
Fluorescence
Encyclopedia

Electromagnetic radiation
Electromagnetic radiation is a form of energy that exhibits wave-like behavior as it travels through space...
of a different wavelength
Wavelength
In physics, the wavelength of a sinusoidal wave is the spatial period of the wave—the distance over which the wave's shape repeats.It is usually determined by considering the distance between consecutive corresponding points of the same phase, such as crests, troughs, or zero crossings, and is a...
. It is a form of luminescence
Luminescence
Luminescence is emission of light by a substance not resulting from heat; it is thus a form of cold body radiation. It can be caused by chemical reactions, electrical energy, subatomic motions, or stress on a crystal. This distinguishes luminescence from incandescence, which is light emitted by a...
. In most cases, emitted light has a longer wavelength, and therefore lower energy, than the absorbed radiation. However, when the absorbed electromagnetic radiation is intense, it is possible for one electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
to absorb two photon
Photon
In physics, a photon is an elementary particle, the quantum of the electromagnetic interaction and the basic unit of light and all other forms of electromagnetic radiation. It is also the force carrier for the electromagnetic force...
s; this two-photon absorption
Two-photon absorption
Two-photon absorption is the simultaneous absorption of two photons of identical or different frequencies in order to excite a molecule from one state to a higher energy electronic state. The energy difference between the involved lower and upper states of the molecule is equal to the sum of the...
can lead to emission of radiation having a shorter wavelength than the absorbed radiation.
The most striking examples of fluorescence occur when the absorbed radiation is in the ultraviolet
Ultraviolet
Ultraviolet light is electromagnetic radiation with a wavelength shorter than that of visible light, but longer than X-rays, in the range 10 nm to 400 nm, and energies from 3 eV to 124 eV...
region of the spectrum
Spectrum
A spectrum is a condition that is not limited to a specific set of values but can vary infinitely within a continuum. The word saw its first scientific use within the field of optics to describe the rainbow of colors in visible light when separated using a prism; it has since been applied by...
, and thus invisible to the human eye, and the emitted light is in the visible region.
Fluorescence has many practical applications, including mineralogy
Mineralogy
Mineralogy is the study of chemistry, crystal structure, and physical properties of minerals. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization.-History:Early writing...
, gemology
Gemology
Gemology or gemmology is the science dealing with natural and artificial gems and gemstones. It is considered a geoscience and a branch of mineralogy...
, chemical sensors (fluorescence spectroscopy
Fluorescence spectroscopy
Fluorescence spectroscopy aka fluorometry or spectrofluorometry, is a type of electromagnetic spectroscopy which analyzes fluorescence from a sample. It involves using a beam of light, usually ultraviolet light, that excites the electrons in molecules of certain compounds and causes them to emit...
), fluorescent labelling
Fluorescent labelling
Fluorescent labelling is the process of covalently attaching a fluorophore to another molecule, such as a protein or nucleic acid. This is generally accomplished using a reactive derivative of the fluorophore that selectively binds to a functional group contained in the target molecule...
, dye
Dye
A dye is a colored substance that has an affinity to the substrate to which it is being applied. The dye is generally applied in an aqueous solution, and requires a mordant to improve the fastness of the dye on the fiber....
s, biological detectors, and, most commonly, fluorescent lamp
Fluorescent lamp
A fluorescent lamp or fluorescent tube is a gas-discharge lamp that uses electricity to excite mercury vapor. The excited mercury atoms produce short-wave ultraviolet light that then causes a phosphor to fluoresce, producing visible light. A fluorescent lamp converts electrical power into useful...
s.
History
An early observation of fluorescence was described by Nicolás MonardesNicolás Monardes
Nicolás Bautista Monardes was a Spanish physician and botanist.The genus Monarda was named for him.Monardes published several books of varying importance. In Diálogo llamado pharmacodilosis , he examines humanism and suggests studying several classical authors, principally Pedanius Dioscorides...
in 1565 in the infusion
Infusion
An infusion is the outcome of steeping plants with desired chemical compounds or flavors in water or oil.-History:The first recorded use of essential oils was in the 10th or 11th century by the Persian polymath Avicenna, possibly in The Canon of Medicine.-Preparation techniques:An infusion is very...
of a certain type of wood (Lignum nephriticum). The chemical compound responsible for this fluorescence is matlanine, which is the oxidation product of one of the flavonoids found in this wood. In 1819 Edward D. Clarke and in 1822 René Just Haüy
René Just Haüy
René Just Haüy – 3 June 1822 in Paris) was a French mineralogist, commonly styled the Abbé Haüy after he was made an honorary canon of Notre Dame. He is often referred to as the "Father of Modern Crystallography." -Biography:...
described fluorescence in florites, Sir David Brewster described the phenomenon for chlorophyll
Chlorophyll
Chlorophyll is a green pigment found in almost all plants, algae, and cyanobacteria. Its name is derived from the Greek words χλωρος, chloros and φύλλον, phyllon . Chlorophyll is an extremely important biomolecule, critical in photosynthesis, which allows plants to obtain energy from light...
in 1833 and Sir John Herschel did the same for quinine
Quinine
Quinine is a natural white crystalline alkaloid having antipyretic , antimalarial, analgesic , anti-inflammatory properties and a bitter taste. It is a stereoisomer of quinidine which, unlike quinine, is an anti-arrhythmic...
in 1845.
In his 1852 paper on the "Refrangibility" (wavelength
Wavelength
In physics, the wavelength of a sinusoidal wave is the spatial period of the wave—the distance over which the wave's shape repeats.It is usually determined by considering the distance between consecutive corresponding points of the same phase, such as crests, troughs, or zero crossings, and is a...
change) of light, George Gabriel Stokes
George Gabriel Stokes
Sir George Gabriel Stokes, 1st Baronet FRS , was an Irish mathematician and physicist, who at Cambridge made important contributions to fluid dynamics , optics, and mathematical physics...
, described a property of fluorspar and uranium glass
Uranium glass
Uranium glass is glass which has had uranium, usually in oxide diuranate form, added to a glass mix before melting. The proportion usually varies from trace levels to about 2% by weight uranium, although some 19th-century pieces were made with up to 25% uranium.Uranium glass was once made into...
