
Enzyme assay
Encyclopedia

Assay
An assay is a procedure in molecular biology for testing or measuring the activity of a drug or biochemical in an organism or organic sample. A quantitative assay may also measure the amount of a substance in a sample. Bioassays and immunoassays are among the many varieties of specialized...
s are laboratory
Laboratory
A laboratory is a facility that provides controlled conditions in which scientific research, experiments, and measurement may be performed. The title of laboratory is also used for certain other facilities where the processes or equipment used are similar to those in scientific laboratories...
methods for measuring enzymatic
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
activity. They are vital for the study of enzyme kinetics
Enzyme kinetics
Enzyme kinetics is the study of the chemical reactions that are catalysed by enzymes. In enzyme kinetics, the reaction rate is measured and the effects of varying the conditions of the reaction investigated...
and enzyme inhibition
Enzyme inhibitor
An enzyme inhibitor is a molecule that binds to enzymes and decreases their activity. Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors. They are also used as herbicides and pesticides...
.
Enzyme units
Amounts of enzymes can either be expressed as long as possible molarMole (unit)
The mole is a unit of measurement used in chemistry to express amounts of a chemical substance, defined as an amount of a substance that contains as many elementary entities as there are atoms in 12 grams of pure carbon-12 , the isotope of carbon with atomic weight 12. This corresponds to a value...
amounts, as with any other chemical, or measured in terms of activity, in enzyme unit
Enzyme unit
The enzyme unit is a unit for the amount of a particular enzyme.One U is defined as the amount of the enzyme that catalyzes the conversion of 1 micro mole of substrate per minute...
s.
Enzyme activity
Enzyme activity = moles of substrate converted per unit time = rate × reaction volume. Enzyme activity is a measure of the quantity of active enzyme present and is thus dependent on conditions, which should be specified. The SI unit is the katalKatal
The katal is the SI unit of catalytic activity. It is a derived SI unit for expressing quantity values of catalytic activity of enzymes and other catalysts. Its use is recommended by the General Conference on Weights and Measures and other international organizations. It replaces the non-SI enzyme...
, 1 katal = 1 mol s−1, but this is an excessively large unit. A more practical and commonly used value is 1 enzyme unit
Enzyme unit
The enzyme unit is a unit for the amount of a particular enzyme.One U is defined as the amount of the enzyme that catalyzes the conversion of 1 micro mole of substrate per minute...
(U) = 1 μmol min−1. 1 U corresponds to 16.67 nano
Nano
Nano- is a prefix meaning a billionth. Used primarily in the metric system, this prefix denotes a factor of 10−9 or . It is frequently encountered in science and electronics for prefixing units of time and length, such as 30 nanoseconds , 100 nanometres or in the case of electrical capacitance,...
katals.
Enzyme activity as given in katal generally refers to that of the assumed natural target substrate of the enzyme. Enzyme activity can also be given as that of certain standardized substrates, such as gelatin
Gelatin
Gelatin is a translucent, colorless, brittle , flavorless solid substance, derived from the collagen inside animals' skin and bones. It is commonly used as a gelling agent in food, pharmaceuticals, photography, and cosmetic manufacturing. Substances containing gelatin or functioning in a similar...
, then measured in gelatin digesting units (GDU), or milk proteins, then measured in milk clotting units (MCU). The units GDU and MCU are based on how fast one gram of the enzyme will digest gelatin or milk proteins, respectively. 1 GDU equals approximately 1.5 MCU.
Specific activity
The specific activity of an enzyme is another common unit. This is the activity of an enzyme per milligram of total protein (expressed in μmol min−1mg−1). Specific activity gives a measurement of the activity of the enzyme. It is the amount of product formed by an enzymeEnzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
in a given amount of time under given conditions per milligram of total protein. Specific activity is equal to the rate of reaction multiplied by the volume of reaction divided by the mass of total protein. The SI unit is katal kg−1, but a more practical unit is μmol mg−1 min−1. Specific activity is a measure of enzyme processivity, at a specific (usually saturating)substrate concentration, and is usually constant for a pure enzyme. For elimination of errors arising from differences in cultivation batches and/or misfolded enzyme etc. an active site titration needs to be done. This is a measure of the amount of active enzyme, calculated by e.g. titrating the amount of active sites present by employing an irreversible inhibitor. The specific activity should then be expressed as μmol min−1 mg−1 active enzyme.
