
Nuclear binding energy
Encyclopedia
Nuclear binding energy is the energy required to split a nucleus of an atom
into its component parts. The component parts are neutrons and protons, which are collectively called nucleon
s. If the binding energy
for the products is higher when light nuclei fuse, or when heavy nuclei split, either of these processes will result in a release of the extra binding energy, and this energy is referred to as nuclear energy or, more loosely, nuclear power
.
The mass
of an atom's nucleus is always less than the sum of the individual masses of the constituent protons and neutrons. This notable difference is a measure of the nuclear binding energy, which is a result of forces that hold the nucleus together. Because these forces result in the removal of energy when the nucleus is formed, and this energy has mass, mass is removed from the total mass of the original particles, and the mass is missing in the resulting nucleus. This missing mass is known as the mass defect, and represents the energy released when the nucleus is formed. When the nucleus splits
into pieces, this energy may be emitted as photons (gamma rays) or as the mass or kinetic energy of a number of different ejected particles. Total mass is conserved throughout the process, and during each nuclear transmutation
, the "mass defect" mass is relocated to, or carried away by, other particles which are no longer a part of the original nucleus.
The nuclear binding energies and forces are on the order of a million times greater than the electron binding energies
of light atoms like hydrogen.
The mass defect of a nucleus represents the mass of the energy of binding of the nucleus, and is the difference between the mass
of a nucleus and the sum
of the masses of the nucleons of which it is composed. Determining the relevant nuclear binding energy encompasses three steps of calculation, which involves the creation of mass defect by removing the mass as released energy.
 Nuclear binding energy is explained by the basic principles involved in nuclear physics.
Nuclear binding energy is explained by the basic principles involved in nuclear physics.
s or radioactive decay
; the former are called endothermic
reactions and the latter exothermic
reactions. Energy is consumed or liberated because of differences in the nuclear binding energy between the incoming and outgoing products of the nuclear transmutation.
The best-known classes of exothermic nuclear transmutations are fission and fusion. Nuclear energy may be liberated by atomic fission, when heavy atomic nuclei like uranium
and plutonium
are broken apart into lighter nuclei. The energy from fission is used to generate electric power
in hundreds of locations worldwide. Nuclear energy is also released during atomic fusion, when light nuclei
like hydrogen
are combined to form heavier nuclei such as helium
. The Sun
and other stars use nuclear fusion to generate thermal energy which is later radiated from the surface, a type of stellar nucleosynthesis
. In any exothermic nuclear process, nuclear mass may ultimately be converted to thermal energy
, which is given off as heat and in doing so, carries away the mass with it.
In order to quantify the energy released or absorbed in any nuclear transmutation, one must know the nuclear binding energies of the nuclear components involved in the transmutation.
is :

The chemical energy of the hydrogen atom is the separation energy of an electron
from a proton. It is given by the Rydberg constant
from the Bohr
theory of the hydrogen
atom :
 .
.
The relative change of mass is the hydrogen chemical energy divided by its mass, here the proton mass :

It is so small as to be unmeasurable directly by weighing, but can be calculated, using the Einstein formula, from the measured chemical energy. Nuclear energy is usually "explained" by a hypothetical "strong force". However, it has been shown that it may be obtained by a similar formula with values intermediate between the Einstein mass and the Rydberg constant
:

This value is not far from the deuteron binding energy, , which is also the neutron-proton separation energy. The relative change in mass is :
, which is also the neutron-proton separation energy. The relative change in mass is :