, materials which he viewed as having the power to convert invisible ultra-violet radiation into radiation of longer wavelengths that are visible, he named the phenomenon fluorescence. He chose the name "to denote the general appearance of a solution of sulphate of quinine and similar media". The name was derived from the mineral fluorite
Fluorite
Fluorite is a halide mineral composed of calcium fluoride, CaF2. It is an isometric mineral with a cubic habit, though octahedral and more complex isometric forms are not uncommon...
(calcium difluoride), some examples of which contain traces of divalent europium
Europium
Europium is a chemical element with the symbol Eu and atomic number 63. It is named after the continent of Europe. It is a moderately hard silvery metal which readily oxidizes in air and water...
, which serves as the fluorescent activator to emit blue light. In a key experiment he used a prism to isolate ultraviolet radiation from sunlight and observed blue light emitted by a ethanol solution of quinine exposed by it.
Photochemistry
Fluorescence occurs when an orbital electronElectron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
of a molecule, atom or nanostructure
Nanostructure
A nanostructure is an object of intermediate size between molecular and microscopic structures.In describing nanostructures it is necessary to differentiate between the number of dimensions on the nanoscale. Nanotextured surfaces have one dimension on the nanoscale, i.e., only the thickness of the...
relaxes to its ground state
Ground state
The ground state of a quantum mechanical system is its lowest-energy state; the energy of the ground state is known as the zero-point energy of the system. An excited state is any state with energy greater than the ground state...
by emitting a photon
Photon
In physics, a photon is an elementary particle, the quantum of the electromagnetic interaction and the basic unit of light and all other forms of electromagnetic radiation. It is also the force carrier for the electromagnetic force...
of light after being excited
Excited state
Excitation is an elevation in energy level above an arbitrary baseline energy state. In physics there is a specific technical definition for energy level which is often associated with an atom being excited to an excited state....
to a higher quantum state by some type of energy:
Excitation:

Fluorescence (emission):

here
 is a generic term for photon energy with h = Planck's constant and
is a generic term for photon energy with h = Planck's constant and  = frequency
= frequencyFrequency
Frequency is the number of occurrences of a repeating event per unit time. It is also referred to as temporal frequency.The period is the duration of one cycle in a repeating event, so the period is the reciprocal of the frequency...
of light. (The specific frequencies of exciting and emitted light are dependent on the particular system.)
State S0 is called the ground state of the fluorophore
Fluorophore
A fluorophore, in analogy to a chromophore, is a component of a molecule which causes a molecule to be fluorescent. It is a functional group in a molecule which will absorb energy of a specific wavelength and re-emit energy at a different wavelength...
(fluorescent molecule) and S1 is its first (electronically) excited state.
A molecule in its excited state, S1, can relax by various competing pathways. It can undergo 'non-radiative relaxation' in which the excitation energy is dissipated as heat
Heat
In physics and thermodynamics, heat is energy transferred from one body, region, or thermodynamic system to another due to thermal contact or thermal radiation when the systems are at different temperatures. It is often described as one of the fundamental processes of energy transfer between...
(vibrations) to the solvent. Excited organic molecules can also relax via conversion to a triplet state
Triplet state
A spin triplet is a set of three quantum states of a system, each with total spin S = 1 . The system could consist of a single elementary massive spin 1 particle such as a W or Z boson, or be some multiparticle state with total spin angular momentum of one.In physics, spin is the angular momentum...
, which may subsequently relax via phosphorescence
Phosphorescence
Phosphorescence is a specific type of photoluminescence related to fluorescence. Unlike fluorescence, a phosphorescent material does not immediately re-emit the radiation it absorbs. The slower time scales of the re-emission are associated with "forbidden" energy state transitions in quantum...
or by a secondary non-radiative relaxation step.
Relaxation of an S1 state can also occur through interaction with a second molecule through fluorescence quenching
Quenching (fluorescence)
Quenching refers to any process which decreases the fluorescence intensity of a given substance. A variety of processes can result in quenching, such as excited state reactions, energy transfer, complex-formation and collisional quenching. As a consequence, quenching is often heavily dependent on...
. Molecular oxygen
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
(O2) is an extremely efficient quencher of fluorescence just because of its unusual triplet ground state.
Molecules that are excited through light absorption or via a different process (e.g. as the product of a reaction) can transfer energy to a second 'sensitized' molecule, which is converted to its excited state and can then fluoresce. This process is used in lightstick
Lightstick
A glow stick is a single-use translucent plastic tube containing isolated substances which when combined make light through a chemical reaction-induced chemiluminescence which does not require an electrical power source...
s to produce different colors.
Quantum yield
The fluorescence quantum yieldQuantum yield
The quantum yield of a radiation-induced process is the number of times that a defined event occurs per photon absorbed by the system. The "event" may represent a chemical reaction, for example the decomposition of a reactant molecule:...
gives the efficiency of the fluorescence process. It is defined as the ratio of the number of photons emitted to the number of photons absorbed.
The maximum fluorescence quantum yield is 1.0 (100%); every photon
Photon
In physics, a photon is an elementary particle, the quantum of the electromagnetic interaction and the basic unit of light and all other forms of electromagnetic radiation. It is also the force carrier for the electromagnetic force...
absorbed results in a photon emitted. Compounds with quantum yields of 0.10 are still considered quite fluorescent. Another way to define the quantum yield of fluorescence, is by the rate of excited state decay:
where
 is the rate of spontaneous emission
is the rate of spontaneous emissionSpontaneous emission
Spontaneous emission is the process by which a light source such as an atom, molecule, nanocrystal or nucleus in an excited state undergoes a transition to a state with a lower energy, e.g., the ground state and emits a photon...
of radiation and

is the sum of all rates of excited state decay. Other rates of excited state decay are caused by mechanisms other than photon emission and are, therefore, often called "non-radiative rates", which can include:
dynamic collisional quenching, near-field dipole-dipole interaction (or resonance energy transfer), internal conversion, and intersystem crossing
Intersystem crossing
Intersystem crossing is a radiationless process involving a transition between two electronic states with different spin multiplicity.-Singlet and triplet states:...
