
Shapiro reaction
Encyclopedia
The Shapiro reaction or tosylhydrazone decomposition is an organic reaction
in which a ketone
or aldehyde
is converted to an alkene
through an intermediate hydrazone
in the presence of 2 equivalents of strong base
. The reaction was discovered by Robert H. Shapiro
in 1975. The Shapiro reaction was used in the Nicolaou Taxol total synthesis
.

or an aldehyde
is reacted with p-toluenesulfonylhydrazide to a p-toluenesulfonylhydrazone (or tosylhydrazone
) which is an imine
or hydrazone
. Two equivalents of a strong base
such as n-butyllithium
then abstract first the hydrazone proton
and then the less acidic α carbonyl proton leaving a carbanion
. The carbanion proceeds in an elimination reaction
creating the carbon to carbon double bond
and converting the hydrazone group into a lithium diazonium
group. This group expels nitrogen
leaving a vinyllithium compound.
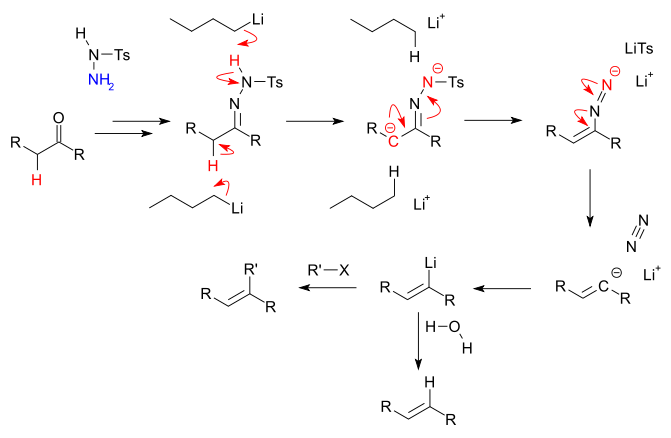
, in the Shapiro reaction in particular water
is added, resulting in hydrolysis to the alkene
. Other reactions of vinyllithium compounds include alkylation reactions with for instance alkyl halides.
 Importantly, the Shapiro reaction cannot be used to synthesize 1-lithioalkenes (and the resulting functionalized derivatives), as sulfonylhydrazones derived from aldehydes undergo exclusive addition of the organolithium base to the carbon of the C–N double bond.
Importantly, the Shapiro reaction cannot be used to synthesize 1-lithioalkenes (and the resulting functionalized derivatives), as sulfonylhydrazones derived from aldehydes undergo exclusive addition of the organolithium base to the carbon of the C–N double bond.
The Bamford-Stevens reaction
is also a tosylhydrazone decomposition but in this reaction a much weaker base can only abstract the hydrazone proton and not the α carbonyl proton and the reaction mechanism
now involves a carbene
or in a protic solvent
a carbocation
.
Organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, photochemical reactions and redox reactions. In organic synthesis,...
in which a ketone
Ketone
In organic chemistry, a ketone is an organic compound with the structure RCR', where R and R' can be a variety of atoms and groups of atoms. It features a carbonyl group bonded to two other carbon atoms. Many ketones are known and many are of great importance in industry and in biology...
or aldehyde
Aldehyde
An aldehyde is an organic compound containing a formyl group. This functional group, with the structure R-CHO, consists of a carbonyl center bonded to hydrogen and an R group....
is converted to an alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
through an intermediate hydrazone
Hydrazone
Hydrazones are a class of organic compounds with the structure R1R2C=NNH2. They are related to ketones and aldehydes by the replacement of the oxygen with the NNH2 functional group...
in the presence of 2 equivalents of strong base
Base (chemistry)
For the term in genetics, see base A base in chemistry is a substance that can accept hydrogen ions or more generally, donate electron pairs. A soluble base is referred to as an alkali if it contains and releases hydroxide ions quantitatively...
. The reaction was discovered by Robert H. Shapiro
Robert H. Shapiro
Robert H. Shapiro was an American chemist. Shapiro was educated at the Stanford University where he received his MS and Ph. D in 1923. In 1965 he became professor at the University of Colorado at Boulder, and from 1980 until his retirement in 1989 he worked at the James Madison University...
in 1975. The Shapiro reaction was used in the Nicolaou Taxol total synthesis
Nicolaou Taxol total synthesis
The Nicolaou Taxol total synthesis, published by K. C. Nicolaou and his group in 1994 concerns the total synthesis of Taxol. This organic synthesis was included in Nicolaou's book, 'Classics in Total Synthesis'....
.