Knowing the formulas characterizing the nuclear and chemical energies, one obtains their ratio :
 hundreds of thousands
hundreds of thousands
The two preceding calculated values are comparable with the one million and one per cent ratios evaluated from experimental binding energies.
The symbols used are
Mass energy of the proton
Nuclear energy
Chemical energy
Proton mass
Electron mass :
Fine structure constant :
(negative attracts positive). Furthermore, electrons are sometimes shared by neighboring atoms
or transferred to them (by processes of quantum physics), and this link between atoms creates many chemical compounds.
The force of electric attraction does not hold nuclei together, because all protons carry a positive charge and repel each other. Thus, Electric forces
do not hold nuclei together, because they act in the opposite direction. It has been established that binding neutrons to nuclei clearly requires a non-electrical attraction.
Therefore another force, called the nuclear force
, holds nuclei together. The nuclear force must be stronger than the electric repulsion at short distances, but weaker far away, or else different nuclei might tend to clump together. Therefore it has short-range characteristics. An analogy to the nuclear force is the force between two small magnets: magnets are very difficult to separate when stuck together, but once pulled a short distance apart, the force between them drops almost to zero.
Unlike gravity or electrical forces, the nuclear force is effective only at very short distances. At greater distances, the protons repel each other because they are positively charged, and charges of the same kind repel. For that reason, the protons forming the nuclei of ordinary hydrogen
; for instance, in a balloon filled with hydrogen; do not combine to form helium
(a process which also would require some to combine with electrons and become neutrons). They cannot get close enough for the nuclear force, which attracts them to each other, to become important. Only under conditions of extreme pressure
and temperature
(for example, within the core of a star
), can such a process take place.
a proton and a neutron; in helium, two protons and two neutrons, and in carbon, nitrogen and oxygen - six, seven and eight of each particle, respectively. A helium nucleus weighs less than the sum of the weights of its components. The same phenomenon is found for carbon, nitrogen and oxygen. For example, the carbon nucleus is slightly lighter than three helium nuclei which can be combined to make a carbon nucleus. This is an illustration of the mass defect.
's famous formula E = mc2, expressing the equivalence of energy and mass. By this formula, adding energy also increases mass (both weight and inertia), whereas removing energy decreases mass.
If a combination of particles contains extra energy - for instance, in a molecule of the explosive TNT - weighing it will reveal some extra mass, compared to its end products after an explosion. On the other hand, if one must inject energy to separate it into its components, the initial weight will be less than that of the components.
The latter scenario is the case with nuclei such as helium: to break them up into protons and neutrons, one must inject energy. On the other hand, if a process existed going in the opposite direction, by which hydrogen atoms could be combined to form helium, then energy would be released. The energy can be computed using E = mc2 for each nucleus, where m is the difference between the mass of the helium nucleus and the mass of four protons (plus two electrons, absorbed to create the neutrons of helium).
For elements heavier than oxygen, the energy which can be released by assembling them from lighter elements decreases, up to iron
. For nuclei heavier than iron, one actually releases energy by breaking them up into 2 fragments. That is how energy is extracted by breaking up uranium nuclei in nuclear power reactors.
The reason the trend reverses after iron is the growing positive charge of the nuclei. The electric force may be weaker than the nuclear force, but its range is greater: in an iron nucleus, each proton repels 25 other protons, while the nuclear force only binds close neighbors.
As nuclei grow bigger still, this disruptive effect becomes steadily more significant. By the time polonium
is reached (84 protons), nuclei can no longer accommodate their large positive charge, but emit their excess protons quite rapidly in the process of alpha radioactivity—the emission of helium nuclei, each containing two protons and two neutrons. (Helium nuclei are an especially stable combination.) This process becomes so rapid that nuclei with more than 94 protons are not found naturally on Earth.
process works as follows: five billion years ago, the new sun having formed when gravity pulled together a vast cloud of gas and dust, from which the Earth and other planets also arose. The gravitational pull released energy and heated the early Sun, much in the way Helmholtz proposed.
Heat is the motion of atoms and molecules: the higher the temperature, the greater is their velocity and the more violent are their collisions. When the temperature at the center of the newly-formed Sun became great enough for collisions between nuclei to overcome their electric repulsion, nuclei began to stick together and protons were combined into helium, with some protons changing in the process to neutrons (plus positrons, positive electrons, which combine with electrons and are destroyed). This released nuclear energy and kept up the high temperature of the Sun's core, and the heat also kept the gas pressure high, keeping the Sun puffed up and stopping gravity from pulling it together any more.
That is the "nuclear fusion" process which still takes place inside the Sun. Different nuclear reactions may predominate at different stages of the Sun's existence, including the proton-proton reaction and the carbon-nitrogen cycle which involves heavier nuclei, but whose final product is still the combination of protons to form helium.
A branch of physics, the study of controlled nuclear fusion
, has tried since the 1950s to derive useful power from nuclear fusion reactions which combine small nuclei into bigger ones, typically to heat boilers, whose steam could turn turbines and produce electricity. Unfortunately, no earthly laboratory can match one feature of the solar powerhouse; the great mass of the Sun, whose weight keeps the hot plasma compressed and confines the nuclear furnace to the Sun's core. Instead, physicists use strong magnetic fields to confine the plasma, and for fuel they use heavy forms of hydrogen, which burn more easily. Magnetic traps can be rather unstable, and any plasma hot enough and dense enough to undergo nuclear fusion tends to slip out of them after a short time. Even with ingenious tricks, the confinement in most cases lasts only a small fraction of a second.
For elements that weigh more than iron
(a nucleus with 26 protons), the fusion process no longer releases energy. In even heavier nuclei energy is consumed, not released, by combining similar sized nuclei. With such large nuclei, overcoming the electric repulsion (which affects all protons in the nucleus) requires more energy than what is released by the nuclear attraction (which is effective mainly between close neighbors). Conversely, energy could actually be released by breaking apart nuclei heavier than iron.
With the nuclei of elements heavier than lead
, the electric repulsion is so strong that some of them spontaneously eject positive fragments, usually nuclei of helium which form very stable combinations (alpha particles). This spontaneous break-up is one of the forms of radioactivity behavior exhibited by some nuclei.
Nuclei heavier than uranium
spontaneously break up too quickly to be found in nature, although they can be produced artificially. Generally, the heavier the nuclei are, the faster they spontaneously decay.
Iron nuclei (in particular isotope 56) are the most stable nuclei, and the best sources of energy are therefore nuclei whose weights are as far removed from iron as possible. One can combine the lightest ones—nuclei of hydrogen (protons)--to form nuclei of helium, and that is how the Sun generates its energy. Or else one can break up the heaviest ones—nuclei of uranium—into smaller fragments, and that is what nuclear power reactors do.
overcomes the repulsion and causes them to stick together. The nuclear force is a close-range force, and virtually no effect of this force is observed outside the nucleus. The nuclear force has a strong dependence on distance—it is a short range force. The nuclear force also pulls neutrons together, or neutrons and protons.
The energy of the nucleus is negative with regard to the energy of the particles pulled apart to infinite distance (just like the gravitational energy of planets of the solar system), because energy must be utilized to split a nucleus into its individual protons and neutrons. Mass spectrometers have measured the masses of nuclei, which are always less than the sum of the masses of protons and neutrons which form them, and the difference—by the formula E = mc2 --gives the binding energy of the nucleus.
The conversion of protons to neutrons is the result of another nuclear force, known as the weak (nuclear) force. The weak force, like the strong force, has a short range, but is much weaker than the strong force. The weak force tries to make the number of neutrons and protons into the most energetically stable configuration. For nuclei containing less than 40 particles, these numbers are usually about equal. Protons and neutrons are closely related and are sometimes collectively known as nucleons. As the number of particles increases toward a maximum of about 209, the number of neutrons to maintain stability begins to outstrip the number of protons, until the ratio of neutrons to protons is about three to two.
The protons of hydrogen will combine to helium only if they have enough velocity to overcome each other's mutual repulsion sufficiently to get within range of the strong nuclear attraction. This means the fusion only occurs within a very hot gas. Hydrogen hot enough for combining to helium requires an enormous pressure to keep it confined, but suitable conditions exist in the central regions
of the Sun, where such pressure is provided by the enormous weight of the layers above the core, pressed inwards by the Sun's strong gravity. The process of combining protons to form helium is an example of nuclear fusion.
The earth's oceans contain a large amount of hydrogen that could theoretically be used for fusion, and helium byproduct of fusion does not harm the environment, so some consider nuclear fusion to be a good alternative to supply humanities energy needs. Experiments to generate electricity from fusion have so far have only partially succeeded. Sufficiently hot hydrogen must be ionized and confined. One technique is to use very strong magnetic fields, because charged particles (like those trapped in the Earth's radiation belt) are guided by magnetic field lines. Fusion experiments also rely on heavy hydrogen which fuses more easily, and gas densities can be moderate. But even with these techniques far more net energy is consumed by the fusion experiments than is yielded by the process.
s of light nuclei, such as carbon, nitrogen and oxygen, the most stable combination of neutrons and of protons are when the numbers are equal (this continues to element 20, calcium). However, in heavier nuclei, the disruptive energy of protons increases, since they are confined to a tiny volume and repel each other. The energy of the strong force holding the nucleus together also increases, but at a slower rate, as if inside the nucleus, only nucleons close to each other are tightly bound, not ones more widely separated.
The net binding energy of a nucleus is that of the nuclear attraction, minus the disruptive energy of the electric force. As nuclei get heavier than helium, their net binding energy per nucleon (deduced from the difference in mass between the nucleus and the sum of masses of component nucleons) grows more and more slowly, reaching its peak at iron. As nucleons are added, the total nuclear binding energy always increases—but the total disruptive energy of electric forces (positive protons repelling other protons) also increases, and past iron, the second increase outweighs the first. 56Fe is the most efficiently bound nucleus.
To reduce the disruptive energy, the weak interaction allows the number of neutrons to exceed that of protons—for instance, the main isotope of iron has 26 protons and 30 neutrons. Isotopes also exist where the number of neutrons differs from the most stable number for that number of protons. If the ratio of protons is too far from stability, nucleons may spontaneously change from proton to neutron, or neutron to proton.
The two methods for this conversion are mediated by the weak force, and involve types of beta decay
. In the simplest beta decay, neutrons are converted to protons by emitting a negative electron and an antineutrino. This is always possible because neutrons are more massive than protons by an equivalent of about 2.5 electrons. In the opposite process, which only happens within a nucleus, and not to free particles, a proton may become a neutron by ejecting a positron
. This is permitted if enough energy is available between parent and daughter nuclides to do this (the required energy difference is equal to 1.022 MeV, which is the mass of 2 electrons). If the mass difference between parent and daughter is less than this, a proton-rich nucleus may still convert protons to neutrons by the process of electron capture
, in which a proton simply captures one of the atom's K orbital electrons, emits a neutrino, and becomes a neutron.
Among the heaviest nuclei, starting with tellurium nuclei (element 52) containing 106 or more nucleons, electric forces may be so destabilizing that entire chunks of the nucleus may be ejected, usually as alpha particles which consist of 2 protons and 2 neutrons (alpha particles are fast helium nuclei). Alpha particles are extremely stable. This type of decay becomes more and more probable as elements rise in atomic weight past 106.
The curve of binding energy is a graph which plots the binding energy per nucleon against atomic mass. This curve has its main peak at iron and then slowly decreases again, and also a narrow isolated peak at helium, which as noted is very stable. The heaviest nuclei in nature, uranium 238U, are unstable, but having a lifetime of 4.5 billion years, close to the age of the Earth, they are still relatively abundant; they (and other nuclei heavier than iron) may have formed in a supernova explosion preceding the formation of the solar system. The most common isotope of thorium, 232Th, also undergoes α particle emission, and its half-life (time over which half a number of atoms decays) is even longer, by several times. In each of these, radioactive decay produces daughter isotopes which are also unstable, starting a chain of decays which ends in some stable isotope of lead.
of atom
(s). The conversion of nuclear mass
-energy
to a form of energy which can remove some mass when the energy is removed, is consistent with the mass-energy equivalence
formula ΔE = Δmc², in which ΔE = energy release, Δm = mass defect, and c = the speed of light
in a vacuum
(a physical constant
). When this equation is used in this way, the mass "changes" only because it is removed from the system, not because it is converted or destroyed (the removed binding energy retains and accounts for the missing mass, which is a conserved quantity).
Nuclear energy was first discovered by French
physicist Henri Becquerel
in 1896, when he found that photographic plates stored in the dark near uranium
were blackened like X-ray
plates (X-rays had recently been discovered in 1895).
Nuclear chemistry
can be used as a form of alchemy
to turn lead
into gold
or change any atom to any other atom (although this may require many intermediate steps). Radionuclide
(radioisotope) production often involves irradiation of another isotope
(or more precisely a nuclide
), with alpha particle
s, beta particle
s, or gamma ray
s.
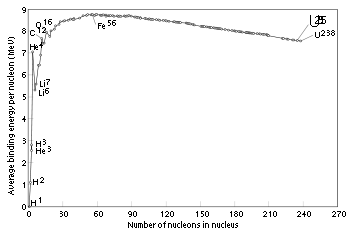
Nickel-62
has the highest binding energy per nucleon
of any isotope
. If an atom of lower average binding energy is changed into an atom of higher average binding energy, energy is given off. The chart shows that fusion of hydrogen
, the combination to form heavier atoms, releases energy, as does fission of uranium, the breaking up of a larger nucleus into smaller parts. Stability varies between isotopes: the isotope U-235
is much less stable than the more common U-238
.
Nuclear energy is released by three exoenergetic (or exothermic
) processes:
atom
s of that isotope, and mostly in MeV
. As a consequence, the listed mass deficits are not a measure for the stability or binding energy of isolated nuclei, but for the whole atoms. This has very practical reasons, because it is very hard to totally ion
ize heavy elements, i.e. strip them of all of their electron
s.
This practice is useful for other reasons, too: Stripping all the electrons from a heavy unstable nucleus (thus producing a bare nucleus) will change the lifetime of the nucleus, indicating that the nucleus cannot be treated independently (Experiments at the heavy ion accelerator GSI
). This is also evident from phenomena like electron capture
. Theoretically, in orbital models of heavy atoms, the electron orbits partially inside the nucleus (it doesn't orbit
in a strict sense, but has a non-vanishing probability of being located inside the nucleus).
A nuclear decay happens to the nucleus, meaning that properties ascribed to the nucleus will change in the event. In the field of physics the concept of "mass deficit" as a measure for "binding energy" means "mass deficit of the neutral atom" (not just the nucleus) and is a measure for stability of the whole atom.
 In the periodic table of elements, the series of light elements from hydrogen
In the periodic table of elements, the series of light elements from hydrogen
up to sodium
is observed to exhibit generally increasing binding energy per nucleon as the atomic mass
increases. This increase is generated by increasing forces per nucleon in the nucleus, as each additional nucleon is attracted by other nearby nucleons, and thus more tightly bound to the whole.
The region of increasing binding energy is followed by a region of relative stability (saturation) in the sequence from magnesium
through xenon
. In this region, the nucleus has become large enough that nuclear forces no longer completely extend efficiently across its width. Attractive nuclear forces in this region, as atomic mass increases, are nearly balanced by repellent electromagnetic forces between protons, as the atomic number
increases.
Finally, in elements heavier than xenon, there is a decrease in binding energy per nucleon as atomic number increases. In this region of nuclear size, electromagnetic repulsive forces are beginning to overcome the strong nuclear force attraction.
At the peak of binding energy, nickel-62
is the most tightly bound nucleus (per nucleon), followed by iron-58 and iron-56
. This is the approximate basic reason why iron and nickel are very common metals in planetary cores, since they are produced profusely as end products in supernova
e and in the final stages of silicon burning in stars. However, it is not binding energy per defined nucleon (as defined above) which controls which exact nuclei are made, because within stars, neutrons are free to convert to protons to release even more energy, per generic nucleon, if the result is a stable nucleus with a larger fraction of protons. Thus, iron-56 has the most binding energy of any group of 56 nucleons (because of its relatively larger fraction of protons), even while having less binding energy per nucleon than nickel-62, if this binding energy is computed by comparing Ni-62 with its disassembly products of 28 protons and 34 neutrons. In fact, it has been argued that photodisintegration
of 62Ni to form 56Fe may be energetically possible in an extremely hot star core, due to this beta decay conversion of neutrons to protons.
It is generally believed that iron-56 is more common than nickel isotopes in the universe for mechanistic reasons, because its unstable progenitor nickel-56 is copiously made by staged build-up of 14 helium nuclei inside supernovas, where it has no time to decay to iron before being released into the interstellar medium in a matter of a few minutes as a star explodes. However, nickel-56 then decays to iron-56
within a few weeks. The gamma ray light curve of such a process has been observed to happen in type IIa supernovae, such as SN1987a. In a star, there are no good ways to create nickel-62 by alpha-addition processes, or else there would presumably be more of this highly stable nuclide in the universe.
Nuclear fusion
produces energy by combining the very lightest elements into more tightly bound elements (such as hydrogen into helium
), and nuclear fission
produces energy by splitting the heaviest elements (such as uranium
and plutonium
) into more tightly bound elements (such as barium
and krypton
). Both processes produce energy, because middle-sized nuclei are the most tightly bound of all.
As seen above in the example of deuterium, nuclear binding energies are large enough that they may be easily measured as fractional mass
deficits, according to the equivalence of mass and energy. The atomic binding energy is simply the amount of energy (and mass) released, when a collection of free nucleons are joined together to form a nucleus
.
Nuclear binding energy can be computed from the difference in mass of a nucleus, and the sum of the masses of the number of free neutrons and protons that make up the nucleus. Once this mass difference, called the mass defect or mass deficiency, is known, Einstein's mass-energy equivalence
formula E = mc² can be used to compute the binding energy of any nucleus. Early nuclear physicists used to refer to computing this value as a "packing fraction" calculation.
For example, the atomic mass unit
(1 u) is defined to be 1/12 of the mass of a 12C atom—but the atomic mass of a 1H atom (which is a proton plus electron) is 1.007825 u, so each nucleon in 12C has lost, on average, about 0.8% of its mass in the form of binding energy.

