
Protein domain
Encyclopedia
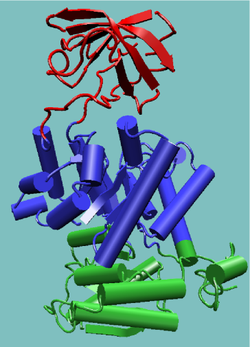
Tertiary structure
In biochemistry and molecular biology, the tertiary structure of a protein or any other macromolecule is its three-dimensional structure, as defined by the atomic coordinates.-Relationship to primary structure:...
that can evolve, function, and exist independently of the rest of the protein chain. Each domain forms a compact three-dimensional structure and often can be independently stable and folded
Protein folding
Protein folding is the process by which a protein structure assumes its functional shape or conformation. It is the physical process by which a polypeptide folds into its characteristic and functional three-dimensional structure from random coil....
. Many proteins consist of several structural domains. One domain may appear in a variety of different proteins. Molecular evolution
Molecular evolution
Molecular evolution is in part a process of evolution at the scale of DNA, RNA, and proteins. Molecular evolution emerged as a scientific field in the 1960s as researchers from molecular biology, evolutionary biology and population genetics sought to understand recent discoveries on the structure...
uses domains as building blocks and these may be recombined in different arrangements to create protein
Protein
Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. A polypeptide is a single linear polymer chain of amino acids bonded together by peptide bonds between the carboxyl and amino groups of...
s with different functions. Domains vary in length from between about 25 amino acids up to 500 amino acids in length. The shortest domains such as zinc fingers are stabilized by metal ions or disulfide bridges. Domains often form functional units, such as the calcium-binding EF hand domain of calmodulin
Calmodulin
Calmodulin is a calcium-binding protein expressed in all eukaryotic cells...
. Because they are independently stable, domains can be "swapped" by genetic engineering
Genetic engineering
Genetic engineering, also called genetic modification, is the direct human manipulation of an organism's genome using modern DNA technology. It involves the introduction of foreign DNA or synthetic genes into the organism of interest...
between one protein and another to make chimeric proteins.
Background
The concept of the domain was first proposed in 1973 by Wetlaufer after X-raycrystallographic studies of hen lysozyme
Lysozyme
Lysozyme, also known as muramidase or N-acetylmuramide glycanhydrolase, are glycoside hydrolases, enzymes that damage bacterial cell walls by catalyzing hydrolysis of 1,4-beta-linkages between N-acetylmuramic acid and N-acetyl-D-glucosamine residues in a peptidoglycan and between...
and papain
Papain
Papain, also known as papaya proteinase I, is a cysteine protease enzyme present in papaya and mountain papaya .-Papain family:...
and by limited proteolysis studies of immunoglobulins. Wetlaufer defined domains as stable units of protein structure
Protein structure
Proteins are an important class of biological macromolecules present in all organisms. Proteins are polymers of amino acids. Classified by their physical size, proteins are nanoparticles . Each protein polymer – also known as a polypeptide – consists of a sequence formed from 20 possible L-α-amino...
that could fold autonomously. In the past domains have been described as units of:
- compact structure
- function and evolution
- folding.
Each definition is valid and will often overlap, i.e. a compact structural domain that is found amongst diverse proteins is likely to fold independently within its structural environment. Nature often brings several domains together to form multidomain and multifunctional proteins with a vast number of possibilities. In a multidomain protein, each domain may fulfill its own function independently, or in a concerted manner with its neighbours. Domains can either serve as modules for building up large assemblies such as virus particles or muscle fibres, or can provide specific catalytic or binding sites as found in enzymes or regulatory proteins.
An appropriate example is pyruvate kinase
Pyruvate kinase
Pyruvate kinase is an enzyme involved in glycolysis. It catalyzes the transfer of a phosphate group from phosphoenolpyruvate to ADP, yielding one molecule of pyruvate and one molecule of ATP.-Reaction:The reaction with pyruvate kinase:...
, a glycolytic enzyme that plays an important role in regulating the flux from fructose-1,6-biphosphate to pyruvate. It contains an all-β regulatory domain, an α/β-substrate binding domain and an α/β-nucleotide binding domain, connected by several polypeptide linkers (see figure, right). Each domain in this protein occurs in diverse sets of protein families.
The central α/β-barrel substrate binding domain is one of the most common enzyme
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
folds. It is seen in many different enzyme families catalysing completely unrelated reactions. The α/β-barrel is commonly called the TIM barrel
TIM barrel
The TIM barrel is a conserved protein fold consisting of eight α-helices and eight parallel β-strands that alternate along the peptide backbone. The structure is named after triosephosphate isomerase, a conserved glycolytic enzyme. TIM barrels are quite common among the conserved protein folds...
named after triose phosphate isomerase, which was the first such structure to be solved. It is currently classified into 26 homologous families in the CATH domain database. The TIM barrel is formed from a sequence of β-α-β motifs closed by the first and last strand hydrogen bonding together, forming an eight stranded barrel. There is debate about the evolutionary origin of this domain. One study has suggested
that a single ancestral enzyme could have diverged into several families, while another suggests that a stable TIM-barrel structure has evolved
through convergent evolution.