Related terminology
The rate of a reaction is the concentration of substrate disappearing (or product produced) per unit time (mol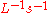 ).
).The % purity is 100% × (specific activity of enzyme sample / specific activity of pure enzyme). The impure sample has lower specific activity because some of the mass is not actually enzyme. If the specific activity of 100% pure enzyme is known, then an impure sample will have a lower specific activity, allowing purity to be calculated.
Types of assay
All enzyme assays measure either the consumption of substrate or production of product over time. A large number of different methods of measuring the concentrations of substrates and products exist and many enzymes can be assayed in several different ways. Biochemists usually study enzyme-catalysed reactions using four types of experiments:- Initial rate experiments. When an enzyme is mixed with a large excess of the substrate, the enzyme-substrate intermediate builds up in a fast initial transient. Then the reaction achieves a steady-state kinetics in which enzyme substrate intermediates remains approximately constant over time and the reaction rate changes relatively slowly. Rates are measured for a short period after the attainment of the quasi-steady state, typically by monitoring the accumulation of product with time. Because the measurements are carried out for a very short period and because of the large excess of substrate, the approximation free substrate is approximately equal to the initial substrate can be made. The initial rate experiment is the simplest to perform and analyze, being relatively free from complications such as back-reaction and enzyme degradation. It is therefore by far the most commonly used type of experiment in enzyme kinetics.
- Progress curve experiments. In these experiments, the kinetic parameters are determined from expressions for the species concentrations as a function of time. The concentration of the substrate or product is recorded in time after the initial fast transient and for a sufficiently long period to allow the reaction to approach equilibrium. We note in passing that, while they are less common now, progress curve experiments were widely used in the early period of enzyme kinetics.
- Transient kinetics experiments. In these experiments, reaction behaviour is tracked during the initial fast transient as the intermediate reaches the steady-state kinetics period. These experiments are more difficult to perform than either of the above two classes because they require specialist techniques (such as flash photolysisFlash photolysisFlash photolysis is a pump-probe laboratory technique, in which a sample is firstly excited by a strong pulse of light from a laser of nanosecond, picosecond, or femtosecond pulse width or by a short-pulse light source such as a flash lamp...
of caged compounds) or rapid mixing (such as stopped-flowStopped flowA stopped flow instrument is a rapid mixing device used to study the chemical kinetics of a reaction in solution. After two or more solutions containing the reagents are mixed, they are studied by whatever experimental methods are deemed suitable. Different forms of spectroscopy and scattering of...
, quenched flow or continuous flow).
- Relaxation experiments. In these experiments, an equilibrium mixture of enzyme, substrate and product is perturbed, for instance by a temperatureTemperature JumpTemperature jump is a technique used in the study of chemical kinetics. It usually involves the discharging of a capacitor through a small volume Temperature jump is a technique used in the study of chemical kinetics. It usually involves the discharging of a capacitor (in the kV range) through a...
, pressurePressure jumpPressure jump is a technique used in the study of chemical kinetics. It involves making rapid changes to the pressure of an experimental system and observing the return to equilibrium or steady state...
or pH jump, and the return to equilibrium is monitored. The analysis of these experiments requires consideration of the fully reversible reaction. Moreover, relaxation experiments are relatively insensitive to mechanistic details and are thus not typically used for mechanism identification, although they can be under appropriate conditions.
Enzyme assays can be split into two groups according to their sampling method: continuous assays, where the assay gives a continuous reading of activity, and discontinuous assays, where samples are taken, the reaction stopped and then the concentration of substrates/products determined.

Continuous assays
Continuous assays are most convenient, with one assay giving the rate of reaction with no further work necessary. There are many different types of continuous assays.Spectrophotometric
In spectrophotometricUltraviolet-visible spectroscopy
Ultraviolet-visible spectroscopy or ultraviolet-visible spectrophotometry refers to absorption spectroscopy or reflectance spectroscopy in the ultraviolet-visible spectral region. This means it uses light in the visible and adjacent ranges...
assays, you follow the course of the reaction by measuring a change in how much light the assay solution absorbs. If this light is in the visible region you can actually see a change in the color of the assay, these are called colorimetric assays. The MTT assay
MTT assay
The MTT assay and the MTS assay are colorimetric assays for measuring the activity of enzymes that reduce MTT or close dyes to formazan dyes, giving a purple color. A main application allows to assess the viability and the proliferation of cells...