where the binding energy is in MeV for the following numerical values of the constants: ;
;  ;
;  ;
;  ;
;  .
.
The first term is called the saturation contribution and ensures that the B.E. per nucleon is the same for all nuclei to a first approximation. The term
is called the saturation contribution and ensures that the B.E. per nucleon is the same for all nuclei to a first approximation. The term  is a surface tension effect and is proportional to the number of nucleons that are situated on the nuclear surface; it is largest for light nuclei. The term
is a surface tension effect and is proportional to the number of nucleons that are situated on the nuclear surface; it is largest for light nuclei. The term  is the Coulomb electrostatic repulsion; this becomes more important as
is the Coulomb electrostatic repulsion; this becomes more important as  increases. The symmetry correction term
increases. The symmetry correction term  takes into account the fact that in the absence of other effects the most stable arrangement has equal numbers of protons and neutrons; this is because the n-p interaction in a nucleus is stronger than either the n-n or p-p interaction. The pairing term
takes into account the fact that in the absence of other effects the most stable arrangement has equal numbers of protons and neutrons; this is because the n-p interaction in a nucleus is stronger than either the n-n or p-p interaction. The pairing term  is purely empirical; it is + for even-even nuclei and - for odd-odd nuclei.
is purely empirical; it is + for even-even nuclei and - for odd-odd nuclei.
= 931.494028(±0.000023) MeV. To calculate the "binding energy" we use the formula P*(mp+me) + N * mn - mnuclide where P denotes the number of protons of the nuclides and N its number of neutrons. We take
mp = 938.2723 Mev, me = 0.5110 MeV and mn = 939.5656 MeV. The letter A denotes the sum of P and N (number of nucleons in the nuclide). If we assume the reference nucleon has the mass of a neutron (so that all "total" binding energies calculated are maximal) we could define the total binding energy as the difference from the mass of the nucleus, and the mass of a collection of A free neutrons. In other words, it would be [(P+N)* mn] - mnuclide. The "total binding energy per nucleon" would be this value divided by A.
56Fe has the lowest nucleon-specific mass of the four nuclides listed in this table, but this does not imply it is the strongest bound atom per hadron, unless the choice of beginning hadrons is completely free. Iron releases the largest energy if any 56 nucleons are allowed to build a nuclide—changing one to another if necessary, The highest binding energy per hadron, with the hadrons starting as the same number of protons Z and total nucleons A as in the bound nucleus, is 62Ni. Thus, the true absolute value of the total binding energy of a nucleus depends on what we are allowed to construct the nucleus out of. If all nuclei of mass number A were to be allowed to be constructed of A neutrons, then Fe-56 would release the most energy per nucleon, since it has a larger fraction of protons than Ni-62. However, if nucleons are required to be constructed of only the same number of protons and neutrons that they contain, then nickel-62 is the most tightly bound nucleus, per nucleon.
In the table above it can be seen that the decay of a neutron, as well as the transformation of tritium into helium-3, releases energy; hence, it manifests a stronger bound new state when measured against the mass of an equal number of neutrons (and also a lighter state per number of total hadrons). Such reactions are not driven by changes in binding energies as calculated from previously fixed N and Z numbers of neutrons and protons, but rather in decreases in the total mass of the nuclide/per nucleon, with the reaction.
Atomic nucleus
The nucleus is the very dense region consisting of protons and neutrons at the center of an atom. It was discovered in 1911, as a result of Ernest Rutherford's interpretation of the famous 1909 Rutherford experiment performed by Hans Geiger and Ernest Marsden, under the direction of Rutherford. The...
into its component parts. The component parts are neutrons and protons, which are collectively called nucleon
Nucleon
In physics, a nucleon is a collective name for two particles: the neutron and the proton. These are the two constituents of the atomic nucleus. Until the 1960s, the nucleons were thought to be elementary particles...
s. If the binding energy
Binding energy
Binding energy is the mechanical energy required to disassemble a whole into separate parts. A bound system typically has a lower potential energy than its constituent parts; this is what keeps the system together—often this means that energy is released upon the creation of a bound state...
for the products is higher when light nuclei fuse, or when heavy nuclei split, either of these processes will result in a release of the extra binding energy, and this energy is referred to as nuclear energy or, more loosely, nuclear power
Nuclear power
Nuclear power is the use of sustained nuclear fission to generate heat and electricity. Nuclear power plants provide about 6% of the world's energy and 13–14% of the world's electricity, with the U.S., France, and Japan together accounting for about 50% of nuclear generated electricity...
.
The mass
Mass
Mass can be defined as a quantitive measure of the resistance an object has to change in its velocity.In physics, mass commonly refers to any of the following three properties of matter, which have been shown experimentally to be equivalent:...
of an atom's nucleus is always less than the sum of the individual masses of the constituent protons and neutrons. This notable difference is a measure of the nuclear binding energy, which is a result of forces that hold the nucleus together. Because these forces result in the removal of energy when the nucleus is formed, and this energy has mass, mass is removed from the total mass of the original particles, and the mass is missing in the resulting nucleus. This missing mass is known as the mass defect, and represents the energy released when the nucleus is formed. When the nucleus splits
Nuclear transmutation
Nuclear transmutation is the conversion of one chemical element or isotope into another. In other words, atoms of one element can be changed into atoms of other element by 'transmutation'...
into pieces, this energy may be emitted as photons (gamma rays) or as the mass or kinetic energy of a number of different ejected particles. Total mass is conserved throughout the process, and during each nuclear transmutation
Nuclear transmutation
Nuclear transmutation is the conversion of one chemical element or isotope into another. In other words, atoms of one element can be changed into atoms of other element by 'transmutation'...
, the "mass defect" mass is relocated to, or carried away by, other particles which are no longer a part of the original nucleus.
The nuclear binding energies and forces are on the order of a million times greater than the electron binding energies
Ionization energy
The ionization energy of a chemical species, i.e. an atom or molecule, is the energy required to remove an electron from the species to a practically infinite distance. Large atoms or molecules have a low ionization energy, while small molecules tend to have higher ionization energies.The property...
of light atoms like hydrogen.
The mass defect of a nucleus represents the mass of the energy of binding of the nucleus, and is the difference between the mass
Mass
Mass can be defined as a quantitive measure of the resistance an object has to change in its velocity.In physics, mass commonly refers to any of the following three properties of matter, which have been shown experimentally to be equivalent:...
of a nucleus and the sum
SUM
SUM can refer to:* The State University of Management* Soccer United Marketing* Society for the Establishment of Useful Manufactures* StartUp-Manager* Software User’s Manual,as from DOD-STD-2 167A, and MIL-STD-498...
of the masses of the nucleons of which it is composed. Determining the relevant nuclear binding energy encompasses three steps of calculation, which involves the creation of mass defect by removing the mass as released energy.
Introduction