The TIM-barrel in pyruvate kinase is 'discontinuous', meaning that more than one segment of the polypeptide is required to form the domain. This is likely to be the result of the insertion of one domain into another during the protein's evolution. It has been shown from known structures that about a quarter of structural domains are discontinuous. The inserted β-barrel regulatory domain is 'continuous', made up of a single stretch of polypeptide.
Covalent association of two domains represents a functional and structural advantage since there is an increase in stability when compared with the same structures non-covalently associated. Other, advantages are the protection of intermediates within inter-domain enzymatic clefts that may
otherwise be unstable in aqueous environments, and a fixed stoichiometric ratio of the enzymatic activity necessary for a sequential set of reactions.
Domains are units of protein structure
The primary structure (string of amino acids) of a proteinProtein
Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. A polypeptide is a single linear polymer chain of amino acids bonded together by peptide bonds between the carboxyl and amino groups of...
ultimately encodes its uniquely folded 3D conformation. The most important factor governing the folding of a protein into 3D structure is the distribution of polar and non-polar side chains. Folding is driven by the burial of hydrophobic side chains into the interior of the molecule so to avoid contact with the aqueous environment. Generally proteins have a core of hydrophobic residues
Residue (chemistry)
In chemistry, residue is the material remaining after a distillation or an evaporation, or to a portion of a larger molecule, such as a methyl group. It may also refer to the undesired byproducts of a reaction....
surrounded by a shell of hydrophilic residues. Since the peptide bonds themselves are polar they are neutralised by hydrogen bonding with each other when in the hydrophobic environment. This gives rise to regions of the polypeptide that form regular 3D structural patterns called secondary structure. There are two main types of secondary structure: α-helices
Alpha helix
A common motif in the secondary structure of proteins, the alpha helix is a right-handed coiled or spiral conformation, in which every backbone N-H group donates a hydrogen bond to the backbone C=O group of the amino acid four residues earlier...
and β-sheet
Beta sheet
The β sheet is the second form of regular secondary structure in proteins, only somewhat less common than the alpha helix. Beta sheets consist of beta strands connected laterally by at least two or three backbone hydrogen bonds, forming a generally twisted, pleated sheet...
s.
Some simple combinations of secondary structure elements have been found to frequently occur in protein structure
Protein structure
Proteins are an important class of biological macromolecules present in all organisms. Proteins are polymers of amino acids. Classified by their physical size, proteins are nanoparticles . Each protein polymer – also known as a polypeptide – consists of a sequence formed from 20 possible L-α-amino...
and are referred to as supersecondary structure
Supersecondary structure
A supersecondary structure is a compact three-dimensional protein structure of several adjacent elements of secondary structure that is smaller than a protein domain or a subunit. Supersecondary structures can act as nucleations in the process of protein folding. Examples include β-hairpins,...
or motifs
Structural motif
In a chain-like biological molecule, such as a protein or nucleic acid, a structural motif is a supersecondary structure, which appears also in a variety of other molecules...
. For example, the β-hairpin motif consists of two adjacent antiparallel β-strands joined by a small loop. It is present in most antiparallel β structures both as an isolated ribbon and as part of more complex β-sheets. Another common super-secondary structure is the β-α-β motif, which is frequently used to connect two parallel β-strands. The central α-helix connects the C-termini of the first strand to the N-termini of the second strand, packing its side chains against the β-sheet and therefore shielding the hydrophobic residues of the β-strands from the surface.
Structural alignment
Structural alignment
Structural alignment attempts to establish homology between two or more polymer structures based on their shape and three-dimensional conformation. This process is usually applied to protein tertiary structures but can also be used for large RNA molecules...
is an important tool for determining domains.
Tertiary structure of domains
Several motifs pack together to form compact, local, semi-independent units called domains.The overall 3D structure of the polypeptide chain is referred to as the protein's tertiary structure. Domains are the fundamental units of tertiary structure, each domain containing an individual hydrophobic core built from secondary structural units connected by loop regions. The packing of the polypeptide is usually much tighter in the interior than the exterior of the domain producing a solid-like core and a fluid-like surface. In fact, core residues are often conserved in a protein family, whereas the residues in loops are less conserved, unless they are involved in the protein's function. Protein tertiary structure can be divided into four main classes based on the secondary structural content of the domain.
- All-α domains have a domain core built exclusively from α-helices. This class is dominated by small folds, many of which form a simple bundle with helices running up and down.
- All-β domains have a core composed of antiparallel β-sheets, usually two sheets packed against each other. Various patterns can be identified in the arrangement of the strands, often giving rise to the identification of recurring motifs, for example the Greek key motif.
- α+β domains are a mixture of all-α and all-β motifs. Classification of proteins into this class is difficult because of overlaps to the other three classes and therefore is not used in the CATHCATHThe CATH Protein Structure Classification is a semi-automatic, hierarchical classification of protein domains published in 1997 by Christine Orengo, Janet Thornton and their colleagues....
domain database. - α/β domains are made from a combination of β-α-β motifs that predominantly form a parallel β-sheet surrounded by amphipathic α-helices. The secondary structures are arranged in layers or barrels.