. Thus, if the rate of any pathway changes, both the excited state lifetime and the fluorescence quantum yield will be affected.
Fluorescence quantum yields are measured by comparison to a standard. The quinine
Quinine
Quinine is a natural white crystalline alkaloid having antipyretic , antimalarial, analgesic , anti-inflammatory properties and a bitter taste. It is a stereoisomer of quinidine which, unlike quinine, is an anti-arrhythmic...
salt quinine sulfate in a sulfuric acid
Sulfuric acid
Sulfuric acid is a strong mineral acid with the molecular formula . Its historical name is oil of vitriol. Pure sulfuric acid is a highly corrosive, colorless, viscous liquid. The salts of sulfuric acid are called sulfates...
solution is a common fluorescence standard.
Lifetime
The fluorescence lifetime refers to the average time the molecule stays in its excited state before emitting a photon. Fluorescence typically follows first-order kinetics:
where
 is the concentration of excited state molecules at time
is the concentration of excited state molecules at time  ,
,  is the initial concentration and
is the initial concentration and 
Gamma
Gamma is the third letter of the Greek alphabet. In the system of Greek numerals it has a value of 3. It was derived from the Phoenician letter Gimel . Letters that arose from Gamma include the Roman C and G and the Cyrillic letters Ge Г and Ghe Ґ.-Greek:In Ancient Greek, gamma represented a...
is the decay rate or the inverse of the fluorescence lifetime. This is an instance of exponential decay. Various radiative and non-radiative processes can de-populate the excited state. In such case the total decay rate is the sum over all rates:

where
 is the total decay rate,
is the total decay rate,  the radiative decay rate and
the radiative decay rate and  the non-radiative decay rate. It is similar to a first-order chemical reaction in which the first-order rate constant is the sum of all of the rates (a parallel kinetic model). If the rate of spontaneous emission, or any of the other rates are fast, the lifetime is short. For commonly used fluorescent compounds, typical excited state decay times for photon emissions with energies from the UV
the non-radiative decay rate. It is similar to a first-order chemical reaction in which the first-order rate constant is the sum of all of the rates (a parallel kinetic model). If the rate of spontaneous emission, or any of the other rates are fast, the lifetime is short. For commonly used fluorescent compounds, typical excited state decay times for photon emissions with energies from the UVUltraviolet
Ultraviolet light is electromagnetic radiation with a wavelength shorter than that of visible light, but longer than X-rays, in the range 10 nm to 400 nm, and energies from 3 eV to 124 eV...
to near infrared are within the range of 0.5 to 20 nanoseconds. The fluorescence lifetime is an important parameter for practical applications of fluorescence such as fluorescence resonance energy transfer
Fluorescence resonance energy transfer
Förster resonance energy transfer , also known as fluorescence resonance energy transfer, resonance energy transfer or electronic energy transfer , is a mechanism describing energy transfer between two chromophores.A donor chromophore, initially in its electronic excited state, may transfer energy...
.
Rules
There are several rules
Rule of thumb
A rule of thumb is a principle with broad application that is not intended to be strictly accurate or reliable for every situation. It is an easily learned and easily applied procedure for approximately calculating or recalling some value, or for making some determination...
that deal with fluorescence. The Kasha–Vavilov rule
Kasha's rule
Kasha's rule is a principle in the photochemistry of electronically excited molecules. The rule states that photon emission occurs in appreciable yield only from the lowest excited state of a given multiplicity...
dictates that the quantum yield of luminescence is independent of the wavelength of exciting radiation. This is not always true and is violated severely in many simple molecules. A somewhat more reliable statement, although still with exceptions, would be that the fluorescence spectrum shows very little dependence on the wavelength of exciting radiation.
The Jablonski diagram
Jablonski diagram
A Jablonski diagram, named after the Polish physicist Aleksander Jabłoński, is a diagram that illustrates the electronic states of a molecule and the transitions between them. The states are arranged vertically by energy and grouped horizontally by spin multiplicity. Nonradiative transitions are...
describes most of the relaxation mechanisms for excited state molecules.
Applications
There are many natural and synthetic compounds that exhibit fluorescence, and they have a number of applications. Some deep-sea animals, such as the greeneyeGreeneye
Greeneyes are deep-sea aulopiform marine fishes in the small family Chlorophthalmidae. Thought to have a circumglobal distribution in tropical and temperate waters, the family contains just 18 species in two genera...
, use fluorescence.
Lighting

Fluorescent lamp
A fluorescent lamp or fluorescent tube is a gas-discharge lamp that uses electricity to excite mercury vapor. The excited mercury atoms produce short-wave ultraviolet light that then causes a phosphor to fluoresce, producing visible light. A fluorescent lamp converts electrical power into useful...
relies on fluorescence. Inside the glass
Glass
Glass is an amorphous solid material. Glasses are typically brittle and optically transparent.The most familiar type of glass, used for centuries in windows and drinking vessels, is soda-lime glass, composed of about 75% silica plus Na2O, CaO, and several minor additives...
tube is a partial vacuum and a small amount of mercury
Mercury (element)
Mercury is a chemical element with the symbol Hg and atomic number 80. It is also known as quicksilver or hydrargyrum...
. An electric discharge in the tube causes the mercury atoms to emit ultraviolet light. The tube is lined with a coating of a fluorescent material, called the phosphor
Phosphor
A phosphor, most generally, is a substance that exhibits the phenomenon of luminescence. Somewhat confusingly, this includes both phosphorescent materials, which show a slow decay in brightness , and fluorescent materials, where the emission decay takes place over tens of nanoseconds...
