of highly efficient tandem pn-DSCs. However, unlike n-DSCs, fast charge recombination following dye-sensitized hole injection usually resulted in low photocurrents in p-DSC and thus hampered the efficiency of the overall device.
Researchers have found that using dyes comprising a perylenemonoimid (PMI) as the acceptor and an oligothiophene coupled to triphenylamine as the donor greatly improve the performance of p-DSC by reducing charge recombination rate following dye-sensitized hole injection. The researchers constructed a tandem DSC device with NiO on the p-DSC side and TiO2 on the n-DSC side. Photocurrent matching was achieved through adjustment of NiO and TiO2 film thicknesses to control the optical absorptions and therefore match the photocurrents of both electrodes. The energy conversion efficiency of the device is 1.91%, which exceeds the efficiency of its individual components, but is still much lower than that of high performance n-DSC devices (6%–11%). The results are still promising since the tandem DSC was in itself rudimentary. The dramatic improvement in performance in p-DSC can eventually lead to tandem devices with much greater efficiency than lone n-DSCs.
Development
Infrared
Infrared light is electromagnetic radiation with a wavelength longer than that of visible light, measured from the nominal edge of visible red light at 0.74 micrometres , and extending conventionally to 300 µm...
light. The wide spectral response results in the dye having a deep brown-black color, and is referred to simply as "black dye". The dyes have an excellent chance of converting a photon into an electron, originally around 80% but improving to almost perfect conversion in more recent dyes, the overall efficiency is about 90%, with the "lost" 10% being largely accounted for by the optical losses in top electrode.
A solar cell must be capable of producing electricity for at least twenty years, without a significant decrease in efficiency (life span
Service life
A product's service life is its expected lifetime, or the acceptable period of use in service. It is the time that any manufactured item can be expected to be 'serviceable' or supported by its manufacturer....
). The "black dye" system was subjected to 50 million cycles, the equivalent of ten years' exposure to the sun in Switzerland. No discernible performance decrease was observed. However the dye is subject to breakdown in high-light situations. Over the last decade an extensive research program has been carried out to address these concerns. The newer dyes included 1-ethyl-3 methylimidazolium tetrocyanoborate [EMIB(CN)4] which is extremely light- and temperature-stable, copper-diselenium [Cu(In,GA)Se2] which offers higher conversion efficiencies, and others with varying special-purpose properties.
DSSCs are still at the start of their development cycle. Efficiency gains are possible and have recently started more widespread study. These include the use of quantum dot
Quantum dot
A quantum dot is a portion of matter whose excitons are confined in all three spatial dimensions. Consequently, such materials have electronic properties intermediate between those of bulk semiconductors and those of discrete molecules. They were discovered at the beginning of the 1980s by Alexei...
s for conversion of higher-energy (higher frequency) light into multiple electrons, using solid-state electrolytes for better temperature response, and changing the doping of the TiO2 to better match it with the electrolyte being used.
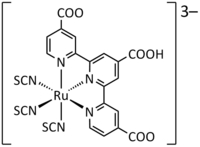
2003
A group of researchers at the Swiss Federal Institute of Technology has reportedly increased the thermostability of DSC by using amphiphilic ruthenium sensitizer in conjunction with quasi-solid-state gel electrolyte. The stability of the device matches that of a conventional inorganic silicon based solar cell. The cell sustained heating for 1,000 h at 80 °C.The group has previously prepared an ruthenium amphiphilic dye Z-907 (cis-Ru(H2dcbpy)(dnbpy)(NCS)2,where the ligand H2dcbpy is 4,4′-dicarboxylic acid-2,2′-bipyridine and dnbpy is 4,4′-dinonyl-2,2′-bipyridine) to increase dye tolerance to water in the electrolytes. In addition, the group also prepared a quasi-solid-state gel electrolye with a 3-methoxypropionitrile (MPN)-based liquid electrolyte that was solidified by a photochemically stable fluorine polymer, poly(vinylidenefluoride-co-hexafluoropropylene (PVDF-HFP).
The use of the amphiphilic Z-907 dye in conjunction with the polymer gel electrolyte in DSC achieved an energy conversion efficiency of 6.1%. More importantly, the device was stable under thermal stress and soaking with light. The high conversion efficiency of the cell was sustained after heating for 1,000 h at 80 °C, maintaining 94% of its initial value. After
accelerated testing in a solar simulator
Solar Simulator
A solar simulator is a device that provides illumination approximating natural sunlight. The purpose of the solar simulator is to provide a controllable indoor test facility under laboratory conditions, used for the testing of solar cells, sun screen, plastics, and other materials and...
for 1,000 h of light-soaking at 55 °C (100 mW cm−2) the efficiency had decreased by less than 5% for cells covered with an ultraviolet absorbing polymer film. These results are well within the limit for that of traditional inorganic silicon solar cells.