Domains have limits on size
Domains have limits on size. The size of individual structural domains varies from 36 residues in E-selectin to 692 residues in lipoxygenase-1, but the majority, 90%, have less than 200 residues with an average of approximately 100 residues. Very short domains, less than 40 residues, are often stabilised by metal ions or disulfide bonds. Larger domains, greater than 300 residues, are likely to consist of multiple hydrophobic cores.Domains and quaternary structure
Many proteins have a quaternary structure, which consists of several polypeptide chains that associate into an oligomeric molecule. Each polypeptide chain in such a protein is called a subunit. Hemoglobin, for example, consists of two α and two β subunits. Each of the four chains has an all-α globin fold with a heme pocket.Domain swapping is a mechanism for forming oligomeric assemblies. In domain swapping, a secondary or tertiary element of a monomeric protein is replaced by the same element of another protein. Domain swapping can range from secondary structure elements to whole structural domains. It also represents a model of evolution for functional adaptation by oligomerisation, e.g. oligomeric enzymes that have their active site at subunit interfaces.
Domains as evolutionary modules
Nature is a tinkerer and not an inventor, new sequences are adapted from pre-existing sequences rather than invented. Domains are the common material used by nature to generate new sequences, they can be thought of as genetically mobile units, referred to as 'modules'. Often, the C and N termini of domains are close together in space, allowing them to easily be "slotted into" parent structures during the process of evolution. Many domain families are found in all three forms of life, ArchaeaArchaea
The Archaea are a group of single-celled microorganisms. A single individual or species from this domain is called an archaeon...
, Bacteria
Bacteria
Bacteria are a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria have a wide range of shapes, ranging from spheres to rods and spirals...
and Eukarya. Domains that are repeatedly found in diverse proteins are often referred to as modules, examples can be found among extracellular proteins associated with clotting, fibrinolysis, complement, the extracellular matrix, cell surface adhesion molecules and cytokine receptors.
Molecular evolution
Molecular evolution
Molecular evolution is in part a process of evolution at the scale of DNA, RNA, and proteins. Molecular evolution emerged as a scientific field in the 1960s as researchers from molecular biology, evolutionary biology and population genetics sought to understand recent discoveries on the structure...
gives rise to families of related proteins with similar sequence and structure. However, sequence similarities can be extremely low between proteins that share the same structure. Protein structures may be similar because proteins have diverged from a common ancestor. Alternatively, some folds may be more favored than others as they represent stable arrangements of secondary structures and some proteins may converge towards these folds over the course of evolution . There are currently about 45,000 experimentally determined protein 3D structures deposited within the Protein Data Bank
Protein Data Bank
The Protein Data Bank is a repository for the 3-D structural data of large biological molecules, such as proteins and nucleic acids....
(PDB). However this set contains a lot of identical or very similar structures. All proteins should be classified to structural families to understand their evolutionary relationships. Structural comparisons are best achieved at the domain level. For this reason many algorithms have been developed to automatically assign domains in proteins with known 3D structure, see 'Domain definition from structural co-ordinates'.
The CATH domain database classifies domains into approximately 800 fold families, ten of these folds are highly populated and are referred to as 'super-folds'. Super-folds are defined as folds for which there are at least three structures without significant sequence similarity. The most populated is the α/β-barrel super-fold as described previously.
Multidomain proteins
The majority of genomic proteins, two-thirds in unicellular organisms and more than 80% in metazoa, are multidomain proteins created as a result of geneduplication events. Many domains in multidomain structures could have once existed as independent proteins. More and more domains in eukaryotic multidomain proteins can be found as independent proteins in prokaryotes. For example, vertebrates have a multi-enzyme polypeptide containing the GAR synthetase
Phosphoribosylamine-glycine ligase
In enzymology, a phosphoribosylamine-glycine ligase is an enzyme that catalyzes the chemical reactionThe 3 substrates of this enzyme are ATP, 5-phospho-D-ribosylamine, and glycine, whereas its 3 products are ADP, phosphate, and N1-glycinamide.This enzyme belongs to the family of ligases,...
, AIR synthetase and GAR transformylase modules (GARs-AIRs-GARt; GAR: glycinamide ribonucleotide synthetase/transferase; AIR: aminoimidazole ribonucleotide synthetase). In insects, the polypeptide appears as GARs-(AIRs)2-GARt, in yeast GARs-AIRs is encoded separately from GARt, and in bacteria each domain is encoded separately.
Origin
Multidomain proteins are likely to have emerged from a selective pressure during evolutionEvolution
Evolution is any change across successive generations in the heritable characteristics of biological populations. Evolutionary processes give rise to diversity at every level of biological organisation, including species, individual organisms and molecules such as DNA and proteins.Life on Earth...
to create new functions. Various proteins have diverged from common ancestors by different combinations and associations of domains. Modular units frequently move about, within and between biological systems through mechanisms of genetic shuffling:
- transposition of mobile elements including horizontal transfers (between species);
- gross rearrangements such as inversions, translocations, deletions and duplications;
- homologous recombinationHomologous recombinationHomologous recombination is a type of genetic recombination in which nucleotide sequences are exchanged between two similar or identical molecules of DNA. It is most widely used by cells to accurately repair harmful breaks that occur on both strands of DNA, known as double-strand breaks...