Nuclear energy
An absorption or release of nuclear energy occurs in nuclear reactionNuclear reaction
In nuclear physics and nuclear chemistry, a nuclear reaction is semantically considered to be the process in which two nuclei, or else a nucleus of an atom and a subatomic particle from outside the atom, collide to produce products different from the initial particles...
s or radioactive decay
Radioactive decay
Radioactive decay is the process by which an atomic nucleus of an unstable atom loses energy by emitting ionizing particles . The emission is spontaneous, in that the atom decays without any physical interaction with another particle from outside the atom...
; the former are called endothermic
Endothermic
In thermodynamics, the word endothermic describes a process or reaction in which the system absorbs energy from the surroundings in the form of heat. Its etymology stems from the prefix endo- and the Greek word thermasi,...
reactions and the latter exothermic
Exothermic
In thermodynamics, the term exothermic describes a process or reaction that releases energy from the system, usually in the form of heat, but also in the form of light , electricity , or sound...
reactions. Energy is consumed or liberated because of differences in the nuclear binding energy between the incoming and outgoing products of the nuclear transmutation.
The best-known classes of exothermic nuclear transmutations are fission and fusion. Nuclear energy may be liberated by atomic fission, when heavy atomic nuclei like uranium
Uranium
Uranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons...
and plutonium
Plutonium
Plutonium is a transuranic radioactive chemical element with the chemical symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, forming a dull coating when oxidized. The element normally exhibits six allotropes and four oxidation...
are broken apart into lighter nuclei. The energy from fission is used to generate electric power
Electric power
Electric power is the rate at which electric energy is transferred by an electric circuit. The SI unit of power is the watt.-Circuits:Electric power, like mechanical power, is represented by the letter P in electrical equations...
in hundreds of locations worldwide. Nuclear energy is also released during atomic fusion, when light nuclei
Atomic nucleus
The nucleus is the very dense region consisting of protons and neutrons at the center of an atom. It was discovered in 1911, as a result of Ernest Rutherford's interpretation of the famous 1909 Rutherford experiment performed by Hans Geiger and Ernest Marsden, under the direction of Rutherford. The...
like hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
are combined to form heavier nuclei such as helium
Helium
Helium is the chemical element with atomic number 2 and an atomic weight of 4.002602, which is represented by the symbol He. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas that heads the noble gas group in the periodic table...
. The Sun
Sun
The Sun is the star at the center of the Solar System. It is almost perfectly spherical and consists of hot plasma interwoven with magnetic fields...
and other stars use nuclear fusion to generate thermal energy which is later radiated from the surface, a type of stellar nucleosynthesis
Stellar nucleosynthesis
Stellar nucleosynthesis is the collective term for the nuclear reactions taking place in stars to build the nuclei of the elements heavier than hydrogen. Some small quantity of these reactions also occur on the stellar surface under various circumstances...
. In any exothermic nuclear process, nuclear mass may ultimately be converted to thermal energy
Thermal energy
Thermal energy is the part of the total internal energy of a thermodynamic system or sample of matter that results in the system's temperature....
, which is given off as heat and in doing so, carries away the mass with it.
In order to quantify the energy released or absorbed in any nuclear transmutation, one must know the nuclear binding energies of the nuclear components involved in the transmutation.
Nuclear and chemical energies
Nuclear energy is typically hundreds of thousands or millions of times greater than chemical energy or approximately 1% of the mass energy from the Einstein mass formula. The mass of a protonProton
The proton is a subatomic particle with the symbol or and a positive electric charge of 1 elementary charge. One or more protons are present in the nucleus of each atom, along with neutrons. The number of protons in each atom is its atomic number....
is :

The chemical energy of the hydrogen atom is the separation energy of an electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
from a proton. It is given by the Rydberg constant
Rydberg constant
The Rydberg constant, symbol R∞, named after the Swedish physicist Johannes Rydberg, is a physical constant relating to atomic spectra in the science of spectroscopy. Rydberg initially determined its value empirically from spectroscopy, but Niels Bohr later showed that its value could be calculated...
from the Bohr
Bohr
Bohr may refer to:People:* Niels Bohr , Danish atomic physicist, Nobel Prize in physics 1922* Aage Bohr , Danish nuclear physicist, Nobel Prize in physics 1975, son of Niels Bohr...
theory of the hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
atom :
 .
.The relative change of mass is the hydrogen chemical energy divided by its mass, here the proton mass :

It is so small as to be unmeasurable directly by weighing, but can be calculated, using the Einstein formula, from the measured chemical energy. Nuclear energy is usually "explained" by a hypothetical "strong force". However, it has been shown that it may be obtained by a similar formula with values intermediate between the Einstein mass and the Rydberg constant
Rydberg constant
The Rydberg constant, symbol R∞, named after the Swedish physicist Johannes Rydberg, is a physical constant relating to atomic spectra in the science of spectroscopy. Rydberg initially determined its value empirically from spectroscopy, but Niels Bohr later showed that its value could be calculated...
:

This value is not far from the deuteron binding energy,
 , which is also the neutron-proton separation energy. The relative change in mass is :
, which is also the neutron-proton separation energy. The relative change in mass is :
Knowing the formulas characterizing the nuclear and chemical energies, one obtains their ratio :
 hundreds of thousands
hundreds of thousandsThe two preceding calculated values are comparable with the one million and one per cent ratios evaluated from experimental binding energies.
The symbols used are
Mass energy of the proton

Nuclear energy

Chemical energy

Proton mass

Electron mass :

Fine structure constant :