The enhanced performance may arise from a decrease in solvent permeation across the sealant due to the application of the polymer gel electrolyte. The polymer gel electrolyte is quasi-solid at room temperature, and becomes a viscous liquid (viscosity: 4.34 mPa·s) at 80 °C compared with the traditional liquid electrolyte (viscosity: 0.91 mPa·s). The much improved stabilities of the device under both thermal stress and soaking with light has never before been seen in DSCs, and they match the durability criteria applied to solar cells for outdoor use, which makes these devices viable for practical application.
2006
The first successful solid-hybrid dye-sensitized solar cells were reported.To improve electron transport in these solar cells, while maintaining the high surface area needed for dye adsorption, two researchers have designed alternate semiconductor morphologies, such as arrays of nanowire
Nanowire
A nanowire is a nanostructure, with the diameter of the order of a nanometer . Alternatively, nanowires can be defined as structures that have a thickness or diameter constrained to tens of nanometers or less and an unconstrained length. At these scales, quantum mechanical effects are important —...
s and a combination of nanowires and nanoparticle
Nanoparticle
In nanotechnology, a particle is defined as a small object that behaves as a whole unit in terms of its transport and properties. Particles are further classified according to size : in terms of diameter, coarse particles cover a range between 10,000 and 2,500 nanometers. Fine particles are sized...
s, to provide a direct path to the electrode via the semiconductor conduction band. Such structures may provide a means to improve the quantum efficiency of DSSCs in the red region of the spectrum, where their performance is currently limited.
On August 2006, to prove the chemical and thermal robustness of the 1-ethyl-3 methylimidazolium tetracyanoborate solar cell, the researchers subjected the devices to heating at 80 °C in the dark for 1000 hours, followed by light soaking at 60 °C for 1000 hours. After dark heating and light soaking, 90% of the initial photovoltaic efficiency was maintained – the first time such excellent thermal stability has been observed for a liquid electrolyte that exhibits such a high conversion efficiency. Contrary to silicon solar cells, whose performance declines with increasing temperature, the dye-sensitized solar-cell devices were only negligibly influenced when increasing the operating temperature
Operating temperature
An operating temperature is the temperature at which an electrical or mechanical device operates. The device will operate effectively within a specified temperature range which varies based on the device function and application context, and ranges from the minimum operating temperature to the...
from ambient to 60 °C.
April 2007
Wayne Campbell at Massey UniversityMassey University
Massey University is one of New Zealand's largest universities with approximately 36,000 students, 20,000 of whom are extramural students.The University has campuses in Palmerston North , Wellington and Auckland . Massey offers most of its degrees extramurally within New Zealand and internationally...
, New Zealand, has experimented with a wide variety of organic dyes based on porphyrin
Porphyrin
Porphyrins are a group of organic compounds, many naturally occurring. One of the best-known porphyrins is heme, the pigment in red blood cells; heme is a cofactor of the protein hemoglobin. Porphyrins are heterocyclic macrocycles composed of four modified pyrrole subunits interconnected at...
. In nature, porphyrin is the basic building block of the hemoprotein
Hemoprotein
A hemeprotein , or heme protein, is a metalloprotein containing a heme prosthetic group- an organic compound that allows a protein to carry out a function that it cannot do alone....
s, which include chlorophyll
Chlorophyll
Chlorophyll is a green pigment found in almost all plants, algae, and cyanobacteria. Its name is derived from the Greek words χλωρος, chloros and φύλλον, phyllon . Chlorophyll is an extremely important biomolecule, critical in photosynthesis, which allows plants to obtain energy from light...
in plants and hemoglobin
Hemoglobin
Hemoglobin is the iron-containing oxygen-transport metalloprotein in the red blood cells of all vertebrates, with the exception of the fish family Channichthyidae, as well as the tissues of some invertebrates...
in animals. He reports efficiency on the order of 5.6% using these low-cost dyes.
June 2008
An article published in Nature Materials demonstrated cell efficiencies of 8.2% using a new solvent-free liquid redox electrolyte consisting of a melt of three salts, as an alternative to using organic solvents as an electrolyte solution. Although the efficiency with this electrolyte is less than the 11% being delivered using the existing iodine-based solutions, the team is confident the efficiency can be improved.2009
A group of researchers at Georgia Tech made dye-sensitized solar cells with a higher effective surface areaSurface area
Surface area is the measure of how much exposed area a solid object has, expressed in square units. Mathematical description of the surface area is considerably more involved than the definition of arc length of a curve. For polyhedra the surface area is the sum of the areas of its faces...
by wrapping the cells around a quartz
Quartz
Quartz is the second-most-abundant mineral in the Earth's continental crust, after feldspar. It is made up of a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall formula SiO2. There are many different varieties of quartz,...
optical fiber
Optical fiber
An optical fiber is a flexible, transparent fiber made of a pure glass not much wider than a human hair. It functions as a waveguide, or "light pipe", to transmit light between the two ends of the fiber. The field of applied science and engineering concerned with the design and application of...