; - slippage of DNA polymeraseDNA polymeraseA DNA polymerase is an enzyme that helps catalyze in the polymerization of deoxyribonucleotides into a DNA strand. DNA polymerases are best known for their feedback role in DNA replication, in which the polymerase "reads" an intact DNA strand as a template and uses it to synthesize the new strand....
during replication.
Types of organisation
The simplest multidomain organisation seen in proteins is that of a single domain repeated in tandem. The domains may interact with each other or remain isolated, like beads on string. The giant 30,000 residue muscle protein titinTitin
Titin , also known as connectin, is a protein that in humans is encoded by the TTN gene. Titin is a giant protein that functions as a molecular spring which is responsible for the passive elasticity of muscle. It is composed of 244 individually folded protein domains connected by unstructured...
comprises about 120 fibronectin-III-type and Ig-type domains. In the serine proteases, a gene duplication event has led to the formation of a two β-barrel domain enzyme. The repeats have diverged so widely that there is no obvious sequence similarity between them. The active site is located at a cleft between the two β-barrel domains, in which functionally important residues are contributed from each domain. Genetically engineered mutants of the chymotrypsin
Chymotrypsin
Chymotrypsin is a digestive enzyme that can perform proteolysis. Chymotrypsin preferentially cleaves peptide amide bonds where the carboxyl side of the amide bond is a tyrosine, tryptophan, or phenylalanine. These amino acids contain an aromatic ring in their sidechain that fits into a...
serine protease
Serine protease
Serine proteases are enzymes that cleave peptide bonds in proteins, in which serine serves as the nucleophilic amino acid at the active site.They are found ubiquitously in both eukaryotes and prokaryotes...
were shown to have some proteinase activity even though their active site residues were abolished and it has therefore been postulated that the duplication event enhanced the enzyme's activity.
Modules frequently display different connectivity relationships, as illustrated by the kinesin
Kinesin
A kinesin is a protein belonging to a class of motor proteins found in eukaryotic cells. Kinesins move along microtubule filaments, and are powered by the hydrolysis of ATP . The active movement of kinesins supports several cellular functions including mitosis, meiosis and transport of cellular...
s and ABC transporters. The kinesin motor domain can be at either end of a polypeptide chain that includes a coiled-coil region and a cargo domain. ABC transporters are built with up to four domains consisting of two unrelated modules, ATP-binding cassette and an integral membrane module, arranged in various combinations.
Not only do domains recombine, but there are many examples of a domain having been inserted into another. Sequence or structural similarities to other
domains demonstrate that homologues of inserted and parent domains can exist independently. An example is that of the 'fingers' inserted into the 'palm' domain within the polymerases of the Pol I family. Since a domain can be inserted into another, there should always be at least one continuous domain in a multidomain protein. This is the main difference between definitions of structural domains and evolutionary/functional domains. An evolutionary domain will be limited to one or two connections between domains, whereas structural domains can have unlimited connections, within a given criterion of the existence of a common core. Several structural domains could be assigned to an evolutionary domain.
Folding
Protein folding - the unsolved problem : Since the seminal work of Anfinsen over forty years ago, the goal to completely understand the mechanism by which a polypeptide rapidly folds into its stable native conformation remains elusive. Many experimental folding studies have contributed much to our understanding, but the principles that govern protein folding are still based on those discovered in the very first studies of folding. Anfinsen showed that the native state of a protein is thermodynamically stable, the conformation being at a global minimum of its free energy.Folding is a directed search of conformational space allowing the protein to fold on a biologically feasible time scale. The Levinthal paradox
Levinthal paradox
Levinthal's paradox is a thought experiment, also constituting a self-reference in the theory of protein folding. In 1969, Cyrus Levinthal noted that, because of the very large number of degrees of freedom in an unfolded polypeptide chain, the molecule has an astronomical number of possible...
states that if an averaged sized protein would sample all possible conformations before finding the one with the lowest energy, the whole process would take billions of years. Proteins typically fold within 0.1 and 1000 seconds, therefore the protein folding process must be directed some way through a specific folding pathway. The forces
that direct this search are likely to be a combination of local and global influences whose effects are felt at various stages of the reaction.
Advances in experimental and theoretical studies have shown that folding can be viewed in terms of energy landscapes, where folding kinetics is considered as a progressive organisation of an ensemble of partially folded structures through which a protein passes on its way to the folded structure. This has been described in terms of a folding funnel
Folding funnel
The folding funnel hypothesis is a specific version of the energy landscape theory of protein folding, which assumes that a protein's native state corresponds to its free energy minimum under the solution conditions usually encountered in cells...
, in which an unfolded protein has a large number of conformational states available and there are fewer states available to the folded protein. A funnel implies that for protein folding there is a decrease in energy and loss of entropy with increasing tertiary structure formation. The local roughness of the funnel reflects kinetic traps, corresponding to the accumulation of misfolded intermediates. A folding chain progresses toward lower intra-chain free-energies by increasing its compactness. The chains conformational options become increasingly narrowed ultimately toward one native structure.