Reaction mechanism
In a prelude to the actual Shapiro reaction a ketoneKetone
In organic chemistry, a ketone is an organic compound with the structure RCR', where R and R' can be a variety of atoms and groups of atoms. It features a carbonyl group bonded to two other carbon atoms. Many ketones are known and many are of great importance in industry and in biology...
or an aldehyde
Aldehyde
An aldehyde is an organic compound containing a formyl group. This functional group, with the structure R-CHO, consists of a carbonyl center bonded to hydrogen and an R group....
is reacted with p-toluenesulfonylhydrazide to a p-toluenesulfonylhydrazone (or tosylhydrazone
Tosylhydrazone
A tosylhydrazone in organic chemistry is a functional group with the general structure RR'C=NH-Ts where Ts is a tosyl group. Organic compounds having this functional group can be accessed by reaction of an aldehyde or ketone with tosylhydrazine.-Synthesis:...
) which is an imine
Imine
An imine is a functional group or chemical compound containing a carbon–nitrogen double bond, with the nitrogen attached to a hydrogen atom or an organic group. If this group is not a hydrogen atom, then the compound is known as a Schiff base...
or hydrazone
Hydrazone
Hydrazones are a class of organic compounds with the structure R1R2C=NNH2. They are related to ketones and aldehydes by the replacement of the oxygen with the NNH2 functional group...
. Two equivalents of a strong base
Base (chemistry)
For the term in genetics, see base A base in chemistry is a substance that can accept hydrogen ions or more generally, donate electron pairs. A soluble base is referred to as an alkali if it contains and releases hydroxide ions quantitatively...
such as n-butyllithium
N-Butyllithium
n-Butyllithium is an organolithium reagent. It is widely used as a polymerization initiator in the production of elastomers such as polybutadiene or styrene-butadiene-styrene...
then abstract first the hydrazone proton
Hydron (chemistry)
In chemistry, a hydron is the general name for a cationic form of atomic hydrogen : most commonly a "proton". However, hydron includes cations of hydrogen regardless of their isotopic composition: thus it refers collectively to protons , deuterons , and tritons...
and then the less acidic α carbonyl proton leaving a carbanion
Carbanion
A carbanion is an anion in which carbon has an unshared pair of electrons and bears a negative charge usually with three substituents for a total of eight valence electrons. The carbanion exists in a trigonal pyramidal geometry. Formally a carbanion is the conjugate base of a carbon acid.where B...
. The carbanion proceeds in an elimination reaction
Elimination reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism...
creating the carbon to carbon double bond
Double bond
A double bond in chemistry is a chemical bond between two chemical elements involving four bonding electrons instead of the usual two. The most common double bond, that between two carbon atoms, can be found in alkenes. Many types of double bonds between two different elements exist, for example in...
and converting the hydrazone group into a lithium diazonium
Diazonium compound
Diazonium compounds or diazonium salts are a group of organic compounds sharing a common functional group R-N2+ X- where R can be any organic residue such alkyl or aryl and X is an inorganic or organic anion such as a halogen...
group. This group expels nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
leaving a vinyllithium compound.

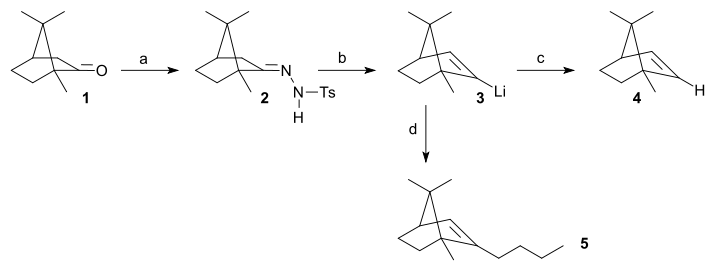
Scope
The position of the alkene in the product is controlled by the site of deprotonation by the organolithium base. In general, the kinetically favored, less substituted site of differentially substituted tosylhydrazones is deprotonated selectively, leading to the less substiuted vinyllithium intermediate. Although many secondary reactions exist for the vinyllithium functional groupFunctional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
, in the Shapiro reaction in particular water
Water
Water is a chemical substance with the chemical formula H2O. A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at ambient conditions, but it often co-exists on Earth with its solid state, ice, and gaseous state . Water also exists in a...
is added, resulting in hydrolysis to the alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
. Other reactions of vinyllithium compounds include alkylation reactions with for instance alkyl halides.

The Bamford-Stevens reaction
Bamford-Stevens reaction
The Bamford–Stevens reaction is a chemical reaction whereby treatment of tosylhydrazones with strong base gives alkenes. It is named for the British chemist William Randall Bamford and the Scottish chemist Thomas Stevens Stevens...
is also a tosylhydrazone decomposition but in this reaction a much weaker base can only abstract the hydrazone proton and not the α carbonyl proton and the reaction mechanism
Reaction mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs.Although only the net chemical change is directly observable for most chemical reactions, experiments can often be designed that suggest the possible sequence of steps in...
now involves a carbene
Carbene
In chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is RR'C:, but the carbon can instead be double-bonded to one group. The term "carbene" may also merely refer to the compound H2C:, also called...
or in a protic solvent
Protic solvent
In chemistry a protic solvent is a solvent that has a hydrogen atom bound to an oxygen or a nitrogen . In general terms, any molecular solvent that contains dissociable H+ is called a protic solvent. The molecules of such solvents can donate an H+...
a carbocation
Carbocation
A carbocation is an ion with a positively-charged carbon atom. The charged carbon atom in a carbocation is a "sextet", i.e. it has only six electrons in its outer valence shell instead of the eight valence electrons that ensures maximum stability . Therefore carbocations are often reactive,...
.