, which absorbs the ultraviolet and re-emits visible light. Fluorescent lighting
Lighting
Lighting or illumination is the deliberate application of light to achieve some practical or aesthetic effect. Lighting includes the use of both artificial light sources such as lamps and light fixtures, as well as natural illumination by capturing daylight...
is more energy-efficient than incandescent lighting elements. However, the uneven spectrum
Spectrum
A spectrum is a condition that is not limited to a specific set of values but can vary infinitely within a continuum. The word saw its first scientific use within the field of optics to describe the rainbow of colors in visible light when separated using a prism; it has since been applied by...
of traditional fluorescent lamps may cause certain colors to appear different than when illuminated by incandescent light or daylight
Daylight
Daylight or the light of day is the combination of all direct and indirect sunlight outdoors during the daytime. This includes direct sunlight, diffuse sky radiation, and both of these reflected from the Earth and terrestrial objects. Sunlight scattered or reflected from objects in outer space is...
. The mercury vapor emission spectrum is dominated by a short-wave UV line at 254 nm (which provides most of the energy to the phosphors), accompanied by visible light emission at 436 nm (blue), 546 nm (green) and 579 nm (yellow-orange). These three lines can be observed superimposed on the white continuum using a hand spectroscope, for light emitted by the usual white fluorescent tubes. These same visible lines, accompanied by the emission lines of trivalent europium and trivalent terbium, and further accompanied by the emission continuum of divalent europium in the blue region, comprise the more discontinuous light emission of the modern trichromatic phosphor systems used in many compact fluorescent lamp
Compact fluorescent lamp
A compact fluorescent lamp , also called compact fluorescent light, energy-saving light, and compact fluorescent tube, is a fluorescent lamp designed to replace an incandescent lamp; some types fit into light fixtures formerly used for incandescent lamps...
and traditional lamps where better color rendition is a goal.
Fluorescent lights were first available to the public at the 1939 New York World's Fair
1939 New York World's Fair
The 1939–40 New York World's Fair, which covered the of Flushing Meadows-Corona Park , was the second largest American world's fair of all time, exceeded only by St. Louis's Louisiana Purchase Exposition of 1904. Many countries around the world participated in it, and over 44 million people...
. Improvements since then have largely been better phosphors, longer life, and more consistent internal discharge, and easier-to-use shapes (such as compact fluorescent lamps). Some high-intensity discharge (HID) lamps couple their even-greater electrical efficiency with phosphor enhancement for better color rendition.
White light-emitting diode
Light-emitting diode
A light-emitting diode is a semiconductor light source. LEDs are used as indicator lamps in many devices and are increasingly used for other lighting...
s (LEDs) became available in the mid-1990s as LED lamp
LED lamp
An LED lamp is a solid-state lamp that uses light-emitting diodes as the source of light. The LEDs involved may be conventional semiconductor light-emitting diodes, organic LEDs , or polymer light-emitting diodes devices, although PLED technologies are not currently commercially available.Since...
s, in which blue light emitted from the semiconductor
Semiconductor
A semiconductor is a material with electrical conductivity due to electron flow intermediate in magnitude between that of a conductor and an insulator. This means a conductivity roughly in the range of 103 to 10−8 siemens per centimeter...
strikes phosphors deposited on the tiny chip. The combination of the blue light that continues through the phosphor and the green to red fluorescence from the phosphors produces a net emission of white light.
Glow sticks sometimes utilize fluorescent materials to absorb light from the chemiluminescent reaction and emit light of a different color.
Analytical chemistry
Many analytical procedures involve the use of a fluorometerFluorometer
A fluorometer or fluorimeter is a device used to measure parameters of fluorescence: its intensity and wavelength distribution of emission spectrum after excitation by a certain spectrum of light. These parameters are used to identify the presence and the amount of specific molecules in a medium...
, usually with a single exciting wavelength and single detection wavelength. Because of the sensitivity that the method affords, fluorescent molecule concentrations as low as 1 part per trillion can be measured.
Fluorescence in several wavelengths can be detected by an array detector
Chromatography detector
A chromatography detector is a device used in gas chromatography or liquid chromatography to visualize components of the mixture being eluted off the chromatography column. There are two general types of detectors: destructive and non-destructive...
, to detect compounds from HPLC
High-performance liquid chromatography
High-performance liquid chromatography , HPLC, is a chromatographic technique that can separate a mixture of compounds and is used in biochemistry and analytical chemistry to identify, quantify and purify the individual components of the mixture.HPLC typically utilizes different types of stationary...
flow. Also, TLC
Thin layer chromatography
Thin layer chromatography is a chromatography technique used to separate mixtures. Thin layer chromatography is performed on a sheet of glass, plastic, or aluminum foil, which is coated with a thin layer of adsorbent material, usually silica gel, aluminium oxide, or cellulose...
plates can be visualized if the compounds or a coloring reagent is fluorescent. Fluorescence is most effective when there is a larger ratio of atoms at lower energy levels in a Boltzmann distribution
Boltzmann distribution
In chemistry, physics, and mathematics, the Boltzmann distribution is a certain distribution function or probability measure for the distribution of the states of a system. It underpins the concept of the canonical ensemble, providing its underlying distribution...
. There is, then, a higher probability of excitement and release of photons by lower-energy atoms, making analysis more efficient.
Equipment
Usually the setup of a Fluorescence assay involves a Light source, which may emit an array different wavelengths of light. In general, a single wavelength is required for proper analysis, so, in order to selectively filter the light, it is passed through an excitation monochromator, and then that chosen wavelength is passed through the sample cell. After absorption and re-emission of the energy, many wavelengths may emerge due to Stokes shiftStokes shift
Stokes shift is the difference between positions of the band maxima of the absorption and emission spectra of the same electronic transition. It is named after Irish physicist George G. Stokes. When a system absorbs a photon, it gains energy and enters an excited state...
and various electron transitions. To separate and analyze them, the fluorescent radiation is passed through an Emission monochromator
Monochromator
A monochromator is an optical device that transmits a mechanically selectable narrow band of wavelengths of light or other radiation chosen from a wider range of wavelengths available at the input...
, and observed selectively by a detector.
Biochemistry and medicine
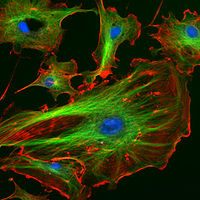
Autofluorescence
Autofluorescence is the natural emission of light by biological entities such as mitochondria and lysosomes, and is used to distinguish the light originating from artificially added fluorescent markers...