, a redox assay using a tetrazolium dye as substrate is an example of a colorimetric assay.
UV light is often used, since the common coenzymes NADH
Nicotinamide adenine dinucleotide
Nicotinamide adenine dinucleotide, abbreviated NAD, is a coenzyme found in all living cells. The compound is a dinucleotide, since it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine base and the other nicotinamide.In metabolism, NAD is involved...
and NADPH
Nicotinamide adenine dinucleotide
Nicotinamide adenine dinucleotide, abbreviated NAD, is a coenzyme found in all living cells. The compound is a dinucleotide, since it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine base and the other nicotinamide.In metabolism, NAD is involved...
absorb UV light in their reduced
Redox
Redox reactions describe all chemical reactions in which atoms have their oxidation state changed....
forms, but do not in their oxidized
Redox
Redox reactions describe all chemical reactions in which atoms have their oxidation state changed....
forms. An oxidoreductase
Oxidoreductase
In biochemistry, an oxidoreductase is an enzyme that catalyzes the transfer of electrons from one molecule to another...
using NADH as a substrate could therefore be assayed by following the decrease in UV absorbance at a wavelength of 340 nm as it consumes the coenzyme.
Direct versus coupled assays
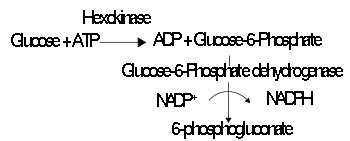
Hexokinase
A hexokinase is an enzyme that phosphorylates a six-carbon sugar, a hexose, to a hexose phosphate. In most tissues and organisms, glucose is the most important substrate of hexokinases, and glucose-6-phosphate the most important product....
, which can be assayed by coupling its production of glucose-6-phosphate to NADPH production, using glucose-6-phosphate dehydrogenase
Glucose-6-phosphate dehydrogenase
Glucose-6-phosphate dehydrogenase is a cytosolic enzyme in the pentose phosphate pathway , a metabolic pathway that supplies reducing energy to cells by maintaining the level of the co-enzyme nicotinamide adenine dinucleotide phosphate...
.
Fluorometric
FluorescenceFluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation of a different wavelength. It is a form of luminescence. In most cases, emitted light has a longer wavelength, and therefore lower energy, than the absorbed radiation...
is when a molecule emits light of one wavelength
Wavelength
In physics, the wavelength of a sinusoidal wave is the spatial period of the wave—the distance over which the wave's shape repeats.It is usually determined by considering the distance between consecutive corresponding points of the same phase, such as crests, troughs, or zero crossings, and is a...
after absorbing light of a different wavelength. Fluorometric assays use a difference in the fluorescence
Fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation of a different wavelength. It is a form of luminescence. In most cases, emitted light has a longer wavelength, and therefore lower energy, than the absorbed radiation...
of substrate from product to measure the enzyme reaction. These assays are in general much more sensitive than spectrophotometric assays, but can suffer from interference caused by impurities and the instability of many fluorescent compounds when exposed to light.
An example of these assays is again the use of the nucleotide coenzymes NADH and NADPH. Here, the reduced forms are fluorescent and the oxidised forms non-fluorescent. Oxidation reactions can therefore be followed by a decrease in fluorescence and reduction reactions by an increase. Synthetic substrates that release a fluorescent dye in an enzyme-catalyzed reaction are also available, such as 4-methylumbelliferyl-β-D-galactoside for assaying β-galactosidase.
Calorimetric
Calorimetry
Calorimetry is the science of measuring the heat of chemical reactions or physical changes. Calorimetry is performed with a calorimeter. The word calorimetry is derived from the Latin word calor, meaning heat...
is the measurement of the heat released or absorbed by chemical reactions. These assays are very general, since many reactions involve some change in heat and with use of a microcalorimeter, not much enzyme or substrate is required. These assays can be used to measure reactions that are impossible to assay in any other way.