. The researchers removed the cladding
Cladding (fiber optics)
Cladding is one or more layers of material of lower refractive index, in intimate contact with a core material of higher refractive index. The cladding causes light to be confined to the core of the fiber by total internal reflection at the boundary between the two. Light propagation in the...
from optical fibers, grew zinc oxide
Zinc oxide
Zinc oxide is an inorganic compound with the formula ZnO. It is a white powder that is insoluble in water. The powder is widely used as an additive into numerous materials and products including plastics, ceramics, glass, cement, rubber , lubricants, paints, ointments, adhesives, sealants,...
nanowire
Nanowire
A nanowire is a nanostructure, with the diameter of the order of a nanometer . Alternatively, nanowires can be defined as structures that have a thickness or diameter constrained to tens of nanometers or less and an unconstrained length. At these scales, quantum mechanical effects are important —...
s along the surface, treated them with dye molecules, surrounded the fibers by an electrolyte
Electrolyte
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible....
and a metal film that carries electrons off the fiber. The cells are six times more efficient than a zinc oxide cell with the same surface area. Photons bounce inside the fiber as they travel, so there are more chances to interact with the solar cell and produce more current. These devices only collect light at the tips, but future fiber cells could be made to absorb light along the entire length of the fiber, which would require a coating that is conductive
Electrical conductor
In physics and electrical engineering, a conductor is a material which contains movable electric charges. In metallic conductors such as copper or aluminum, the movable charged particles are electrons...
as well as transparent
Transparency and translucency
In the field of optics, transparency is the physical property of allowing light to pass through a material; translucency only allows light to pass through diffusely. The opposite property is opacity...
. Max Shtein of the University of Michigan
University of Michigan
The University of Michigan is a public research university located in Ann Arbor, Michigan in the United States. It is the state's oldest university and the flagship campus of the University of Michigan...
said a sun-tracking system
Solar tracker
A solar tracker is a generic term used to describe devices that orient various payloads toward the sun. Payloads can be photovoltaic panels, reflectors, lenses or other optical devices....
would not be necessary for such cells, and would work on cloudy days when light is diffuse.
2010
Researchers at the École Polytechnique Fédérale de LausanneÉcole polytechnique fédérale de Lausanne
The École polytechnique fédérale de Lausanne is one of the two Swiss Federal Institutes of Technology and is located in Lausanne, Switzerland.The school was founded by the Swiss Federal Government with the stated mission to:...
and at the Université du Québec à Montréal
Université du Québec à Montréal
The Université du Québec à Montréal is one of four universities in Montreal, Quebec, Canada.-Basic facts:The UQAM is the largest constituent element of the Université du Québec , a public university system with other branches in Gatineau , Rimouski, Rouyn-Noranda, Quebec City, Chicoutimi, and...
claim to have overcome two of the DSC's major issues:
- "new molecules" have been created for the electrolyteElectrolyteIn chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible....
, resulting in a liquid or gel that is transparent and non-corrosive, which can increase the photovoltage and improve the cell's output and stability. - At the cathodeCathodeA cathode is an electrode through which electric current flows out of a polarized electrical device. Mnemonic: CCD .Cathode polarity is not always negative...
, platinum was replaced by cobalt sulfideCobalt sulfideCobalt sulfide is the name for chemical compounds with a formula CoxSy. Well-characterized species include minerals with the formula CoS2 and Co3S4, and the synthetic material Co9S8...
, which is far less expensive, more efficient, more stable and easier to produce in the laboratory.
2011
DyesolDyesol
Dyesol is a maker of dye-sensitized solar cell materials and technology, including components,and research and development...
and Tata steel
Tata Steel
Tata Steel is a multinational steel company headquartered in Jamshedpur, India and part of Tata Group. It is the world's seventh-largest steel company, with an annual crude steel capacity of 31 million tonnes, and the largest private-sector steel company in India measured by domestic production...
announced in June the development of the world´s largest dye sensitized photovoltaic module, printed onto steel in a continuous line.