). In fact, a protein
Protein
Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. A polypeptide is a single linear polymer chain of amino acids bonded together by peptide bonds between the carboxyl and amino groups of...
or other component can be "labelled" with a extrinsic fluorophore
Fluorophore
A fluorophore, in analogy to a chromophore, is a component of a molecule which causes a molecule to be fluorescent. It is a functional group in a molecule which will absorb energy of a specific wavelength and re-emit energy at a different wavelength...
, a fluorescent dye
Dye
A dye is a colored substance that has an affinity to the substrate to which it is being applied. The dye is generally applied in an aqueous solution, and requires a mordant to improve the fastness of the dye on the fiber....
that can be a small molecule, protein, or quantum dot, finding a large use in many biological applications.
The quantification of a dye is done with a spectrofluorometer
Spectrofluorometer
The spectrofluorometer is an instrument which takes advantage of fluorescent properties of some compounds in order to provide information regarding their concentration and chemical environment in a sample...
and finds additional applications in:
- When scanning the fluorescent intensity across a plane one has fluorescence microscopyFluorescence microscopeA fluorescence microscope is an optical microscope used to study properties of organic or inorganic substances using the phenomena of fluorescence and phosphorescence instead of, or in addition to, reflection and absorption...
of tissues, cells, or subcellular structures, which is accomplished by labelling an antibody with a fluorophore and allowing the antibody to find its target antigen within the sample. Labelling multiple antibodies with different fluorophores allows visualization of multiple targets within a single image (multiple channels). DNA microarrays are a variant of this. - Automated sequencing of DNADNADeoxyribonucleic acid is a nucleic acid that contains the genetic instructions used in the development and functioning of all known living organisms . The DNA segments that carry this genetic information are called genes, but other DNA sequences have structural purposes, or are involved in...
by the chain termination method; each of four different chain terminating bases has its own specific fluorescent tag. As the labelled DNA molecules are separated, the fluorescent label is excited by a UV source, and the identity of the base terminating the molecule is identified by the wavelength of the emitted light. - Ethidium bromide fluoresces orange when intercalatingIntercalation (chemistry)In chemistry, intercalation is the reversible inclusion of a molecule between two other molecules . Examples include DNA intercalation and graphite intercalation compounds.- DNA intercalation :...
DNADNADeoxyribonucleic acid is a nucleic acid that contains the genetic instructions used in the development and functioning of all known living organisms . The DNA segments that carry this genetic information are called genes, but other DNA sequences have structural purposes, or are involved in...
and when exposed to UV light. - FACS (fluorescence-activated cell sorting). One of several important cell sorting techniques used in the separation of different cell lines (especially those isolated from animal tissues).
- DNA detection: the compound ethidium bromideEthidium bromideEthidium bromide is an intercalating agent commonly used as a fluorescent tag in molecular biology laboratories for techniques such as agarose gel electrophoresis. It is commonly abbreviated as "EtBr", which is also an abbreviation for bromoethane...
, when free to change its conformation in solution, has very little fluorescence. Ethidium bromide's fluorescence is greatly enhanced after it binds to DNA, so this compound is very useful in visualising the location of DNA fragments in agarose gel electrophoresis. Ethidium bromide may be carcinogenic – an arguably safer alternative is the dye SYBR GreenSYBR GreenSYBR Green I is an asymmetrical cyanine dye used as a nucleic acid stain in molecular biology. SYBR Green I binds to DNA. The resulting DNA-dye-complex absorbs blue light and emits green light . The stain preferentially binds to double-stranded DNA, but will stain single-stranded DNA with lower...
. - Immunology: An antibody is first prepared by having a fluorescent chemical group attached, and the sites (e.g., on a microscopic specimen) where the antibody has bound can be seen, and even quantified, by the fluorescence.
- FLIM (Fluorescence Lifetime Imaging Microscopy) can be used to detect certain bio-molecular interactions that manifest themselves by influencing fluorescence lifetimes.
- FRET Fluorescence resonance energy transferFluorescence resonance energy transferFörster resonance energy transfer , also known as fluorescence resonance energy transfer, resonance energy transfer or electronic energy transfer , is a mechanism describing energy transfer between two chromophores.A donor chromophore, initially in its electronic excited state, may transfer energy...
is used to study protein interactions, detect specific nucleic acid sequences and used as biosensors, while fluorescence lifetime (FLIM) can give an additional layer of information. - Biotechnology: biosensors using fluorescence are being studied as possible Fluorescent glucose biosensors.
- Cell and molecular biology: detection of colocalizationColocalizationIn fluorescence microscopy, colocalization refers to observation of the spatial overlap between two different fluorescent labels, each having a separate emission wavelength, to see if the different "targets" are located in the same area of the cell or very near to one another...
using fluorescence-labelled antibodies for selective detection of the antigens of interest using specialized software, such as CoLocalizer ProCoLocalizer ProCoLocalizer Pro is a scientific software application, developed by , that allows researchers to analyze colocalization in the images obtained using fluorescence microscopy. Due to high popularity of Macintosh computers in medicine and biology, the software is designed specifically for Mac OS X...
.
Gemology, mineralogy, geology and forensics
FingerprintFingerprint
A fingerprint in its narrow sense is an impression left by the friction ridges of a human finger. In a wider use of the term, fingerprints are the traces of an impression from the friction ridges of any part of a human hand. A print from the foot can also leave an impression of friction ridges...
s can be visualized with fluorescent compounds such as ninhydrin
Ninhydrin
Ninhydrin is a chemical used to detect ammonia or primary and secondary amines. When reacting with these free amines, a deep blue or purple color known as Ruhemann's purple is produced...
. Blood and other substances are sometimes detected by fluorescent reagents, in particular where their location was not previously known.