The nuclear force
Electrons and nuclei are kept together by electric attractionElectric charge
Electric charge is a physical property of matter that causes it to experience a force when near other electrically charged matter. Electric charge comes in two types, called positive and negative. Two positively charged substances, or objects, experience a mutual repulsive force, as do two...
(negative attracts positive). Furthermore, electrons are sometimes shared by neighboring atoms
Covalent bond
A covalent bond is a form of chemical bonding that is characterized by the sharing of pairs of electrons between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding....
or transferred to them (by processes of quantum physics), and this link between atoms creates many chemical compounds.
The force of electric attraction does not hold nuclei together, because all protons carry a positive charge and repel each other. Thus, Electric forces
Electromagnetism
Electromagnetism is one of the four fundamental interactions in nature. The other three are the strong interaction, the weak interaction and gravitation...
do not hold nuclei together, because they act in the opposite direction. It has been established that binding neutrons to nuclei clearly requires a non-electrical attraction.
Therefore another force, called the nuclear force
Nuclear force
The nuclear force is the force between two or more nucleons. It is responsible for binding of protons and neutrons into atomic nuclei. The energy released causes the masses of nuclei to be less than the total mass of the protons and neutrons which form them...
, holds nuclei together. The nuclear force must be stronger than the electric repulsion at short distances, but weaker far away, or else different nuclei might tend to clump together. Therefore it has short-range characteristics. An analogy to the nuclear force is the force between two small magnets: magnets are very difficult to separate when stuck together, but once pulled a short distance apart, the force between them drops almost to zero.
Unlike gravity or electrical forces, the nuclear force is effective only at very short distances. At greater distances, the protons repel each other because they are positively charged, and charges of the same kind repel. For that reason, the protons forming the nuclei of ordinary hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
; for instance, in a balloon filled with hydrogen; do not combine to form helium
Helium
Helium is the chemical element with atomic number 2 and an atomic weight of 4.002602, which is represented by the symbol He. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas that heads the noble gas group in the periodic table...
(a process which also would require some to combine with electrons and become neutrons). They cannot get close enough for the nuclear force, which attracts them to each other, to become important. Only under conditions of extreme pressure
Pressure
Pressure is the force per unit area applied in a direction perpendicular to the surface of an object. Gauge pressure is the pressure relative to the local atmospheric or ambient pressure.- Definition :...
and temperature
Temperature
Temperature is a physical property of matter that quantitatively expresses the common notions of hot and cold. Objects of low temperature are cold, while various degrees of higher temperatures are referred to as warm or hot...
(for example, within the core of a star
Star
A star is a massive, luminous sphere of plasma held together by gravity. At the end of its lifetime, a star can also contain a proportion of degenerate matter. The nearest star to Earth is the Sun, which is the source of most of the energy on Earth...
), can such a process take place.
Physics of nuclei
The nuclei of atoms are found in many different sizes. In hydrogen they contain just one proton, in heavy hydrogenDeuterium
Deuterium, also called heavy hydrogen, is one of two stable isotopes of hydrogen. It has a natural abundance in Earth's oceans of about one atom in of hydrogen . Deuterium accounts for approximately 0.0156% of all naturally occurring hydrogen in Earth's oceans, while the most common isotope ...
a proton and a neutron; in helium, two protons and two neutrons, and in carbon, nitrogen and oxygen - six, seven and eight of each particle, respectively. A helium nucleus weighs less than the sum of the weights of its components. The same phenomenon is found for carbon, nitrogen and oxygen. For example, the carbon nucleus is slightly lighter than three helium nuclei which can be combined to make a carbon nucleus. This is an illustration of the mass defect.
Mass defect
The fundamental reason for the "mass defect" is Albert EinsteinAlbert Einstein
Albert Einstein was a German-born theoretical physicist who developed the theory of general relativity, effecting a revolution in physics. For this achievement, Einstein is often regarded as the father of modern physics and one of the most prolific intellects in human history...
's famous formula E = mc2, expressing the equivalence of energy and mass. By this formula, adding energy also increases mass (both weight and inertia), whereas removing energy decreases mass.
If a combination of particles contains extra energy - for instance, in a molecule of the explosive TNT - weighing it will reveal some extra mass, compared to its end products after an explosion. On the other hand, if one must inject energy to separate it into its components, the initial weight will be less than that of the components.
The latter scenario is the case with nuclei such as helium: to break them up into protons and neutrons, one must inject energy. On the other hand, if a process existed going in the opposite direction, by which hydrogen atoms could be combined to form helium, then energy would be released. The energy can be computed using E = mc2 for each nucleus, where m is the difference between the mass of the helium nucleus and the mass of four protons (plus two electrons, absorbed to create the neutrons of helium).
For elements heavier than oxygen, the energy which can be released by assembling them from lighter elements decreases, up to iron
Iron
Iron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust...
. For nuclei heavier than iron, one actually releases energy by breaking them up into 2 fragments. That is how energy is extracted by breaking up uranium nuclei in nuclear power reactors.
The reason the trend reverses after iron is the growing positive charge of the nuclei. The electric force may be weaker than the nuclear force, but its range is greater: in an iron nucleus, each proton repels 25 other protons, while the nuclear force only binds close neighbors.
As nuclei grow bigger still, this disruptive effect becomes steadily more significant. By the time polonium
Polonium
Polonium is a chemical element with the symbol Po and atomic number 84, discovered in 1898 by Marie Skłodowska-Curie and Pierre Curie. A rare and highly radioactive element, polonium is chemically similar to bismuth and tellurium, and it occurs in uranium ores. Polonium has been studied for...
is reached (84 protons), nuclei can no longer accommodate their large positive charge, but emit their excess protons quite rapidly in the process of alpha radioactivity—the emission of helium nuclei, each containing two protons and two neutrons. (Helium nuclei are an especially stable combination.) This process becomes so rapid that nuclei with more than 94 protons are not found naturally on Earth.
Solar binding energy
The nuclear fusionNuclear fusion
Nuclear fusion is the process by which two or more atomic nuclei join together, or "fuse", to form a single heavier nucleus. This is usually accompanied by the release or absorption of large quantities of energy...
process works as follows: five billion years ago, the new sun having formed when gravity pulled together a vast cloud of gas and dust, from which the Earth and other planets also arose. The gravitational pull released energy and heated the early Sun, much in the way Helmholtz proposed.
Heat is the motion of atoms and molecules: the higher the temperature, the greater is their velocity and the more violent are their collisions. When the temperature at the center of the newly-formed Sun became great enough for collisions between nuclei to overcome their electric repulsion, nuclei began to stick together and protons were combined into helium, with some protons changing in the process to neutrons (plus positrons, positive electrons, which combine with electrons and are destroyed). This released nuclear energy and kept up the high temperature of the Sun's core, and the heat also kept the gas pressure high, keeping the Sun puffed up and stopping gravity from pulling it together any more.
That is the "nuclear fusion" process which still takes place inside the Sun. Different nuclear reactions may predominate at different stages of the Sun's existence, including the proton-proton reaction and the carbon-nitrogen cycle which involves heavier nuclei, but whose final product is still the combination of protons to form helium.
A branch of physics, the study of controlled nuclear fusion
Nuclear power
Nuclear power is the use of sustained nuclear fission to generate heat and electricity. Nuclear power plants provide about 6% of the world's energy and 13–14% of the world's electricity, with the U.S., France, and Japan together accounting for about 50% of nuclear generated electricity...
, has tried since the 1950s to derive useful power from nuclear fusion reactions which combine small nuclei into bigger ones, typically to heat boilers, whose steam could turn turbines and produce electricity. Unfortunately, no earthly laboratory can match one feature of the solar powerhouse; the great mass of the Sun, whose weight keeps the hot plasma compressed and confines the nuclear furnace to the Sun's core. Instead, physicists use strong magnetic fields to confine the plasma, and for fuel they use heavy forms of hydrogen, which burn more easily. Magnetic traps can be rather unstable, and any plasma hot enough and dense enough to undergo nuclear fusion tends to slip out of them after a short time. Even with ingenious tricks, the confinement in most cases lasts only a small fraction of a second.
Combining nuclei
Small nuclei that are larger than hydrogen can combine into bigger ones and release energy, but in combining such nuclei, the amount of energy released is much smaller compared to hydrogen fusion. The reason is that while the overall process releases energy from letting the nuclear attraction do its work, energy must first be injected to force together positively charged protons, which also repel each other with their electric charge.For elements that weigh more than iron
Iron
Iron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust...
(a nucleus with 26 protons), the fusion process no longer releases energy. In even heavier nuclei energy is consumed, not released, by combining similar sized nuclei. With such large nuclei, overcoming the electric repulsion (which affects all protons in the nucleus) requires more energy than what is released by the nuclear attraction (which is effective mainly between close neighbors). Conversely, energy could actually be released by breaking apart nuclei heavier than iron.
With the nuclei of elements heavier than lead
Lead
Lead is a main-group element in the carbon group with the symbol Pb and atomic number 82. Lead is a soft, malleable poor metal. It is also counted as one of the heavy metals. Metallic lead has a bluish-white color after being freshly cut, but it soon tarnishes to a dull grayish color when exposed...
, the electric repulsion is so strong that some of them spontaneously eject positive fragments, usually nuclei of helium which form very stable combinations (alpha particles). This spontaneous break-up is one of the forms of radioactivity behavior exhibited by some nuclei.