Chemiluminescent
Chemiluminescence is the emission of light by a chemical reaction. Some enzyme reactions produce light and this can be measured to detect product formation. These types of assay can be extremely sensitive, since the light produced can be captured by photographic film over days or weeks, but can be hard to quantify, because not all the light released by a reaction will be detected.The detection of horseradish peroxidase
Peroxidase
Peroxidases are a large family of enzymes that typically catalyze a reaction of the form:For many of these enzymes the optimal substrate is hydrogen peroxide, but others are more active with organic hydroperoxides such as lipid peroxides...
by enzymatic chemiluminescence (ECL) is a common method of detecting antibodies in western blotting. Another example is the enzyme luciferase
Luciferase
Luciferase is a generic term for the class of oxidative enzymes used in bioluminescence and is distinct from a photoprotein. One famous example is the firefly luciferase from the firefly Photinus pyralis. "Firefly luciferase" as a laboratory reagent usually refers to P...
, this is found in fireflies and naturally produces light from its substrate luciferin.
Light Scattering
Static light scatteringStatic light scattering
Static light scattering is a technique in physical chemistry that measures the intensity of the scattered light to obtain the average molecular weight Mw of a macromolecule like a polymer or a protein. Measurement of the scattering intensity at many angles allows calculation of the root mean square...
measures the product of weight-averaged molar mass and concentration of macromolecules in solution. Given a fixed total concentration of one or more species over the measurement time, the scattering signal is a direct measure of the weight-averaged molar mass of the solution, which will vary as complexes form or dissociate.
Hence the measurement quantifies the stoichiometry of the complexes as well as kinetics. Light scattering assays of protein kinetics is a very general technique that does not require an enzyme.
Microscale Thermophoresis
Microscale ThermophoresisMicroscale Thermophoresis
Microscale Thermophoresis is a technology for the analysis of biomolecules. Microscale Thermophoresis is the directed movement of particles in a microscopic temperature gradient...
(MST) measures the size, charge and hydration entropy of molecules/substrates in real time. The thermophoretic movement of a fluorescently labeled substrate changes significantly as it is modified by an enzyme. This enzymatic activity can be measured with high time resolution in real time. The material consumption of the all optical MST method is very low, only 5 µl sample volume and 10nM enzyme concentration are needed to measure the enzymatic rate constants for activity and inhibition. MST allows to measure the modification of two different substrates at once (multiplexing
Multiplexing
The multiplexed signal is transmitted over a communication channel, which may be a physical transmission medium. The multiplexing divides the capacity of the low-level communication channel into several higher-level logical channels, one for each message signal or data stream to be transferred...
) if both substrates are labeled with different fluorophores. Thus substrate competition experiments can be performed.
Discontinuous assays
Discontinuous assays are when samples are taken from an enzyme reaction at intervals and the amount of product production or substrate consumption is measured in these samples.Radiometric
Radiometric assays measure the incorporation of radioactivity into substrates or its release from substrates. The radioactive isotopes most frequently used in these assays are 14C, 32P, 35S and 125I. Since radioactive isotopes can allow the specific labelling of a single atom of a substrate, these assays are both extremely sensitive and specific. They are frequently used in biochemistry and are often the only way of measuring a specific reaction in crude extracts (the complex mixtures of enzymes produced when you lyse cells).Radioactivity is usually measured in these procedures using a scintillation counter
Scintillation counter
A scintillation counter measures ionizing radiation. The sensor, called a scintillator, consists of a transparent crystal, usually phosphor, plastic , or organic liquid that fluoresces when struck by ionizing radiation. A sensitive photomultiplier tube measures the light from the crystal...
.
Chromatographic
Chromatographic assays measure product formation by separating the reaction mixture into its components by chromatographyChromatography
Chromatography is the collective term for a set of laboratory techniques for the separation of mixtures....
. This is usually done by high-performance liquid chromatography
High-performance liquid chromatography
High-performance liquid chromatography , HPLC, is a chromatographic technique that can separate a mixture of compounds and is used in biochemistry and analytical chemistry to identify, quantify and purify the individual components of the mixture.HPLC typically utilizes different types of stationary...
(HPLC), but can also use the simpler technique of thin layer chromatography
Thin layer chromatography
Thin layer chromatography is a chromatography technique used to separate mixtures. Thin layer chromatography is performed on a sheet of glass, plastic, or aluminum foil, which is coated with a thin layer of adsorbent material, usually silica gel, aluminium oxide, or cellulose...
. Although this approach can need a lot of material, its sensitivity can be increased by labelling the substrates/products with a radioactive or fluorescent tag. Assay sensitivity has also been increased by switching protocols to improved chromatographic instruments (e.g. ultra-high pressure liquid chromatography) that operate at pump pressure a few-fold higher than HPLC instruments (see High-performance liquid chromatography#Pump pressure).