Gemstone
Gemstone
A gemstone or gem is a piece of mineral, which, in cut and polished form, is used to make jewelry or other adornments...
s, mineral
Mineral
A mineral is a naturally occurring solid chemical substance formed through biogeochemical processes, having characteristic chemical composition, highly ordered atomic structure, and specific physical properties. By comparison, a rock is an aggregate of minerals and/or mineraloids and does not...
s, fiber
Fiber
Fiber is a class of materials that are continuous filaments or are in discrete elongated pieces, similar to lengths of thread.They are very important in the biology of both plants and animals, for holding tissues together....
s, and many other materials that may be encountered in forensics or with a relationship to various collectible
Collectible
A collectable or collectible is any object regarded as being of value or interest to a collector . There are numerous types of collectables and terms to denote those types. An antique is a collectable that is old...
s may have a distinctive fluorescence or may fluoresce differently under short-wave ultraviolet, long-wave ultraviolet, or X-ray
X-ray
X-radiation is a form of electromagnetic radiation. X-rays have a wavelength in the range of 0.01 to 10 nanometers, corresponding to frequencies in the range 30 petahertz to 30 exahertz and energies in the range 120 eV to 120 keV. They are shorter in wavelength than UV rays and longer than gamma...
s.
Many types of calcite
Calcite
Calcite is a carbonate mineral and the most stable polymorph of calcium carbonate . The other polymorphs are the minerals aragonite and vaterite. Aragonite will change to calcite at 380-470°C, and vaterite is even less stable.-Properties:...
and amber will fluoresce under shortwave UV. Rubies
Ruby
A ruby is a pink to blood-red colored gemstone, a variety of the mineral corundum . The red color is caused mainly by the presence of the element chromium. Its name comes from ruber, Latin for red. Other varieties of gem-quality corundum are called sapphires...
, emerald
Emerald
Emerald is a variety of the mineral beryl colored green by trace amounts of chromium and sometimes vanadium. Beryl has a hardness of 7.5–8 on the 10 point Mohs scale of mineral hardness...
s, and the Hope Diamond
Hope Diamond
The Hope Diamond, also known as "Le bleu de France" or "Le Bijou du Roi", is a large, , deep-blue diamond, now housed in the Smithsonian Natural History Museum in Washington, D.C. It is blue to the naked eye because of trace amounts of boron within its crystal structure, but exhibits red...
exhibit red fluorescence under short-wave UV light; diamonds also emit light under X ray radiation.
Fluorescence in minerals is caused by a wide range of activators. In some cases, the concentration of the activator must be restricted to below a certain level, to prevent quenching of the fluorescent emission. Furthermore, certain impurities such as iron or copper need to be absent, to prevent quenching of possible fluorescence. Divalent manganese
Manganese
Manganese is a chemical element, designated by the symbol Mn. It has the atomic number 25. It is found as a free element in nature , and in many minerals...
, in concentrations of up to several percent, is responsible for the red or orange fluorescence of calcite
Calcite
Calcite is a carbonate mineral and the most stable polymorph of calcium carbonate . The other polymorphs are the minerals aragonite and vaterite. Aragonite will change to calcite at 380-470°C, and vaterite is even less stable.-Properties:...
, the green fluorescence of willemite
Willemite
Willemite is a zinc silicate mineral and a minor ore of zinc. It is highly fluorescent under shortwave ultraviolet light.It occurs in all different colors in daylight, in fibrous masses, solid brown masses , and apple green gemmy masses.It was discovered in 1830 and named after William I of the...
, the yellow fluorescence of esperite
Esperite
Esperite is a rare complex calcium lead zinc silicate related to beryllonite and trimerite that used to be called calcium larsenite...
, and the orange fluorescence of wollastonite
Wollastonite
Wollastonite is a calcium inosilicate mineral that may contain small amounts of iron, magnesium, and manganese substituting for calcium. It is usually white. It forms when impure limestone or dolostone is subjected to high temperature and pressure sometimes in the presence of silica-bearing fluids...
and clinohedrite
Clinohedrite
Clinohedrite is a rare silicate mineral. Its chemical composition is a hydrous calcium-zinc silicate; CaZn4·H2O. It crystallizes in the monoclinic system and typically occurs as veinlets and fracture coatings. It is commonly colorless, white to pale amethyst in color. It has perfect cleavage and...
. Hexavalent uranium
Uranium
Uranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons...
, in the form of the uranyl cation, fluoresces at all concentrations in a yellow green, and is the cause of fluorescence of minerals such as autunite
Autunite
Autunite with formula: Ca22·10-12H2O is a yellow - greenish fluorescent mineral with a hardness of 2 - 2½. Autunite crystallizes in the tetragonal system and often occurs as tabular square crystals. Due to the moderate uranium content of 48.27% it is radioactive and also used as uranium ore...
or andersonite
Andersonite
Andersonite, Andersonit or Andersonita , or hydrated sodium calcium uranyl carbonate is a rare uranium carbonate mineral that was only described in the last half century. Named for Charles Alfred Anderson of United States Geological Survey, who first described the mineral species, it is found in...
, and, at low concentration, is the cause of the fluorescence of such materials as some samples of hyalite opal. Trivalent chromium
Chromium
Chromium is a chemical element which has the symbol Cr and atomic number 24. It is the first element in Group 6. It is a steely-gray, lustrous, hard metal that takes a high polish and has a high melting point. It is also odorless, tasteless, and malleable...
at low concentration is the source of the red fluorescence of ruby
Ruby
A ruby is a pink to blood-red colored gemstone, a variety of the mineral corundum . The red color is caused mainly by the presence of the element chromium. Its name comes from ruber, Latin for red. Other varieties of gem-quality corundum are called sapphires...
. Divalent europium
Europium
Europium is a chemical element with the symbol Eu and atomic number 63. It is named after the continent of Europe. It is a moderately hard silvery metal which readily oxidizes in air and water...
is the source of the blue fluorescence, when seen in the mineral fluorite
Fluorite
Fluorite is a halide mineral composed of calcium fluoride, CaF2. It is an isometric mineral with a cubic habit, though octahedral and more complex isometric forms are not uncommon...