Dyesol
Dyesol
Dyesol is a maker of dye-sensitized solar cell materials and technology, including components,and research and development...
and Tata steel
Tata Steel
Tata Steel is a multinational steel company headquartered in Jamshedpur, India and part of Tata Group. It is the world's seventh-largest steel company, with an annual crude steel capacity of 31 million tonnes, and the largest private-sector steel company in India measured by domestic production...
announced in November the targeted development of Grid Parity Competitive BIPV solar steel that does not require government subsidised feed in tariffs. Dyesol-TATA Solar Steel Roofing is currently being installed on the Sustainable Building Envelope Centre (SBEC) in Shotton, Wales.
Market introduction
Several commercial providers are promising availability of DSCs in the near future:- DyesolDyesolDyesol is a maker of dye-sensitized solar cell materials and technology, including components,and research and development...
officially opened its new manufacturing facilities in Queanbeyan on the 7th of October 2008. It has subsequently announced partnerships with Tata Steel and Pilkington Glass for the development and large scale manufacture of DSC BIPV. Dyesol has also entered working relationships with Merck, Umicore, CSIRO, Japanese Ministry of Economy and Trade, Singapore Aerospace Manufacturing and TIMO Korea - Solaronix, a Swiss company specialized in the production of DSC materials since 1993, has extended their premises in 2010 to host a manufacturing pilot line of DSC modules.
- SolarPrint founded in 2008 by Dr. Mazhar Bari, Andre Fernon and Roy Horgan. SolarPrint is the first Ireland-based commercial entity involved in the manufacturing of PV technology. SolarPrint's innovation is the solution to the solvent based electrolyte which to date has prohibited the mass commercialisation of DSSC.
- G24innovations, founded in 2006, based in Cardiff, South Wales, UK. On October 17, 2007, claimed the production of the first commercial grade dye sensitised thin films.
- Hydrogen Solar is another company making dye-sensitized cells.
- Konarka, announced in 2002 that they were granted licensee rights to dye-sensitized solar cell technology from the Swiss Federal Institute of TechnologyÉcole polytechnique fédérale de LausanneThe École polytechnique fédérale de Lausanne is one of the two Swiss Federal Institutes of Technology and is located in Lausanne, Switzerland.The school was founded by the Swiss Federal Government with the stated mission to:...
(EPFL). - Aisin Seiki has worked with Toyota Central R&D Labs to develop dye-sensitized solar cells (DSC) for applications in cars and homes.
- Sony Corporation has developed dye-sensitized solar cells with an energy conversion efficiency of 10%, a level seen as necessary for commercial use. Sony has been supplied materials by Australia´s DyesolDyesolDyesol is a maker of dye-sensitized solar cell materials and technology, including components,and research and development...
See also
- Absorption
- Bronsted
- ChromophoreChromophoreA chromophore is the part of a molecule responsible for its color. The color arises when a molecule absorbs certain wavelengths of visible light and transmits or reflects others. The chromophore is a region in the molecule where the energy difference between two different molecular orbitals falls...
- Low-cost solar cell
- Luminescent solar concentrator
- ElutionElutionElution is a term used in analytical and organic chemistry to describe the process of extracting one material from another by washing with a solvent ....
- Organic photovoltaics
- Photoelectrochemical cellPhotoelectrochemical cellPhotoelectrochemical cells or PECs are solar cells which generate electrical energy from light, including visible light. Some photoelectrochemical cells simply produce electrical energy, while others produce hydrogen in a process similar to the electrolysis of water.-Photogeneration cell:In this...
- Solid-state solar cell
- Stationary phase
- Titanium dioxideTitanium dioxideTitanium dioxide, also known as titanium oxide or titania, is the naturally occurring oxide of titanium, chemical formula . When used as a pigment, it is called titanium white, Pigment White 6, or CI 77891. Generally it comes in two different forms, rutile and anatase. It has a wide range of...
External links
- Dye Solar Cells for Real, the assembly guide for making your own solar cells
- Breakthrough claimed by Korean researchers
- Breakthrough in low-cost efficient solar cells
- DSSC Manual (National Science FoundationNational Science FoundationThe National Science Foundation is a United States government agency that supports fundamental research and education in all the non-medical fields of science and engineering. Its medical counterpart is the National Institutes of Health...
) - How to Build Your Own Solar Cell, A Nanocrystalline Dye-Sensitized Solar Cell (DIY)
- Global picture for dye-sensitized solar cells.
- Solar Cells for Cheap – 2006 interview with the inventor Michael Grätzel at TechnologyReview
- Taking nature’s cue for cheaper solar power Wayne Campbell's research on using Porphyrin for highly efficient cells
- What on earth is a Grätzel solar cell, and why is it so important? Interview with Dr Wayne Campbell broadcast 21 April 2007
- A New Ingredient for Solar Cells – Raspberries
- International conference on the industrialisation of DSC.
- Material Safety Data Sheet for Dye N3.