Nuclei heavier than uranium
Uranium
Uranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons...
spontaneously break up too quickly to be found in nature, although they can be produced artificially. Generally, the heavier the nuclei are, the faster they spontaneously decay.
Iron nuclei (in particular isotope 56) are the most stable nuclei, and the best sources of energy are therefore nuclei whose weights are as far removed from iron as possible. One can combine the lightest ones—nuclei of hydrogen (protons)--to form nuclei of helium, and that is how the Sun generates its energy. Or else one can break up the heaviest ones—nuclei of uranium—into smaller fragments, and that is what nuclear power reactors do.
Nuclear binding energy
An example that illustrates nuclear binding energy is carbon nucleus of 12C, which contains 6 protons and 6 neutrons. The protons are all positively charged and repel each other, but the nuclear forceNuclear force
The nuclear force is the force between two or more nucleons. It is responsible for binding of protons and neutrons into atomic nuclei. The energy released causes the masses of nuclei to be less than the total mass of the protons and neutrons which form them...
overcomes the repulsion and causes them to stick together. The nuclear force is a close-range force, and virtually no effect of this force is observed outside the nucleus. The nuclear force has a strong dependence on distance—it is a short range force. The nuclear force also pulls neutrons together, or neutrons and protons.
The energy of the nucleus is negative with regard to the energy of the particles pulled apart to infinite distance (just like the gravitational energy of planets of the solar system), because energy must be utilized to split a nucleus into its individual protons and neutrons. Mass spectrometers have measured the masses of nuclei, which are always less than the sum of the masses of protons and neutrons which form them, and the difference—by the formula E = mc2 --gives the binding energy of the nucleus.
Nuclear fusion
The binding energy of helium is the energy source of the Sun and of most stars. The sun is composed of 74 percent hydrogen (measured by mass), an element whose nucleus is a single proton. Energy is released in the sun when 4 protons combine into a helium nucleus, a process in which two of them are also converted to neutrons.The conversion of protons to neutrons is the result of another nuclear force, known as the weak (nuclear) force. The weak force, like the strong force, has a short range, but is much weaker than the strong force. The weak force tries to make the number of neutrons and protons into the most energetically stable configuration. For nuclei containing less than 40 particles, these numbers are usually about equal. Protons and neutrons are closely related and are sometimes collectively known as nucleons. As the number of particles increases toward a maximum of about 209, the number of neutrons to maintain stability begins to outstrip the number of protons, until the ratio of neutrons to protons is about three to two.
The protons of hydrogen will combine to helium only if they have enough velocity to overcome each other's mutual repulsion sufficiently to get within range of the strong nuclear attraction. This means the fusion only occurs within a very hot gas. Hydrogen hot enough for combining to helium requires an enormous pressure to keep it confined, but suitable conditions exist in the central regions
Solar core
The core of the Sun is considered to extend from the center to about 0.2 to 0.25 solar radius. It is the hottest part of the Sun and of the Solar System. It has a density of up to 150 g/cm³ and a temperature of close to 15,000,000 kelvin...
of the Sun, where such pressure is provided by the enormous weight of the layers above the core, pressed inwards by the Sun's strong gravity. The process of combining protons to form helium is an example of nuclear fusion.
The earth's oceans contain a large amount of hydrogen that could theoretically be used for fusion, and helium byproduct of fusion does not harm the environment, so some consider nuclear fusion to be a good alternative to supply humanities energy needs. Experiments to generate electricity from fusion have so far have only partially succeeded. Sufficiently hot hydrogen must be ionized and confined. One technique is to use very strong magnetic fields, because charged particles (like those trapped in the Earth's radiation belt) are guided by magnetic field lines. Fusion experiments also rely on heavy hydrogen which fuses more easily, and gas densities can be moderate. But even with these techniques far more net energy is consumed by the fusion experiments than is yielded by the process.
The binding energy maximum and ways to approach it by decay
In the main isotopeIsotope
Isotopes are variants of atoms of a particular chemical element, which have differing numbers of neutrons. Atoms of a particular element by definition must contain the same number of protons but may have a distinct number of neutrons which differs from atom to atom, without changing the designation...
s of light nuclei, such as carbon, nitrogen and oxygen, the most stable combination of neutrons and of protons are when the numbers are equal (this continues to element 20, calcium). However, in heavier nuclei, the disruptive energy of protons increases, since they are confined to a tiny volume and repel each other. The energy of the strong force holding the nucleus together also increases, but at a slower rate, as if inside the nucleus, only nucleons close to each other are tightly bound, not ones more widely separated.
The net binding energy of a nucleus is that of the nuclear attraction, minus the disruptive energy of the electric force. As nuclei get heavier than helium, their net binding energy per nucleon (deduced from the difference in mass between the nucleus and the sum of masses of component nucleons) grows more and more slowly, reaching its peak at iron. As nucleons are added, the total nuclear binding energy always increases—but the total disruptive energy of electric forces (positive protons repelling other protons) also increases, and past iron, the second increase outweighs the first. 56Fe is the most efficiently bound nucleus.
To reduce the disruptive energy, the weak interaction allows the number of neutrons to exceed that of protons—for instance, the main isotope of iron has 26 protons and 30 neutrons. Isotopes also exist where the number of neutrons differs from the most stable number for that number of protons. If the ratio of protons is too far from stability, nucleons may spontaneously change from proton to neutron, or neutron to proton.
The two methods for this conversion are mediated by the weak force, and involve types of beta decay
Beta decay
In nuclear physics, beta decay is a type of radioactive decay in which a beta particle is emitted from an atom. There are two types of beta decay: beta minus and beta plus. In the case of beta decay that produces an electron emission, it is referred to as beta minus , while in the case of a...
. In the simplest beta decay, neutrons are converted to protons by emitting a negative electron and an antineutrino. This is always possible because neutrons are more massive than protons by an equivalent of about 2.5 electrons. In the opposite process, which only happens within a nucleus, and not to free particles, a proton may become a neutron by ejecting a positron
Positron
The positron or antielectron is the antiparticle or the antimatter counterpart of the electron. The positron has an electric charge of +1e, a spin of ½, and has the same mass as an electron...
. This is permitted if enough energy is available between parent and daughter nuclides to do this (the required energy difference is equal to 1.022 MeV, which is the mass of 2 electrons). If the mass difference between parent and daughter is less than this, a proton-rich nucleus may still convert protons to neutrons by the process of electron capture
Electron capture
Electron capture is a process in which a proton-rich nuclide absorbs an inner atomic electron and simultaneously emits a neutrino...
, in which a proton simply captures one of the atom's K orbital electrons, emits a neutrino, and becomes a neutron.
Among the heaviest nuclei, starting with tellurium nuclei (element 52) containing 106 or more nucleons, electric forces may be so destabilizing that entire chunks of the nucleus may be ejected, usually as alpha particles which consist of 2 protons and 2 neutrons (alpha particles are fast helium nuclei). Alpha particles are extremely stable. This type of decay becomes more and more probable as elements rise in atomic weight past 106.
The curve of binding energy is a graph which plots the binding energy per nucleon against atomic mass. This curve has its main peak at iron and then slowly decreases again, and also a narrow isolated peak at helium, which as noted is very stable. The heaviest nuclei in nature, uranium 238U, are unstable, but having a lifetime of 4.5 billion years, close to the age of the Earth, they are still relatively abundant; they (and other nuclei heavier than iron) may have formed in a supernova explosion preceding the formation of the solar system. The most common isotope of thorium, 232Th, also undergoes α particle emission, and its half-life (time over which half a number of atoms decays) is even longer, by several times. In each of these, radioactive decay produces daughter isotopes which are also unstable, starting a chain of decays which ends in some stable isotope of lead.
Determining nuclear binding energy
Calculation can be employed to determine the nuclear binding energy of nuclei. The calculation uses three general steps:- Determining the Mass defect
- Conversion of mass defect into energy
- Expressing nuclear binding energy as energy per mole of atoms, or as energy per nucleon.
Conversion of mass defect into energy
Mass defect is defined as the difference between the mass of a nucleus, and the sum of the masses of the nucleons of which it is composed. The mass defect is determined by calculating three quantities. These are: the actual mass of the nucleus, the composition of the nucleus (number of protons and of neutrons), and the masses of a proton and of a neutron. This is then followed by converting the mass defect into energy. This quantity is the nuclear binding energy, however it must be expressed as energy per mole of atoms or as energy per nucleon.Fission and fusion
Nuclear energy is released by the splitting (fission) or merging together (fusion) of the nucleiAtomic nucleus
The nucleus is the very dense region consisting of protons and neutrons at the center of an atom. It was discovered in 1911, as a result of Ernest Rutherford's interpretation of the famous 1909 Rutherford experiment performed by Hans Geiger and Ernest Marsden, under the direction of Rutherford. The...
of atom
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
(s). The conversion of nuclear mass
Mass
Mass can be defined as a quantitive measure of the resistance an object has to change in its velocity.In physics, mass commonly refers to any of the following three properties of matter, which have been shown experimentally to be equivalent:...
-energy
Energy
In physics, energy is an indirectly observed quantity. It is often understood as the ability a physical system has to do work on other physical systems...
to a form of energy which can remove some mass when the energy is removed, is consistent with the mass-energy equivalence
Mass-energy equivalence
In physics, mass–energy equivalence is the concept that the mass of a body is a measure of its energy content. In this concept, mass is a property of all energy, and energy is a property of all mass, and the two properties are connected by a constant...