. Trivalent lanthanide
Lanthanide
The lanthanide or lanthanoid series comprises the fifteen metallic chemical elements with atomic numbers 57 through 71, from lanthanum through lutetium...
s such as terbium
Terbium
Terbium is a chemical element with the symbol Tb and atomic number 65. It is a silvery-white rare earth metal that is malleable, ductile and soft enough to be cut with a knife...
and dysprosium
Dysprosium
Dysprosium is a chemical element with the symbol Dy and atomic number 66. It is a rare earth element with a metallic silver luster. Dysprosium is never found in nature as a free element, though it is found in various minerals, such as xenotime...
are the principal activators of the creamy yellow fluorescence exhibited by the yttrofluorite variety of the mineral fluorite, and contribute to the orange fluorescence of zircon
Zircon
Zircon is a mineral belonging to the group of nesosilicates. Its chemical name is zirconium silicate and its corresponding chemical formula is ZrSiO4. A common empirical formula showing some of the range of substitution in zircon is 1–x4x–y...
. Powellite
Powellite
Powellite is a calcium molybdate mineral with formula CaMoO4. Powellite crystallizes with tetragonal - dipyramidal crystal structure as transparent adamantine blue, greenish brown, yellow to grey typically anhedral forms. It exhibits distinct cleavage and has a brittle to conchoidal fracture. It...
(calcium molybdate) and scheelite
Scheelite
Scheelite is a calcium tungstate mineral with the chemical formula CaWO4. It is an important ore of tungsten. Well-formed crystals are sought by collectors and are occasionally fashioned into gemstones when suitably free of flaws...
(calcium tungstate) fluoresce intrinsically in yellow and blue, respectively. When present together in solid solution, energy is transferred from the higher-energy tungsten
Tungsten
Tungsten , also known as wolfram , is a chemical element with the chemical symbol W and atomic number 74.A hard, rare metal under standard conditions when uncombined, tungsten is found naturally on Earth only in chemical compounds. It was identified as a new element in 1781, and first isolated as...
to the lower-energy molybdenum
Molybdenum
Molybdenum , is a Group 6 chemical element with the symbol Mo and atomic number 42. The name is from Neo-Latin Molybdaenum, from Ancient Greek , meaning lead, itself proposed as a loanword from Anatolian Luvian and Lydian languages, since its ores were confused with lead ores...
, such that fairly low levels of molybdenum
Molybdenum
Molybdenum , is a Group 6 chemical element with the symbol Mo and atomic number 42. The name is from Neo-Latin Molybdaenum, from Ancient Greek , meaning lead, itself proposed as a loanword from Anatolian Luvian and Lydian languages, since its ores were confused with lead ores...
are sufficient to cause a yellow emission for scheelite
Scheelite
Scheelite is a calcium tungstate mineral with the chemical formula CaWO4. It is an important ore of tungsten. Well-formed crystals are sought by collectors and are occasionally fashioned into gemstones when suitably free of flaws...
, instead of blue. Low-iron sphalerite
Sphalerite
Sphalerite is a mineral that is the chief ore of zinc. It consists largely of zinc sulfide in crystalline form but almost always contains variable iron. When iron content is high it is an opaque black variety, marmatite. It is usually found in association with galena, pyrite, and other sulfides...
(zinc sulfide), fluoresces and phosphoresces in a range of colors, influenced by the presence of various trace impurities.
Crude oil (petroleum
Petroleum
Petroleum or crude oil is a naturally occurring, flammable liquid consisting of a complex mixture of hydrocarbons of various molecular weights and other liquid organic compounds, that are found in geologic formations beneath the Earth's surface. Petroleum is recovered mostly through oil drilling...
) fluoresces in a range of colors, from dull-brown for heavy oils and tars through to bright-yellowish and bluish-white for very light oils and condensates. This phenomenon is used in oil exploration
Oil exploration
Hydrocarbon exploration is the search by petroleum geologists and geophysicists for hydrocarbon deposits beneath the Earth's surface, such as oil and natural gas...
drilling to identify very small amounts of oil in drill cuttings and core samples.
Organic liquids
Organic liquids such anthraceneAnthracene
Anthracene is a solid polycyclic aromatic hydrocarbon consisting of three fused benzene rings. It is a component of coal-tar. Anthracene is used in the production of the red dye alizarin and other dyes...
in benzene
Benzene
Benzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6....
, toluene
Toluene
Toluene, formerly known as toluol, is a clear, water-insoluble liquid with the typical smell of paint thinners. It is a mono-substituted benzene derivative, i.e., one in which a single hydrogen atom from the benzene molecule has been replaced by a univalent group, in this case CH3.It is an aromatic...
, or stilbene
Stilbene
-Stilbene, is a diarylethene, i.e., a hydrocarbon consisting of a trans ethene double bond substituted with a phenyl group on both carbon atoms of the double bond. The name stilbene is derived from the Greek word stilbos, which means shining....
in the same solvent
Solvent
A solvent is a liquid, solid, or gas that dissolves another solid, liquid, or gaseous solute, resulting in a solution that is soluble in a certain volume of solvent at a specified temperature...
s fluoresce with ultraviolet
Ultraviolet
Ultraviolet light is electromagnetic radiation with a wavelength shorter than that of visible light, but longer than X-rays, in the range 10 nm to 400 nm, and energies from 3 eV to 124 eV...
or gamma ray
Gamma ray
Gamma radiation, also known as gamma rays or hyphenated as gamma-rays and denoted as γ, is electromagnetic radiation of high frequency . Gamma rays are usually naturally produced on Earth by decay of high energy states in atomic nuclei...
irradiation
Irradiation
Irradiation is the process by which an object is exposed to radiation. The exposure can originate from various sources, including natural sources. Most frequently the term refers to ionizing radiation, and to a level of radiation that will serve a specific purpose, rather than radiation exposure to...
. The decay times of this fluorescence are of the order of nanoseconds, since the duration of the light depends on the lifetime of the excited states of the fluorescent material, in this case anthracene or stilbene.
See also
- Absorption-re-emission atomic line filterAtomic line filterAn atomic line filter is an advanced optical band-pass filter used in the physical sciences for filtering electromagnetic radiation with precision, accuracy, and minimal signal strength loss...
s use the phenomenon of fluorescence to filter light extremely effectively. - Black lightBlack lightA black light, also referred to as a UV light, ultraviolet light, or Wood's lamp, is a lamp that emits ultraviolet radiation in the long-wave range, and little visible light...