Factors to control in assays
- Salt Concentration: Most enzymes cannot tolerate extremely high salt concentrations. The ions interfere with the weak ionic bonds of proteins. Typical enzymes are active in salt concentrations of 1-500 mM. As usual there are exceptions such as the halophilic (salt loving) algaeAlgaeAlgae are a large and diverse group of simple, typically autotrophic organisms, ranging from unicellular to multicellular forms, such as the giant kelps that grow to 65 meters in length. They are photosynthetic like plants, and "simple" because their tissues are not organized into the many...
and bacteriaBacteriaBacteria are a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria have a wide range of shapes, ranging from spheres to rods and spirals...
.
- Effects of Temperature: All enzymes work within a range of temperature specific to the organism. Increases in temperature generally lead to increases in reaction rates. There is a limit to the increase because higher temperatures lead to a sharp decrease in reaction rates. This is due to the denaturating (alteration) of proteinProteinProteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. A polypeptide is a single linear polymer chain of amino acids bonded together by peptide bonds between the carboxyl and amino groups of...
structure resulting from the breakdown of the weak ionicIonic bondAn ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. Pure ionic bonding cannot exist: all ionic compounds have some...
and hydrogen bonding that stabilize the three dimensional structure of the enzyme active site. The "optimum" temperature for human enzymes is usually between 35 and 40 °C. The average temperature for humans is 37 °C. Human enzymes start to denature quickly at temperatures above 40 °C. Enzymes from thermophilic archaeaArchaeaThe Archaea are a group of single-celled microorganisms. A single individual or species from this domain is called an archaeon...
found in the hot springs are stable up to 100 °C. However, the idea of an "optimum" rate of an enzyme reaction is misleading, as the rate observed at any temperature is the product of two rates, the reaction rate and the denaturation rate. If you were to use an assay measuring activity for one second, it would give high activity at high temperatures, however if you were to use an assay measuring product formation over an hour, it would give you low activity at these temperatures.
- Effects of pH: Most enzymes are sensitive to pHPHIn chemistry, pH is a measure of the acidity or basicity of an aqueous solution. Pure water is said to be neutral, with a pH close to 7.0 at . Solutions with a pH less than 7 are said to be acidic and solutions with a pH greater than 7 are basic or alkaline...
and have specific ranges of activity. All have an optimum pH. The pH can stop enzyme activity by denaturating (altering) the three dimensional shape of the enzyme by breaking ionicIonic bondAn ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. Pure ionic bonding cannot exist: all ionic compounds have some...
, and hydrogen bondHydrogen bondA hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, such as nitrogen, oxygen or fluorine, that comes from another molecule or chemical group. The hydrogen must be covalently bonded to another electronegative atom to create the bond...
s. Most enzymes function between a pH of 6 and 8; however pepsin in the stomach works best at a pH of 2 and trypsin at a pH of 8.
- Substrate Saturation: Increasing the substrateSubstrate (biochemistry)In biochemistry, a substrate is a molecule upon which an enzyme acts. Enzymes catalyze chemical reactions involving the substrate. In the case of a single substrate, the substrate binds with the enzyme active site, and an enzyme-substrate complex is formed. The substrate is transformed into one or...
concentration increases the rate of reaction (enzyme activity). However, enzyme saturation limits reaction rates. An enzyme is saturated when the active sites of all the molecules are occupied most of the time. At the saturation point, the reaction will not speed up, no matter how much additional substrate is added. The graph of the reaction rate will plateau.
- Level of crowding, large amounts of macromoleculeMacromoleculeA macromolecule is a very large molecule commonly created by some form of polymerization. In biochemistry, the term is applied to the four conventional biopolymers , as well as non-polymeric molecules with large molecular mass such as macrocycles...
s in a solution will alter the ratesReaction rateThe reaction rate or speed of reaction for a reactant or product in a particular reaction is intuitively defined as how fast or slow a reaction takes place...
and equilibrium constants of enzyme reactions, through an effect called macromolecular crowdingMacromolecular crowdingThe phenomenon of macromolecular crowding alters the properties of molecules in a solution when high concentrations of macromolecules such as proteins are present. Such conditions occur routinely in living cells; for instance, the cytosol of Escherichia coli contains about 300–400 milligrammes per...
.