Advantage of domains in protein folding
The organisation of large proteins by structural domains represents an advantage for protein folding, with each domain being able to individually fold, accelerating the folding process and reducing a potentially large combination of residue interactions. Furthermore, given the observed random distribution of hydrophobic residues in proteins, domain formation appears to be the optimal solution for a large protein to bury its hydrophobic residues while keeping the hydrophilic residues at the surface.However, the role of inter-domain interactions in protein folding and in energetics of stabilisation of the native structure, probably differs for each protein. In T4 lysozyme, the influence of one domain on the other is so strong that the entire molecule is resistant to proteolytic cleavage. In this case, folding is a sequential process where the C-terminal domain is required to fold independently in an early step, and the other domain requires the presence of the folded C-terminal domain for folding and stabilisation.
It has been found that the folding of an isolated domain can take place at the same rate or sometimes faster than that of the integrated domain. Suggesting that unfavourable interactions with the rest of the protein can occur during folding. Several arguments suggest that the slowest step in the folding of large proteins is the pairing of the folded domains. This is either because the domains are not folded entirely correctly or because the small adjustments required for their interaction are energetically unfavourable, such as the removal of water from the domain interface.
Domains and protein flexibility
The presence of multiple domains in proteins gives rise to a great deal of flexibility and mobilityConformational change
A macromolecule is usually flexible and dynamic. It can change its shape in response to changes in its environment or other factors; each possible shape is called a conformation, and a transition between them is called a conformational change...
, leading to protein domain dynamics. Domain motions can be inferred
by comparing different structures of a protein (as in Database of Molecular Motions
Database of Molecular Motions
The Database of Macromolecular Motions is a bioinformatics database that attempts tocategorize macromolecular motions, sometimes also known as conformational change.It was original developed by Mark B...
), or they can be directly
observed using spectra measured by neutron spin echo
Neutron spin echo
Neutron spin echo spectroscopy is an inelastic neutron scattering technique invented by Ferenc Mezei in the 1970's, and developed in collaboration with John Hayter.In recognition of his work and in other areas, Mezei was awarded the first in 1999....
spectroscopy.
They can also be suggested by sampling in extensive
molecular dynamics trajectories. Domain motions are important for:
- catalysis
- regulatory activity
- transport of metabolites
- formation of protein assemblies
- cellular locomotion
One of the largest observed domain motions is the `swivelling'
mechanism in pyruvate phosphate dikinase. The phosphoinositide domain swivels between two states in order to bring a phosphate group from the active site of the nucleotide binding domain to that of the phosphoenolpyruvate/pyruvate domain. The phosphate group is moved over a distance of 45A involving a domain motion of about 100 degrees around a single residue.In enzymes, the closure of one domain onto another captures a substrate by an induced fit, allowing the reaction to take place in a controlled way. A detailed analysis by Gerstein led to the classification of two basic types of domain motion; hinge and shear. Only a relatively small portion of the chain, namely the inter-domain linker and side chains undergo significant conformational changes upon domain rearrangement.
Hinges by secondary structures
A study by Hayward found that the termini of α-helices and β-sheets form hinges in a large number of cases. Many hinges were found to involve two secondary structure elements acting like hinges of a door, allowing an opening and closing motion to occur. This can arise when two neighbouring strands within a β-sheet situated in one domain, diverge apart as they join the other domain. The two resulting termini then form the bending regions between the two domains. α-helices that preserve their hydrogen bonding network when bent are found to behave as mechanical hinges, storing `elastic energy' that drives the closure of domains for rapid capture of a substrate.Helical to extended conformation
The interconversion of helical and extended conformations at the site of a domain boundary is not uncommon. In calmodulin, torsion angles change for five residues in the middle of a domain linking α-helix. The helix is split into two, almost perpendicular, smaller helices separated by four residues of an extended strand.Shear motions
Shear motions involve a small sliding movement of domain interfaces, controlled by the amino acid side chains within the interface. Proteins displaying shear motions often have a layered architecture: stacking of secondary structures. The interdomain linker has merely the role of keeping the domains in close proximity.Domain motion and functional dynamics in enzymes
The analysis of the internal dynamics of structurally different, but functionally similar enzymeshas highlighted a common relationship between the positioning of the
active site and the two principal protein sub-domains. In fact, for several members of the hydrolase superfamily, the catalytic site is located close to the interface separating the two principal quasi-rigid domains. Such positioning appears instrumental for maintaining the precise geometry of the active site, while allowing for an appreciable functionally-oriented modulation of the flanking regions resulting from the relative motion of the two sub-domains.
Domain definition from structural co-ordinates
The importance of domains as structural building blocks and elements of evolution has brought about many automated methods for their identification and classification in proteins of known structure. Automatic procedures for reliable domain assignment is essential for the generation of the domain databases, especially as the number of protein structures is increasing. Although the boundaries of a domain can be determined by visual inspection, construction of an automated method is not straightforward. Problems occur when faced with domains that are discontinuous or highly associated. The fact that there is no standard definition of what a domain really is has meant that domain assignments have varied enormously, with each researcher using a unique set of criteria.A structural domain is a compact, globular sub-structure with more interactions within it than with the rest of the protein.
Therefore, a structural domain can be determined by two visual characteristics; its compactness and its extent of isolation. Measures of local compactness in proteins have been used in many of the early methods of domain assignment and in several of the more recent methods.