formula ΔE = Δmc², in which ΔE = energy release, Δm = mass defect, and c = the speed of light
Speed of light
The speed of light in vacuum, usually denoted by c, is a physical constant important in many areas of physics. Its value is 299,792,458 metres per second, a figure that is exact since the length of the metre is defined from this constant and the international standard for time...
in a vacuum
Vacuum
In everyday usage, vacuum is a volume of space that is essentially empty of matter, such that its gaseous pressure is much less than atmospheric pressure. The word comes from the Latin term for "empty". A perfect vacuum would be one with no particles in it at all, which is impossible to achieve in...
(a physical constant
Physical constant
A physical constant is a physical quantity that is generally believed to be both universal in nature and constant in time. It can be contrasted with a mathematical constant, which is a fixed numerical value but does not directly involve any physical measurement.There are many physical constants in...
). When this equation is used in this way, the mass "changes" only because it is removed from the system, not because it is converted or destroyed (the removed binding energy retains and accounts for the missing mass, which is a conserved quantity).
Nuclear energy was first discovered by French
France
The French Republic , The French Republic , The French Republic , (commonly known as France , is a unitary semi-presidential republic in Western Europe with several overseas territories and islands located on other continents and in the Indian, Pacific, and Atlantic oceans. Metropolitan France...
physicist Henri Becquerel
Henri Becquerel
Antoine Henri Becquerel was a French physicist, Nobel laureate, and the discoverer of radioactivity along with Marie Curie and Pierre Curie, for which all three won the 1903 Nobel Prize in Physics.-Early life:...
in 1896, when he found that photographic plates stored in the dark near uranium
Uranium
Uranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons...
were blackened like X-ray
X-ray
X-radiation is a form of electromagnetic radiation. X-rays have a wavelength in the range of 0.01 to 10 nanometers, corresponding to frequencies in the range 30 petahertz to 30 exahertz and energies in the range 120 eV to 120 keV. They are shorter in wavelength than UV rays and longer than gamma...
plates (X-rays had recently been discovered in 1895).
Nuclear chemistry
Nuclear chemistry
Nuclear chemistry is the subfield of chemistry dealing with radioactivity, nuclear processes and nuclear properties.It is the chemistry of radioactive elements such as the actinides, radium and radon together with the chemistry associated with equipment which are designed to perform nuclear...
can be used as a form of alchemy
Alchemy
Alchemy is an influential philosophical tradition whose early practitioners’ claims to profound powers were known from antiquity. The defining objectives of alchemy are varied; these include the creation of the fabled philosopher's stone possessing powers including the capability of turning base...
to turn lead
Lead
Lead is a main-group element in the carbon group with the symbol Pb and atomic number 82. Lead is a soft, malleable poor metal. It is also counted as one of the heavy metals. Metallic lead has a bluish-white color after being freshly cut, but it soon tarnishes to a dull grayish color when exposed...
into gold
Gold
Gold is a chemical element with the symbol Au and an atomic number of 79. Gold is a dense, soft, shiny, malleable and ductile metal. Pure gold has a bright yellow color and luster traditionally considered attractive, which it maintains without oxidizing in air or water. Chemically, gold is a...
or change any atom to any other atom (although this may require many intermediate steps). Radionuclide
Radionuclide
A radionuclide is an atom with an unstable nucleus, which is a nucleus characterized by excess energy available to be imparted either to a newly created radiation particle within the nucleus or to an atomic electron. The radionuclide, in this process, undergoes radioactive decay, and emits gamma...
(radioisotope) production often involves irradiation of another isotope
Isotope
Isotopes are variants of atoms of a particular chemical element, which have differing numbers of neutrons. Atoms of a particular element by definition must contain the same number of protons but may have a distinct number of neutrons which differs from atom to atom, without changing the designation...
(or more precisely a nuclide
Nuclide
A nuclide is an atomic species characterized by the specific constitution of its nucleus, i.e., by its number of protons Z, its number of neutrons N, and its nuclear energy state....
), with alpha particle
Alpha particle
Alpha particles consist of two protons and two neutrons bound together into a particle identical to a helium nucleus, which is classically produced in the process of alpha decay, but may be produced also in other ways and given the same name...
s, beta particle
Beta particle
Beta particles are high-energy, high-speed electrons or positrons emitted by certain types of radioactive nuclei such as potassium-40. The beta particles emitted are a form of ionizing radiation also known as beta rays. The production of beta particles is termed beta decay...
s, or gamma ray
Gamma ray
Gamma radiation, also known as gamma rays or hyphenated as gamma-rays and denoted as γ, is electromagnetic radiation of high frequency . Gamma rays are usually naturally produced on Earth by decay of high energy states in atomic nuclei...
s.
Nickel-62
Nickel-62
Nickel-62 is an isotope of nickel having 28 protons and 34 neutrons.It is a stable isotope, with the highest binding energy per nucleon of any known nuclide . It is often stated that 56Fe is the "most stable nucleus", but actually 56Fe has the lowest mass per nucleon of all nuclides...
has the highest binding energy per nucleon
Nucleon
In physics, a nucleon is a collective name for two particles: the neutron and the proton. These are the two constituents of the atomic nucleus. Until the 1960s, the nucleons were thought to be elementary particles...
of any isotope
Isotope
Isotopes are variants of atoms of a particular chemical element, which have differing numbers of neutrons. Atoms of a particular element by definition must contain the same number of protons but may have a distinct number of neutrons which differs from atom to atom, without changing the designation...
. If an atom of lower average binding energy is changed into an atom of higher average binding energy, energy is given off. The chart shows that fusion of hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
, the combination to form heavier atoms, releases energy, as does fission of uranium, the breaking up of a larger nucleus into smaller parts. Stability varies between isotopes: the isotope U-235
Uranium-235
- References :* .* DOE Fundamentals handbook: Nuclear Physics and Reactor theory , .* A piece of U-235 the size of a grain of rice can produce energy equal to that contained in three tons of coal or fourteen barrels of oil. -External links:* * * one of the earliest articles on U-235 for the...
is much less stable than the more common U-238
Uranium-238
Uranium-238 is the most common isotope of uranium found in nature. It is not fissile, but is a fertile material: it can capture a slow neutron and after two beta decays become fissile plutonium-239...
.
Nuclear energy is released by three exoenergetic (or exothermic
Exothermic
In thermodynamics, the term exothermic describes a process or reaction that releases energy from the system, usually in the form of heat, but also in the form of light , electricity , or sound...
) processes:
- Radioactive decayRadioactive decayRadioactive decay is the process by which an atomic nucleus of an unstable atom loses energy by emitting ionizing particles . The emission is spontaneous, in that the atom decays without any physical interaction with another particle from outside the atom...
, where a neutron or proton in the radioactive nucleus decays spontaneously by emitting either particles, electromagnetic radiationElectromagnetic radiationElectromagnetic radiation is a form of energy that exhibits wave-like behavior as it travels through space...
(gamma rays), neutrinos (or all of them) - FusionNuclear fusionNuclear fusion is the process by which two or more atomic nuclei join together, or "fuse", to form a single heavier nucleus. This is usually accompanied by the release or absorption of large quantities of energy...
, two atomic nuclei fuse together to form a heavier nucleus - FissionNuclear fissionIn nuclear physics and nuclear chemistry, nuclear fission is a nuclear reaction in which the nucleus of an atom splits into smaller parts , often producing free neutrons and photons , and releasing a tremendous amount of energy...
, the breaking of a heavy nucleus into two (or more rarely three) lighter nuclei
Binding energy for atoms
The binding energy of an atom (including its electrons) is not the same as the binding energy of the atom's nucleus. The measured mass deficits of isotopes are always listed as mass deficits of the neutralNeutral particle
In physics, a neutral particle is a particle with no electric charge. This is not to be confused with a real neutral particle, a neutral particle that is also identical to its own antiparticle.-Stable or long-lived neutral particles:...
atom
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
s of that isotope, and mostly in MeV
MEV
MeV and meV are multiples and submultiples of the electron volt unit referring to 1,000,000 eV and 0.001 eV, respectively.Mev or MEV may refer to:In entertainment:* Musica Elettronica Viva, an Italian musical group...
. As a consequence, the listed mass deficits are not a measure for the stability or binding energy of isolated nuclei, but for the whole atoms. This has very practical reasons, because it is very hard to totally ion
Ion
An ion is an atom or molecule in which the total number of electrons is not equal to the total number of protons, giving it a net positive or negative electrical charge. The name was given by physicist Michael Faraday for the substances that allow a current to pass between electrodes in a...
ize heavy elements, i.e. strip them of all of their electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
s.
This practice is useful for other reasons, too: Stripping all the electrons from a heavy unstable nucleus (thus producing a bare nucleus) will change the lifetime of the nucleus, indicating that the nucleus cannot be treated independently (Experiments at the heavy ion accelerator GSI
Gesellschaft für Schwerionenforschung
The GSI Helmholtz Centre for Heavy Ion Research GmbH in the Wixhausen suburb of Darmstadt, Germany is a federally and state co-funded heavy ion research center. The current director of GSI is Horst Stöcker who succeeded Walter F...
). This is also evident from phenomena like electron capture
Electron capture
Electron capture is a process in which a proton-rich nuclide absorbs an inner atomic electron and simultaneously emits a neutrino...
. Theoretically, in orbital models of heavy atoms, the electron orbits partially inside the nucleus (it doesn't orbit
Orbit
In physics, an orbit is the gravitationally curved path of an object around a point in space, for example the orbit of a planet around the center of a star system, such as the Solar System...
in a strict sense, but has a non-vanishing probability of being located inside the nucleus).
A nuclear decay happens to the nucleus, meaning that properties ascribed to the nucleus will change in the event. In the field of physics the concept of "mass deficit" as a measure for "binding energy" means "mass deficit of the neutral atom" (not just the nucleus) and is a measure for stability of the whole atom.
Nuclear binding energy curve

Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
up to sodium
Sodium
Sodium is a chemical element with the symbol Na and atomic number 11. It is a soft, silvery-white, highly reactive metal and is a member of the alkali metals; its only stable isotope is 23Na. It is an abundant element that exists in numerous minerals, most commonly as sodium chloride...
is observed to exhibit generally increasing binding energy per nucleon as the atomic mass
Atomic mass
The atomic mass is the mass of a specific isotope, most often expressed in unified atomic mass units. The atomic mass is the total mass of protons, neutrons and electrons in a single atom....
increases. This increase is generated by increasing forces per nucleon in the nucleus, as each additional nucleon is attracted by other nearby nucleons, and thus more tightly bound to the whole.
The region of increasing binding energy is followed by a region of relative stability (saturation) in the sequence from magnesium
Magnesium
Magnesium is a chemical element with the symbol Mg, atomic number 12, and common oxidation number +2. It is an alkaline earth metal and the eighth most abundant element in the Earth's crust and ninth in the known universe as a whole...
through xenon
Xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. The element name is pronounced or . A colorless, heavy, odorless noble gas, xenon occurs in the Earth's atmosphere in trace amounts...
. In this region, the nucleus has become large enough that nuclear forces no longer completely extend efficiently across its width. Attractive nuclear forces in this region, as atomic mass increases, are nearly balanced by repellent electromagnetic forces between protons, as the atomic number
Atomic number
In chemistry and physics, the atomic number is the number of protons found in the nucleus of an atom and therefore identical to the charge number of the nucleus. It is conventionally represented by the symbol Z. The atomic number uniquely identifies a chemical element...
increases.
Finally, in elements heavier than xenon, there is a decrease in binding energy per nucleon as atomic number increases. In this region of nuclear size, electromagnetic repulsive forces are beginning to overcome the strong nuclear force attraction.
At the peak of binding energy, nickel-62
Nickel-62
Nickel-62 is an isotope of nickel having 28 protons and 34 neutrons.It is a stable isotope, with the highest binding energy per nucleon of any known nuclide . It is often stated that 56Fe is the "most stable nucleus", but actually 56Fe has the lowest mass per nucleon of all nuclides...
is the most tightly bound nucleus (per nucleon), followed by iron-58 and iron-56
Iron-56
Iron-56 is the most common isotope of iron. About 91.754% of all iron is iron-56.Of all isotopes, iron-56 has the lowest mass per nucleon. With 8.8 MeV binding energy per nucleon, iron-56 is one of the most tightly bound nuclei....
. This is the approximate basic reason why iron and nickel are very common metals in planetary cores, since they are produced profusely as end products in supernova
Supernova
A supernova is a stellar explosion that is more energetic than a nova. It is pronounced with the plural supernovae or supernovas. Supernovae are extremely luminous and cause a burst of radiation that often briefly outshines an entire galaxy, before fading from view over several weeks or months...
e and in the final stages of silicon burning in stars. However, it is not binding energy per defined nucleon (as defined above) which controls which exact nuclei are made, because within stars, neutrons are free to convert to protons to release even more energy, per generic nucleon, if the result is a stable nucleus with a larger fraction of protons. Thus, iron-56 has the most binding energy of any group of 56 nucleons (because of its relatively larger fraction of protons), even while having less binding energy per nucleon than nickel-62, if this binding energy is computed by comparing Ni-62 with its disassembly products of 28 protons and 34 neutrons. In fact, it has been argued that photodisintegration
Photodisintegration
Photodisintegration is a physical process in which an extremely high energy gamma ray interacts with an atomic nucleus and causes it to enter an excited state, which immediately decays by emitting a subatomic particle. A single proton or neutron is effectively knocked out of the nucleus by the...
of 62Ni to form 56Fe may be energetically possible in an extremely hot star core, due to this beta decay conversion of neutrons to protons.
It is generally believed that iron-56 is more common than nickel isotopes in the universe for mechanistic reasons, because its unstable progenitor nickel-56 is copiously made by staged build-up of 14 helium nuclei inside supernovas, where it has no time to decay to iron before being released into the interstellar medium in a matter of a few minutes as a star explodes. However, nickel-56 then decays to iron-56
Iron-56
Iron-56 is the most common isotope of iron. About 91.754% of all iron is iron-56.Of all isotopes, iron-56 has the lowest mass per nucleon. With 8.8 MeV binding energy per nucleon, iron-56 is one of the most tightly bound nuclei....
within a few weeks. The gamma ray light curve of such a process has been observed to happen in type IIa supernovae, such as SN1987a. In a star, there are no good ways to create nickel-62 by alpha-addition processes, or else there would presumably be more of this highly stable nuclide in the universe.
Measuring the binding energy
The fact that the maximum binding energy is found in medium-sized nuclei is a consequence of the trade-off in the effects of two opposing forces which have different range characteristics. The attractive nuclear force (strong nuclear force), which binds protons and neutrons equally to each other, has a limited range due to a rapid exponential decrease in this force with distance. However, the repelling electromagnetic force, which acts between protons to force nuclei apart, falls off with distance much more slowly (as the inverse square of distance). For nuclei larger than about four nucleons in diameter, the additional repelling force of additional protons more than offsets any binding energy which results between further added nucleons as a result of additional strong force interactions; such nuclei become less and less tightly bound as their size increases, though most of them are still stable. Finally, nuclei containing more than 209 nucleons (larger than about 6 nucleons in diameter) are all too large to be stable, and are subject to spontaneous decay to smaller nuclei.Nuclear fusion
Nuclear fusion
Nuclear fusion is the process by which two or more atomic nuclei join together, or "fuse", to form a single heavier nucleus. This is usually accompanied by the release or absorption of large quantities of energy...
produces energy by combining the very lightest elements into more tightly bound elements (such as hydrogen into helium
Helium
Helium is the chemical element with atomic number 2 and an atomic weight of 4.002602, which is represented by the symbol He. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas that heads the noble gas group in the periodic table...
), and nuclear fission
Nuclear fission
In nuclear physics and nuclear chemistry, nuclear fission is a nuclear reaction in which the nucleus of an atom splits into smaller parts , often producing free neutrons and photons , and releasing a tremendous amount of energy...
produces energy by splitting the heaviest elements (such as uranium
Uranium
Uranium is a silvery-white metallic chemical element in the actinide series of the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons...
and plutonium
Plutonium
Plutonium is a transuranic radioactive chemical element with the chemical symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, forming a dull coating when oxidized. The element normally exhibits six allotropes and four oxidation...
) into more tightly bound elements (such as barium
Barium
Barium is a chemical element with the symbol Ba and atomic number 56. It is the fifth element in Group 2, a soft silvery metallic alkaline earth metal. Barium is never found in nature in its pure form due to its reactivity with air. Its oxide is historically known as baryta but it reacts with...
and krypton
Krypton
Krypton is a chemical element with the symbol Kr and atomic number 36. It is a member of Group 18 and Period 4 elements. A colorless, odorless, tasteless noble gas, krypton occurs in trace amounts in the atmosphere, is isolated by fractionally distilling liquified air, and is often used with other...
). Both processes produce energy, because middle-sized nuclei are the most tightly bound of all.
As seen above in the example of deuterium, nuclear binding energies are large enough that they may be easily measured as fractional mass
Mass
Mass can be defined as a quantitive measure of the resistance an object has to change in its velocity.In physics, mass commonly refers to any of the following three properties of matter, which have been shown experimentally to be equivalent:...
deficits, according to the equivalence of mass and energy. The atomic binding energy is simply the amount of energy (and mass) released, when a collection of free nucleons are joined together to form a nucleus
Atomic nucleus
The nucleus is the very dense region consisting of protons and neutrons at the center of an atom. It was discovered in 1911, as a result of Ernest Rutherford's interpretation of the famous 1909 Rutherford experiment performed by Hans Geiger and Ernest Marsden, under the direction of Rutherford. The...
.
Nuclear binding energy can be computed from the difference in mass of a nucleus, and the sum of the masses of the number of free neutrons and protons that make up the nucleus. Once this mass difference, called the mass defect or mass deficiency, is known, Einstein's mass-energy equivalence
Mass-energy equivalence
In physics, mass–energy equivalence is the concept that the mass of a body is a measure of its energy content. In this concept, mass is a property of all energy, and energy is a property of all mass, and the two properties are connected by a constant...
formula E = mc² can be used to compute the binding energy of any nucleus. Early nuclear physicists used to refer to computing this value as a "packing fraction" calculation.
For example, the atomic mass unit
Atomic mass unit
The unified atomic mass unit or dalton is a unit that is used for indicating mass on an atomic or molecular scale. It is defined as one twelfth of the rest mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state, and has a value of...
(1 u) is defined to be 1/12 of the mass of a 12C atom—but the atomic mass of a 1H atom (which is a proton plus electron) is 1.007825 u, so each nucleon in 12C has lost, on average, about 0.8% of its mass in the form of binding energy.
Semiempirical formula for nuclear binding energy
For a nucleus with A nucleons, including Z protons and N neutrons, a semiemipirical formula for the binding energy (B.E.) per nucleon (A) is:
where the binding energy is in MeV for the following numerical values of the constants:
 ;
;  ;
;  ;
;  ;
;  .
.The first term
 is called the saturation contribution and ensures that the B.E. per nucleon is the same for all nuclei to a first approximation. The term
is called the saturation contribution and ensures that the B.E. per nucleon is the same for all nuclei to a first approximation. The term  is a surface tension effect and is proportional to the number of nucleons that are situated on the nuclear surface; it is largest for light nuclei. The term
is a surface tension effect and is proportional to the number of nucleons that are situated on the nuclear surface; it is largest for light nuclei. The term  is the Coulomb electrostatic repulsion; this becomes more important as
is the Coulomb electrostatic repulsion; this becomes more important as  increases. The symmetry correction term
increases. The symmetry correction term  takes into account the fact that in the absence of other effects the most stable arrangement has equal numbers of protons and neutrons; this is because the n-p interaction in a nucleus is stronger than either the n-n or p-p interaction. The pairing term
takes into account the fact that in the absence of other effects the most stable arrangement has equal numbers of protons and neutrons; this is because the n-p interaction in a nucleus is stronger than either the n-n or p-p interaction. The pairing term  is purely empirical; it is + for even-even nuclei and - for odd-odd nuclei.
is purely empirical; it is + for even-even nuclei and - for odd-odd nuclei.Example values deduced from experimentally measured atom nuclide masses
The following table lists some binding energies and mass defect values. Notice also that we use 1 u = 1 a.m.uAtomic mass unit
The unified atomic mass unit or dalton is a unit that is used for indicating mass on an atomic or molecular scale. It is defined as one twelfth of the rest mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state, and has a value of...
= 931.494028(±0.000023) MeV. To calculate the "binding energy" we use the formula P*(mp+me) + N * mn - mnuclide where P denotes the number of protons of the nuclides and N its number of neutrons. We take
mp = 938.2723 Mev, me = 0.5110 MeV and mn = 939.5656 MeV. The letter A denotes the sum of P and N (number of nucleons in the nuclide). If we assume the reference nucleon has the mass of a neutron (so that all "total" binding energies calculated are maximal) we could define the total binding energy as the difference from the mass of the nucleus, and the mass of a collection of A free neutrons. In other words, it would be [(P+N)* mn] - mnuclide. The "total binding energy per nucleon" would be this value divided by A.
| nuclide | P | N | mass excess | total mass | total mass / A | total binding energy / A | mass defect | binding energy | binding energy / A |
|---|---|---|---|---|---|---|---|---|---|
| 56Fe Iron-56 Iron-56 is the most common isotope of iron. About 91.754% of all iron is iron-56.Of all isotopes, iron-56 has the lowest mass per nucleon. With 8.8 MeV binding energy per nucleon, iron-56 is one of the most tightly bound nuclei.... |
26 | 30 | -60.6054 MeV | 55.934937 u | 0.9988382 u | 9.1538 MeV | 0.528479 u | 492.275 MeV | 8.7906 MeV |
| 58Fe | 26 | 32 | -62.1534 MeV | 57.933276 u | 0.9988496 u | 9.1432 MeV | 0.547471 u | 509.966 MeV | 8.7925 MeV |
| 60Ni | 28 | 32 | -64.4721 MeV | 59.930786 u | 0.9988464 u | 9.1462 MeV | 0.565612 u | 526.864 MeV | 8.7811 MeV |
| 62Ni Nickel-62 Nickel-62 is an isotope of nickel having 28 protons and 34 neutrons.It is a stable isotope, with the highest binding energy per nucleon of any known nuclide . It is often stated that 56Fe is the "most stable nucleus", but actually 56Fe has the lowest mass per nucleon of all nuclides... |
28 | 34 | -66.7461 MeV | 61.928345 u | 0.9988443 u | 9.1481 MeV | 0.585383 u | 545.281 MeV | 8.7948 MeV |
56Fe has the lowest nucleon-specific mass of the four nuclides listed in this table, but this does not imply it is the strongest bound atom per hadron, unless the choice of beginning hadrons is completely free. Iron releases the largest energy if any 56 nucleons are allowed to build a nuclide—changing one to another if necessary, The highest binding energy per hadron, with the hadrons starting as the same number of protons Z and total nucleons A as in the bound nucleus, is 62Ni. Thus, the true absolute value of the total binding energy of a nucleus depends on what we are allowed to construct the nucleus out of. If all nuclei of mass number A were to be allowed to be constructed of A neutrons, then Fe-56 would release the most energy per nucleon, since it has a larger fraction of protons than Ni-62. However, if nucleons are required to be constructed of only the same number of protons and neutrons that they contain, then nickel-62 is the most tightly bound nucleus, per nucleon.
| nuclide | P | N | mass excess | total mass | total mass / A | total binding energy / A | mass defect | binding energy | binding energy / A |
|---|---|---|---|---|---|---|---|---|---|
| n Neutron The neutron is a subatomic hadron particle which has the symbol or , no net electric charge and a mass slightly larger than that of a proton. With the exception of hydrogen, nuclei of atoms consist of protons and neutrons, which are therefore collectively referred to as nucleons. The number of... |
0 | 1 | 8.0716 MeV | 1.008665 u | 1.008665 u | 0.0000 MeV | 0 u | 0 MeV | 0 MeV |
| 1H | 1 | 0 | 7.2890 MeV | 1.007825 u | 1.007825 u | 0.7826 MeV | 0.0000000146 u | 0.0000136 MeV | 13.6 eV |
| 2H Deuterium Deuterium, also called heavy hydrogen, is one of two stable isotopes of hydrogen. It has a natural abundance in Earth's oceans of about one atom in of hydrogen . Deuterium accounts for approximately 0.0156% of all naturally occurring hydrogen in Earth's oceans, while the most common isotope ... |
1 | 1 | 13.13572 MeV | 2.014102 u | 1.007051 u | 1.50346 MeV | 0.002388 u | 2.22452 MeV | 1.11226 MeV |
| 3H Tritium Tritium is a radioactive isotope of hydrogen. The nucleus of tritium contains one proton and two neutrons, whereas the nucleus of protium contains one proton and no neutrons... |
1 | 2 | 14.9498 MeV | 3.016049 u | 1.005350 u | 3.08815 MeV | 0.0091058 u | 8.4820 MeV | 2.8273 MeV |
| 3He Helium-3 Helium-3 is a light, non-radioactive isotope of helium with two protons and one neutron. It is rare on Earth, and is sought for use in nuclear fusion research... |
2 | 1 | 14.9312 MeV | 3.016029 u | 1.005343 u | 3.09433 MeV | 0.0082857 u | 7.7181 MeV | 2.5727 MeV |
In the table above it can be seen that the decay of a neutron, as well as the transformation of tritium into helium-3, releases energy; hence, it manifests a stronger bound new state when measured against the mass of an equal number of neutrons (and also a lighter state per number of total hadrons). Such reactions are not driven by changes in binding energies as calculated from previously fixed N and Z numbers of neutrons and protons, but rather in decreases in the total mass of the nuclide/per nucleon, with the reaction.