- Blacklight paintBlacklight paintBlacklight ink or blacklight-reactive Ink is ink that glows under a black light, a source of light whose wavelengths are primarily in the ultraviolet. The paint may or may not be colorful under ordinary light...
- Evos microscope
- Fluorescence correlation spectroscopyFluorescence correlation spectroscopyFluorescence correlation spectroscopy is a correlation analysis of fluctuation of the fluorescence intensity. The analysis provides parameters of the physics under the fluctuations. One of the interesting applications of this is an analysis of the concentration fluctuations of fluorescent...
- Fluorescence in plants
- Fluorescence spectroscopyFluorescence spectroscopyFluorescence spectroscopy aka fluorometry or spectrofluorometry, is a type of electromagnetic spectroscopy which analyzes fluorescence from a sample. It involves using a beam of light, usually ultraviolet light, that excites the electrons in molecules of certain compounds and causes them to emit...
- Fluorescent lampFluorescent lampA fluorescent lamp or fluorescent tube is a gas-discharge lamp that uses electricity to excite mercury vapor. The excited mercury atoms produce short-wave ultraviolet light that then causes a phosphor to fluoresce, producing visible light. A fluorescent lamp converts electrical power into useful...
- Fluorescent multilayer cardFluorescent Multilayer CardUsing the same 3D optical data storage technology as the fluorescent multilayer disc, the fluorescent multilayer card was a memory device proposed by Consallation3D. It consisted of a card containing a square shaped clear part with multiple layers each containing a different fluorescent material...
- Fluorescent Multilayer DiscFluorescent Multilayer DiscFluorescent Multilayer Disc was an optical disc format developed by Constellation 3D that uses fluorescent, rather than reflective materials to store data. Reflective disc formats have a practical limitation of about two layers, primarily due to interference, scatter, and inter-layer cross talk...
- FluorometerFluorometerA fluorometer or fluorimeter is a device used to measure parameters of fluorescence: its intensity and wavelength distribution of emission spectrum after excitation by a certain spectrum of light. These parameters are used to identify the presence and the amount of specific molecules in a medium...
- High-visibility clothingHigh-visibility clothingHigh-visibility clothing, a type of personal protective equipment , is any clothing worn that has highly reflective properties or a colour that is easily discernible from any background. Yellow waistcoats worn by emergency services are a common example....
- Laser-induced fluorescenceLaser-induced fluorescenceLaser-induced fluorescence is a spectroscopic method used for studying structure of molecules, detection of selective species and flow visualization and measurements....
- List of light sources
- Mössbauer effectMössbauer effectThe Mössbauer effect, or recoilless nuclear resonance fluorescence, is a physical phenomenon discovered by Rudolf Mössbauer in 1958. It involves the resonant and recoil-free emission and absorption of γ radiation by atomic nuclei bound in a solid...
, resonant fluorescence of gamma rays - Organic light-emitting diodeOrganic light-emitting diodeAn OLED is a light-emitting diode in which the emissive electroluminescent layer is a film of organic compounds which emit light in response to an electric current. This layer of organic semiconductor material is situated between two electrodes...
s can be fluorescent - PhosphorescencePhosphorescencePhosphorescence is a specific type of photoluminescence related to fluorescence. Unlike fluorescence, a phosphorescent material does not immediately re-emit the radiation it absorbs. The slower time scales of the re-emission are associated with "forbidden" energy state transitions in quantum...
- Phosphor thermometryPhosphor thermometryPhosphor thermometry is an optical method for surface temperature measurement. The method exploits luminescence emitted by phosphor material. Phosphors are fine white or pastel-colored inorganic powders which may be stimulated by any of a variety of means to luminesce, i.e. emit light...
, the use of phosphorescence to measure temperature. - SpectroscopySpectroscopySpectroscopy is the study of the interaction between matter and radiated energy. Historically, spectroscopy originated through the study of visible light dispersed according to its wavelength, e.g., by a prism. Later the concept was expanded greatly to comprise any interaction with radiative...
- Two-photon absorptionTwo-photon absorptionTwo-photon absorption is the simultaneous absorption of two photons of identical or different frequencies in order to excite a molecule from one state to a higher energy electronic state. The energy difference between the involved lower and upper states of the molecule is equal to the sum of the...
- X-ray fluorescenceX-ray fluorescenceX-ray fluorescence is the emission of characteristic "secondary" X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays...
External links
- Fluorophores.org, the database of fluorescent dyes
- Zeiss.com, Interactive Fluorescence Dye and Filter Database Carl ZeissCarl ZeissCarl Zeiss was a German maker of optical instruments commonly known for the company he founded, Carl Zeiss Jena . Zeiss made contributions to lens manufacturing that have aided the modern production of lenses...
Interactive Fluorescence Dye and Filter Database. - ISS.com, Fluorescence Lifetime Standards Tables
- ISS.com, Fluorescence Probes Data Tables
- Fluorescence-Foundation.org
- SHSU.edu, Jablonski diagram
- Wolfram.com, Fluorescence on Scienceworld
- FSU.edu, Basic Concepts in Fluorescence
- Confocal-microscopy.org, Immunofluorescence Protocol
- BBham.ac.uk, An example of use of fluorescence in generating cellular images
- More examples how the fluorescence can be used
- Fluorescence control by Photonic Crystals – ICMM
- The Fluorescent Mineral Society
- Fluorescence in Practice
- "A nano-history of fluorescence" lecture by David Jameson
- Excitation and emission spectra of various fluorescent dyes
- Manawatu Microscopy – first known collaboration environment for Microscopy and Image Analysis featuring Open DataBase of Fluorescent Dyes and Stains and their applications.
- Fluorescence Tutorials
- The Molecular Probes Handbook – a comprehensive resource for fluorescence technology and its applications.
- Scientific research in and fun-diving for bio-fluorescence of marine organisms