Methods
One of the first algorithms used a Cα-Cα distance map together with a hierarchical clustering routine that considered proteins as several small segments, 10 residues in length. The initial segments were clustered one after another based on inter-segment distances; segments with the shortest distances were clustered and considered as single segments thereafter. The stepwise clustering finally included the full protein. Go also exploited the fact that inter-domain distances are normally larger than intra-domain distances; all possible Cα-Cα distances were represented as diagonal plots in which there were distinct patterns for helices, extended strands and combinations of secondary structures.The method by Sowdhamini and Blundell clusters secondary structures in a protein based on their Cα-Cα distances and identifies domains from the pattern in
their dendrogram
Dendrogram
A dendrogram is a tree diagram frequently used to illustrate the arrangement of the clusters produced by hierarchical clustering...
s. As the procedure does not consider the protein as a continuous chain of amino acids there are no problems in treating discontinuous domains. Specific nodes in these dendrograms are identified as tertiary structural clusters of the protein, these include both super-secondary structures and domains. The DOMAK algorithm is used to create the 3Dee domain database. It calculates a 'split value' from the number of each type of contact when the protein is divided arbitrarily into two parts. This split value is
large when the two parts of the structure are distinct.
The method of Wodak and Janin was based on the calculated interface areas between two chain segments repeatedly cleaved at various residue positions. Interface areas were calculated by comparing surface areas of the cleaved segments with that of the native structure. Potential domain boundaries can be identified at a site where the interface area was at a minimum. Other methods have used measures of solvent accessibility to calculate compactness.
The PUU algorithm incorporates a harmonic model used to approximate inter-domain dynamics. The underlying physical concept is that many rigid interactions will occur within each domain and loose interactions will occur between domains. This algorithm is used to define domains in the FSSP
Families of structurally similar proteins
Families of Structurally Similar Proteins or FSSP is a database of structurally superimposed proteins generated using the "Distance-matrix ALIgnment" algorithm. The database is helpful for the comparison of protein structures.-External links:*...
domain database.
Swindells (1995) developed a method, DETECTIVE, for identification of domains in protein structures based on the idea that domains have a hydrophobic
interior. Deficiencies were found to occur when hydrophobic cores from different domains continue through the interface region.
RigidFinder is a novel method for identification of protein rigid blocks (domains and loops) from two different conformations. Rigid blocks are defined as blocks where all inter residue distances are conserved across conformations.
A general method to identify dynamical domains, that is protein
regions that behave approximately as rigid units in the course of
structural fluctuations, has been introduced by Potestio et al. and, among other applications was also used
to compare the consistency of the dynamics-based domain
subdivisions with standard structure-based ones. The method,
termed PiSQRD, is publicly available in the form of a webserver. The latter allows users to optimally subdivide single-chain
or multimeric proteins into quasi-rigid domains based on the collective modes of fluctuation of the system. By default the
latter are calculated through an elastic network model;
alternatively pre-calculated essential dynamical spaces can be
uploaded by the user.
Example domains
- Armadillo repeatsArmadillo repeatsArmadillo repeats are named after the β-catenin-like Armadillo protein of the fruit fly Drosophila. These repeats are about 40 amino acids long and proteins that contain them have many tandemly repeated copies. Each Armadillo repeat is composed of a pair of alpha helices that form a hairpin...
: named after the β-catenin-like Armadillo protein of the fruit fly DrosophilaDrosophila melanogasterDrosophila melanogaster is a species of Diptera, or the order of flies, in the family Drosophilidae. The species is known generally as the common fruit fly or vinegar fly. Starting from Charles W...
.
- Basic Leucine zipper domain (bZIP domainBZIP domainThe Basic Leucine Zipper Domain is found in many DNA binding eukaryotic proteins. One part of the domain contains a region that mediates sequence specific DNA binding properties and the leucine zipper that is required for the dimerization of two DNA binding regions. The DNA binding region...
) : is found in many DNA-binding eukaryoticEukaryoteA eukaryote is an organism whose cells contain complex structures enclosed within membranes. Eukaryotes may more formally be referred to as the taxon Eukarya or Eukaryota. The defining membrane-bound structure that sets eukaryotic cells apart from prokaryotic cells is the nucleus, or nuclear...
proteins. One part of the domain contains a region that mediates sequence-specific DNA-binding properties and the Leucine zipper that is required for the dimerProtein dimerIn biochemistry, a dimer is a macromolecular complex formed by two, usually non-covalently bound, macromolecules like proteins or nucleic acids...
ization of two DNA-binding regions. The DNA-binding region comprises a number of basic aminoacids such as arginineArginineArginine is an α-amino acid. The L-form is one of the 20 most common natural amino acids. At the level of molecular genetics, in the structure of the messenger ribonucleic acid mRNA, CGU, CGC, CGA, CGG, AGA, and AGG, are the triplets of nucleotide bases or codons that codify for arginine during...
and lysineLysineLysine is an α-amino acid with the chemical formula HO2CCH4NH2. It is an essential amino acid, which means that the human body cannot synthesize it. Its codons are AAA and AAG....
- Cadherin repeats : Cadherins function as Ca2+-dependent cell-cell adhesionCell adhesionCellular adhesion is the binding of a cell to a surface, extracellular matrix or another cell using cell adhesion molecules such as selectins, integrins, and cadherins. Correct cellular adhesion is essential in maintaining multicellular structure...
proteins. Cadherin domains are extracellular regions which mediate cell-to-cell homophilic binding between cadherins on the surface of adjacent cells.
- Death effector domainDeath effector domainThe death-effector domain is a protein interaction domain found to regulate a variety of cellular signalling pathways. The DED domain is found in inactive procaspases and proteins that regulate caspase activation in the apoptosis cascade such as FAS-associating death domain-containing protein...
(DED) : allows protein-protein binding by homotypic interactions (DED-DED). CaspaseCaspaseCaspases, or cysteine-aspartic proteases or cysteine-dependent aspartate-directed proteases are a family of cysteine proteases that play essential roles in apoptosis , necrosis, and inflammation....
proteaseProteaseA protease is any enzyme that conducts proteolysis, that is, begins protein catabolism by hydrolysis of the peptide bonds that link amino acids together in the polypeptide chain forming the protein....
s trigger apoptosisApoptosisApoptosis is the process of programmed cell death that may occur in multicellular organisms. Biochemical events lead to characteristic cell changes and death. These changes include blebbing, cell shrinkage, nuclear fragmentation, chromatin condensation, and chromosomal DNA fragmentation...
via proteolytic cascades. Pro-Caspase-8 and pro-caspase-9 bind to specific adaptor molecules via DED domains and this leads to autoactivation of caspases.
- EF handEF handThe EF hand is a helix-loop-helix structural domain found in a large family of calcium-binding proteins. The EF-hand motif contains a helix-loop-helix topology, much like the spread thumb and forefinger of the human hand, in which the Ca2+ ions are coordinated by ligands within the loop...
: a helix-turn-helixHelix-turn-helixIn proteins, the helix-turn-helix is a major structural motif capable of binding DNA. It is composed of two α helices joined by a short strand of amino acids and is found in many proteins that regulate gene expression...
structural motifStructural motifIn a chain-like biological molecule, such as a protein or nucleic acid, a structural motif is a supersecondary structure, which appears also in a variety of other molecules...
found in each structural domain of the signaling protein calmodulinCalmodulinCalmodulin is a calcium-binding protein expressed in all eukaryotic cells...
and in the muscle protein troponin-C.
- Immunoglobulin-like domains : are found in proteins of the immunoglobulin superfamilyImmunoglobulin superfamilyThe immunoglobulin superfamily is a large group of cell surface and soluble proteins that are involved in the recognition, binding, or adhesion processes of cells. Molecules are categorized as members of this superfamily based on shared structural features with immunoglobulins ; they all possess a...
(IgSF). They contain about 70-110 amino acidAmino acidAmino acids are molecules containing an amine group, a carboxylic acid group and a side-chain that varies between different amino acids. The key elements of an amino acid are carbon, hydrogen, oxygen, and nitrogen...
s and are classified into different categories (IgV, IgC1, IgC2 and IgI) according to their size and function. They possess a characteristic fold in which two beta sheetBeta sheetThe β sheet is the second form of regular secondary structure in proteins, only somewhat less common than the alpha helix. Beta sheets consist of beta strands connected laterally by at least two or three backbone hydrogen bonds, forming a generally twisted, pleated sheet...
s form a "sandwich" that is stabilized by interactions between conserved cysteineCysteineCysteine is an α-amino acid with the chemical formula HO2CCHCH2SH. It is a non-essential amino acid, which means that it is biosynthesized in humans. Its codons are UGU and UGC. The side chain on cysteine is thiol, which is polar and thus cysteine is usually classified as a hydrophilic amino acid...
s and other charged amino acidAmino acidAmino acids are molecules containing an amine group, a carboxylic acid group and a side-chain that varies between different amino acids. The key elements of an amino acid are carbon, hydrogen, oxygen, and nitrogen...
s. They are important for protein-to-protein interactions in processes of cell adhesionCell adhesionCellular adhesion is the binding of a cell to a surface, extracellular matrix or another cell using cell adhesion molecules such as selectins, integrins, and cadherins. Correct cellular adhesion is essential in maintaining multicellular structure...
, cell activation, and molecular recognition. These domains are commonly found in molecules with roles in the immune systemImmune systemAn immune system is a system of biological structures and processes within an organism that protects against disease by identifying and killing pathogens and tumor cells. It detects a wide variety of agents, from viruses to parasitic worms, and needs to distinguish them from the organism's own...
.
- Phosphotyrosine-binding domainPhosphotyrosine-binding domainPhosphotyrosine-binding domain in the protein tensin tends to be found at the C-terminus. Tensin is a multi-domain protein that binds to actin filaments and functions as a focal-adhesion molecule...
(PTB) : PTB domains usually bind to phosphorylated tyrosine residues. They are often found in signal transduction proteins. PTB-domain binding specificity is determined by residues to the amino-terminal side of the phosphotyrosine. Examples: the PTB domains of both SHC and IRS-1 bind to a NPXpY sequence. PTB-containing proteins such as SHC and IRS-1 are important for insulinInsulinInsulin is a hormone central to regulating carbohydrate and fat metabolism in the body. Insulin causes cells in the liver, muscle, and fat tissue to take up glucose from the blood, storing it as glycogen in the liver and muscle....
responses of human cells.
- Pleckstrin homology domainPleckstrin homology domainPleckstrin homology domain is a protein domain of approximately 120 amino acids that occurs in a wide range of proteins involved in intracellular signaling or as constituents of the cytoskeleton....
(PH) : PH domains bind phosphoinositides with high affinity. Specificity for PtdIns(3)P, PtdIns(4)P, PtdIns(3,4)P2, PtdIns(4,5)P2, and PtdIns(3,4,5)P3 have all been observed. Given the fact that phosphoinositides are sequestered to various cell membranes (due to their long lipophilic tail) the PH domains usually causes recruitment of the protein in question to a membrane where the protein can exert a certain function in cell signalling, cytoskeletal reorganization or membrane trafficking.
- Src homology 2 domain (SH2) : SH2 domains are often found in signal transduction proteins. SH2 domains confer binding to phosphorylated tyrosine (pTyr). Named after the phosphotyrosine binding domain of the src viral oncogeneOncogeneAn oncogene is a gene that has the potential to cause cancer. In tumor cells, they are often mutated or expressed at high levels.An oncogene is a gene found in the chromosomes of tumor cells whose activation is associated with the initial and continuing conversion of normal cells into cancer...
, which is itself a tyrosine kinaseTyrosine kinaseA tyrosine kinase is an enzyme that can transfer a phosphate group from ATP to a protein in a cell. It functions as an "on" or "off" switch in many cellular functions....
. See also: SH3 domainSH3 domainThe SRC Homology 3 Domain is a small protein domain of about 60 amino acids residues first identified as a conserved sequence in the viral adaptor protein v-Crk and the non-catalytic parts of enzymes such as phospholipase and several cytoplasmic tyrosine kinases such as Abl and Src...
.
- Zinc finger DNA binding domainZinc fingerZinc fingers are small protein structural motifs that can coordinate one or more zinc ions to help stabilize their folds. They can be classified into several different structural families and typically function as interaction modules that bind DNA, RNA, proteins, or small molecules...
(ZnF_GATA) : ZnF_GATA domain-containing proteins are typically transcription factorTranscription factorIn molecular biology and genetics, a transcription factor is a protein that binds to specific DNA sequences, thereby controlling the flow of genetic information from DNA to mRNA...
s that usually bind to the DNA sequence [AT]GATA[AG] of promoters.
See also
- Binding domainBinding domainA binding domain is a protein domain which binds to a specific atom or molecule, such as calcium or DNA. Upon binding, proteins may undergo a conformational change. Binding domains are essential for the function of many proteins....
- Short linear motifShort linear motifIn molecular biology Short Linear Motifs are short stretches of protein sequence that mediate protein protein interaction.The first definition was given by Tim Hunt:...
- ProteinProteinProteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. A polypeptide is a single linear polymer chain of amino acids bonded together by peptide bonds between the carboxyl and amino groups of...
- Protein structureProtein structureProteins are an important class of biological macromolecules present in all organisms. Proteins are polymers of amino acids. Classified by their physical size, proteins are nanoparticles . Each protein polymer – also known as a polypeptide – consists of a sequence formed from 20 possible L-α-amino...
- Protein structure predictionProtein structure predictionProtein structure prediction is the prediction of the three-dimensional structure of a protein from its amino acid sequence — that is, the prediction of its secondary, tertiary, and quaternary structure from its primary structure. Structure prediction is fundamentally different from the inverse...
- Protein structure prediction software
- Protein familyProtein familyA protein family is a group of evolutionarily-related proteins, and is often nearly synonymous with gene family. The term protein family should not be confused with family as it is used in taxonomy....
- Structural biologyStructural biologyStructural biology is a branch of molecular biology, biochemistry, and biophysics concerned with the molecular structure of biological macromolecules, especially proteins and nucleic acids, how they acquire the structures they have, and how alterations in their structures affect their function...
- Structural Classification of ProteinsStructural Classification of ProteinsThe Structural Classification of Proteins database is a largely manual classification of protein structural domains based on similarities of their structures and amino acid sequences. A motivation for this classification is to determine the evolutionary relationship between proteins...
(SCOP) - CATHCATHThe CATH Protein Structure Classification is a semi-automatic, hierarchical classification of protein domains published in 1997 by Christine Orengo, Janet Thornton and their colleagues....
External links
- The Protein Families (Pfam) database clan browser provides easy access to information about protein structural domains. A clan contains two or more Pfam families that have arisen from a single evolutionary origin.
Structural domain databases
- Conserved Domains at the National Center for Biotechnology website
- 3Dee
- CATH
- DALI
- SCOP
- Pawson Lab - Protein interaction domains
- Nash Lab - Protein interaction domains in Signal Transduction
- Definition and assignment of structural domains in proteins.
Sequence domain databases
- InterPro
- Pfam
- PROSITE
- ProDom
- SMART
- NCBI Conserved Domain Database
- SUPERFAMILY Library of HMMs representing superfamilies and database of (superfamily and family) annotations for all completely sequenced organisms

